Abstract
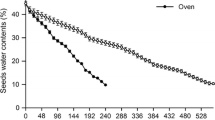
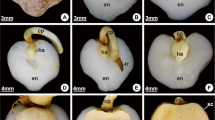
The germination and post-seminal development of Arecaceae are notably complex due to the microscopic dimensions of the embryonic axis, the occurrence of dormancy, and the diversity of reserve compounds. In-depth information on this subject is still limited, especially in terms of the basal sub-family Calamoideae. Mauritiella armata is widely distributed in the Amazon region and is considered a key species in flooded ecosystems (veredas) in the Cerrado biome. We sought to describe histogenesis and reserve compound dynamics during the germination of M. armata, as well as the changes in incubated seeds over time. Seeds with their operculum removed (the structure that limits embryonic growth) were evaluated during germination using standard methods of histology, histochemistry, and electron microscopy. Evaluations were also performed on intact seeds incubated for 180 days. The embryos show characteristics associated with recalcitrant seeds of Arecaceae: a high water content (>80%), differentiated vessel elements, and reduced lipid reserves. Both the embryo and endosperm store abundant reserves of proteins, neutral carbohydrates, and pectins. The completion of germination involves cell divisions and expansions in specific regions of the embryo, in addition to the mobilization of embryonic and endospermic reserves through symplastic and apoplastic flows. Intact seeds show dormancy (not germinating for 180 days), but exhibit continuous development associated with cell growth, differentiation, and reserve mobilization. The anatomical and histochemical characters of M. armata seeds indicate an association between recalcitrance and dormancy related to the species’ adaptation to flooded environments.














Similar content being viewed by others
References
Ávila MA, Azevedo IFP, Antunes JR, Souza CR, Santos RM, Fonseca RS, Nunes YRF (2022) Temperature as the main factor affecting the reproductive phenology of the dioecious palm Mauritiella armata (Arecaceae). Acta Botan Brasil 36:1–12. https://doi.org/10.1590/0102-33062021abb0111
Ávila MAD, Nunes YRF, Souza CS, Machado ADO, Mazzottini-dos-Santos HC, Ribeiro LM, Santos RMD, De Azevedo IFP (2023) Local environment contributes to shape phenological patterns in Mauritia flexuosa L.f. For Ecol Manage 545:121252. https://doi.org/10.1016/j.foreco.2023.121252
Baker WJ, Couvreur TLP (2013) Global biogeography and diversification of palms sheds light on the evolution of tropical lineages. I. Historical biogeography. J Biogeogr 40:274–285. https://doi.org/10.1111/j.1365-2699.2012.02795.x
Baskin CC, Baskin JM (2014) Seeds: ecology, biogeography, and evolution of dormancy and germination. Elsevier/AP, San Diego
Baskin JM, Baskin CC (2014) What kind of seed dormancy might palms have? Seed Sci Res 24:17–22. https://doi.org/10.1017/S0960258513000342
Baskin JM, Baskin CC (2021) The great diversity in kinds of seed dormancy: a revision of the Nikolaeva-Baskin classification system for primary seed dormancy. Seed Sci Res 31:249–277. https://doi.org/10.1017/S096025852100026X
Berjak P, Pammenter NW (2000) What ultrastructure has told us about recalcitrant seeds. Rev Bras Fisiol Veg 12:22–55
Berjak P, Pammenter NW (2008) From Avicennia to Zizania: seed recalcitrance in perspective. Ann Bot 101:213–228
Berjak P, Pammenter NW (2008) Perspectives on seed recalcitrance. S Afr J Bot 74:361. https://doi.org/10.1016/j.sajb.2008.01.032
Bewley JD, Bradford KJ, Hilhorst HWM, Nonogaki H (2013) Seeds: physiology of development, germination and dormancy, 3rd edn. Springer, New York
Brasil (Ministério da Agricultura Pecuária e Abastecimento) (2009) Regras para análise de sementes. Mapa/ACS, Brasília, DF
Carvalho VS, Ribeiro LM, Lopes PSN et al (2015) Dormancy is modulated by seed structures in palms of the cerrado biome. Aust J Bot 63:444. https://doi.org/10.1071/BT14224
da Neves SC, Ribeiro LM, da Cunha IRG et al (2013) Diaspore structure and germination ecophysiology of the babassu palm (Attalea vitrivir). Flora 208:68–78. https://doi.org/10.1016/j.flora.2012.12.007
DeMason DA (1984) Growth parameters in the cotyledon of date seedlings. Bot Gaz 145:176–183. https://doi.org/10.1086/337444
DeMason DA, Thomson WW (1981) Structure and ultrastructure of the cotyledon of date palm (Phoenix dactylifera L.). Bot Gaz 142:320–328. https://doi.org/10.1086/337230
