Abstract
Three iridium(III) polypyridyl complexes [Ir(ppy)2(PYTA)](PF6) (1) (ppy = 2-phenylpyridine), [Ir(bzq)2(PYTA)](PF6) (2) (bzq = benzo[h]quinolone) and [Ir(piq)2(PYTA)](PF6) (3) (piq = 1-phenylisoquinoline, PYTA = 2,4-diamino-6-(2′-pyridyl)-1,3,5-triazine) were synthesized and characterized by elemental analysis, IR, 1H NMR and 13C NMR. The cytotoxic activity of the complexes toward cancer SGC-7901, Eca-109, A549, HeLa, HepG2, BEL-7402 and normal LO2 cell lines was investigated by 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) method. Complex 3 shows the most effective on inhibiting the above cell growth among these complexes. The complexes locate at the lysosomes and mitochondria. AO/EB, Annex V and PI and comet assays indicate that the complexes can induce apoptosis in SGC-7901 cells. Intracellular ROS and mitochondrial membrane potential were examined under fluorescence microscopy. The results demonstrate that the complexes increase the intracellular ROS levels and induce a decrease in the mitochondrial membrane potential. The complexes can enhance intracellular Ca2+ concentration and cause a release of cytochrome c. The autophagy was studied using MDC staining and western blot. Complexes 1–3 can effectively inhibit the cell invasion with a concentration-dependent manner. Additionally, the complexes target tubules and inhibit the polymerization of tubules. The antimicrobial activity of the complexes against S. aureus, E. coli, Salmonella and L. monocytogenes was explored. The mechanism shows that the complexes induce apoptosis in SGC-7901 cells through ROS-mediated lysosomal–mitochondrial, targeting tubules and damage DNA pathways.
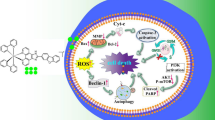
Graphical Abstract
Three iridium(III) complexes [Ir(N–C)2(PYTA)](PF6) (N–C = ppy, 1; bzq, 2; piq, 3) were synthesized and characterized. The anticancer activity of the complexes against SGC-7901 cells was studied by apoptosis, comet assay, autophagy, ROS, mitochondrial membrane potential, intracellular Ca2+ levels, release of cytochrome c, tubules and western blot analysis. The antibacterial activity in vitro was also assayed.














Similar content being viewed by others
References
Mjos KD, Orvig C (2014) Chem Rev 114:4540–4563
Muhammad N, Guo Z (2014) Curr Opin Chem Biol 19:144–153
Komeda S, Casini A (2012) Curr Top Med Chem 12:219–235
Zhang X, Wang F, Zhang C, Wu S, Zheng X, Gong T, Ding R, Chen K, Bai D (2018) Inorg Chem Commun 94:92–97
Liu Z, Romero-Canel I, Qamar B, Hearn JM, Habtemariam A, Barry NPE, Pizarro AM, Clarkson GJ, Sadler PJ (2014) Angew Chem Int Ed 53:4022–4027
Liu Z, Sadler PJ (2016) Accounts Chem Res 47:1174–1185
Leung CH, Zhong HJ, Chan DSH, Ma DL (2013) Coord Chem Rev 257:1764–1776
Barry NPE, Sadler PJ (2012) Chem Soc Rev 41:3264–3279
Sch€afer S, Sheldrick WS (2007) J Organomet Chem 692:1300–1309
Wilbuer Danielle A, Vlecken DH, Schmitz DJ, Kr€aling K, Harms K, Bagowski CP, Meggers E (2010) Angew Chem Int Ed 49:3839–3842
Kastl A, Wilbuer A, Merkel AL, Feng L, Fazio PD, Ocker M, Meggers E (2011) Chem Commun 48:1863–1865
Li Y, Tan CP, Zhang W, He L, Ji LN, Mao ZW (2015) Biomaterials 39:95–104
Ye RR, Tan CP, Ji LN, Mao ZW (2016) Dalton Trans 45:13042–13051
He L, Li Y, Tan CP, Ye RR, Ji LN, Mao ZW (2015) Chem Sci 6:5409–5418
Tang B, Wan D, Wang YJ, Yi QY, Guo BH, Liu YJ (2018) Eur J Med Chem 145:302–314
Yi QY, Wan D, Tang B, Wang YJ, Zhang WY, Du F, He M, Liu YJ (2018) Eur J Med Chem 145:338–349
Zhang WY, Yi QY, Wang YJ, Du F, He M, Tang B, Wan D, Liu YJ, Huang HL (2018) Eur J Med Chem 151:568–584
Wan D, Tang B, Wang YJ, Guo BH, Yin H, Yi QY, Liu YJ (2017) Eur J Med Chem 139:180–190
Song XD, Kong X, He SF, Chen JX, Sun J, Chen BB, Zhao JW, Mao ZW (2017) Eur J Med Chem 138:246–254
Mou ZD, Deng N, Zhang F, Zhang JY, Cen J, Zhang X (2017) Eur J Med Chem 138:72–82
Zhang C, Lai SH, Yang HH, Xing DG, Zeng CC, Tang B, Wan D, Liu YJ (2017) RSC Adv 7:17752–17762
Henze K, Martin W (2012) Nature 426:127–128
McBride HM, Neuspiel M, Wasiak S (2006) Curr Biol 16:R551–R560
Youle RJ, Narendra DP (2011) Nat Rev Mol Cell Biol 12:9–14
Zeng L, Chen Y, Liu J, Huang H, Guan R, Ji L, Chao H (2016) Sci Rep 6:19449
Repnik U, Turk B (2010) Mitochondrion 10:662–669
Reich M, Van Swieten PF, Sommandas V, Kraus M, Fischer R, Weber E, Kalbacher H, Overkleeft HS, Driessen C (2007) J Leukoc Biol 81:990–1001
Guicciardi ME, Leist M, Gores GJ (2004) Oncogene 23:2881–2890
Ghosh M, Carlsson F, Laskar A, Yuan X, Li W (2011) FEBS Lett 585:623–629
Verhey KJ, Gaertig J (2007) Cell Cycle 6:2152–2160
Lin GJ, Jiang GB, Xie YY, Huang HL, Liang ZH, Liu YJ (2013) J Biol Inorg Chem 18:873–882
Case FH (1968) J Heterocycl Chem 5:223–226
Sprouse S, King KA, Spellane PJ, Watts RJ (1984) J Am Chem Soc 106:6647–6653
Mosmann T (1983) J Immunol Methods 65:55–63
Case FH (1968) J Heterocycl Chem 5:223–226
Boya P, Kroemer G (2008) Oncogene 27:6434–6451
Chang Y, Li Y, Ye N, Guo X, Li Z, Sun G, Sun Y (2016) Apoptosis 21:977–996
Rin Jean S, Tulumello DV, Wisnovsky SP, Lei EK, Pereira MP, Kelley SO (2014) ACS Chem Biol 9:323–333
Ly JD, Grubb D, Lawen A (2003) Apoptosis 8:115–128
Lee S, Chen X (2011) Chem Bio Chem 12:2120–2121
Chen LB (1988) Annu Rev Cell Biol 4:155–181
Nicholls DG, Ward MW (2000) Trends Neurosci 3:166–174
Trachootham D, Alexandre J, Huang P (2009) Nat Rev Drug Discov 8:579–591
Ma XW, Zhang LH, Wang LR, Xue X, Sun JH, Wu Y, Zou GZ, Wu X, Wang PC, Wamer WG, Yin JJ, Zheng KY, Liang XJ (2012) ACS Nano 6:10486–10496
Carew JS, Nawrocki ST, Cleveland JL (2007) Autophagy 3:464–467
Biederbick A, Kern HF, Elsasser HP (1995) Eur J Cell Biol 66:3–14
Doonan F, Cotter TG (2008) Methods 44:200–204
Bachand GD, Jain R, Ko R, Bouxsein NF, Van Delinder V (2018) Biomacromol 19:2401–2408
Yuan ZG, Chen SP, Chen CJ, Chen JW, Chen CK, Dai QZ, Gao CM, Jiang YY (2017) Eur J Med Chem 138:1135–1146
Abdel-Rahman LH, Abu-Dief AM, Moustafa H, Hamdan SK (2017) Appl Organomet Chem 31:e3555
Ravi V, Kamakshi D, Praveen KY, Vinoda RM, Rajender RM, Nagamani C, Ravi Ch, Suman ST, Mohan R, Satyanarayana S (2018) New J Chem 42:846–859
Kumar VR, Nagababu P, Srinivas G, Rajender MR, Rani MV, Ravi M, Satyanarayana S (2017) J Coord Chem 70:3790–3809
Acknowledgements
This work was supported by the National Nature Science Foundation of China (No 21877018) and the Natural Science Foundation of Guangdong Province (No 2016A030313728).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yi, QY., Zhang, WY., He, M. et al. Anticancer and antibacterial activity in vitro evaluation of iridium(III) polypyridyl complexes. J Biol Inorg Chem 24, 151–169 (2019). https://doi.org/10.1007/s00775-018-1635-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-018-1635-8