DeMason DA, Sexton R, Gorman M, Reid JSG (1985) Structure and biochemistry of endosperm breakdown in date palm (Phoenix dactylifera L.) seeds. Protoplasma 126:159–167. https://doi.org/10.1007/BF01281791
Dias DS, Ribeiro LM, Lopes PSN et al (2018) Haustorium–endosperm relationships and the integration between developmental pathways during reserve mobilization in Butia capitata (Arecaceae) seeds. Ann Bot 122:267–277. https://doi.org/10.1093/aob/mcy065
Dias GP, Mazzottini-dos-Santos HC, Ribeiro LM et al (2020) Reserve mobilization dynamics and degradation pattern of mannan-rich cell walls in the recalcitrant seed of Mauritia flexuosa (Arecaceae). Plant Physiol Biochem 156:445–460. https://doi.org/10.1016/j.plaphy.2020.09.03
Donohue K, Rubio de Casas R, Burghardt L, Kovach K, Willis CG (2010) Germination, postgermination adaptation, and species ecological ranges. Annu Rev Ecol Evol Syst 41:293–319. https://doi.org/10.1146/annurev-ecolsys-102209-144715
Dransfield J, Uhl NW, Asmussen CBA, Baker WJ, Harley MM, Lewis CE (2008) Genera Palmarum: the evolution and classification of palms, New. Kew Publ, Royal Botanical Garden, Kew
Feder N, O’Brien TP (1968) Plant microtechnique: some principles and new methods. Am J Bot 55:123. https://doi.org/10.2307/2440500
Ferreira CD, de Silva-Cardoso IMA, Ferreira JCB et al (2020) Morphostructural and histochemical dynamics of Euterpe precatoria (Arecaceae) germination. J Plant Res 133:693–713. https://doi.org/10.1007/s10265-020-01219-7
Finch-Savage WE, Leubner-Metzger G (2006) Seed dormancy and the control of germination: Tansley review. New Phytol 171:501–523. https://doi.org/10.1111/j.1469-8137.2006.01787.x
Haccius B, Philip VJ (1979) Embryo development in Cocos nucifera L.: a critical contribution to a general understanding of palm embryogenesis. Plant Syst Evol 132:91–106. https://doi.org/10.1007/BF00983086
Henderson FM (2006) Morphology and anatomy of palm seedlings. Bot. Rev. 72:273–329
Hong TD, Ellis RH (1996) A protocol to determine seed storage behaviour. In: Engels JMM, Toll J (eds) IPGRI Technical Bulletin No. 1. International Plant Genetic Resources Institute, Rome
Karnovsky MJA (1965) A formaldehyde-glutaraldehyde fixate of high osmolality for use in electron microscopy. J Cell Biol 27:137A – 138a
Kirk PW (1970) Neutral red as a lipid fluorochrome. Stain Technol 45:1–4. https://doi.org/10.3109/10520297009063373
Lorenzi H, Noblick LR, Kahn F, Ferreira E (2010) Flora Brasileira – Arecaceae (palmeiras). Instituto Plantarum, Nova Odessa
Marques A, Buijs G, Ligterink W, Hilhorst H (2018) Evolutionary ecophysiology of seed desiccation sensitivity. Funct Plant Biol 45:1083. https://doi.org/10.1071/FP18022
Mazzottini-dos-Santos HC, Ribeiro LM, Oliveira DMT (2017) Roles of the haustorium and endosperm during the development of seedlings of Acrocomia aculeata (Arecaceae): dynamics of reserve mobilization and accumulation. Protoplasma 254:1563–1578. https://doi.org/10.1007/s00709-016-1048-x
Mazzottini-dos-Santos HC, Ribeiro LM, Oliveira DMT (2018) Structural changes in the micropylar region and overcoming dormancy in Cerrado palms seeds. Trees 32:1415–1428. https://doi.org/10.1007/s00468-018-1723-y
Mazzottini-dos-Santos HC, Ribeiro LM, Oliveira DMT, Paiva EAS (2020) Ultrastructural aspects of metabolic pathways and translocation routes during mobilization of seed reserves in Acrocomia aculeata (Arecaceae). Braz J Bot 43:589–600. https://doi.org/10.1007/s40415-020-00622-7
Moura ACF, Ribeiro LM, Mazzottini-dos-Santos HC et al (2019) Cytological and histochemical evaluations reveal roles of the cotyledonary petiole in the germination and seedling development of Mauritia flexuosa (Arecaceae). Protoplasma 256:1299–1316. https://doi.org/10.1007/s00709-019-01375-1
Nunes YRF, Souza CS, Azevedo IFPD et al (2022) Vegetation structure and edaphic factors in veredas reflect different conservation status in these threatened areas. For Ecosyst 9:100036. https://doi.org/10.1016/j.fecs.2022.100036
O’Brien TP, Feder N, McCully ME (1964) Polychromatic staining of plant cell walls by toluidine blue O. Protoplasma 59:368–373
Oliveira NCC, Lopes PSN, Ribeiro LM et al (2013) Seed structure, germination, and reserve mobilization in Butia capitata (Arecaceae). Trees 27:1633–1645. https://doi.org/10.1007/s00468-013-0910-0
Orozco-Segovia A, Batis AI, Rojas-Aréchiga M, Mendonza A (2003) Seed biology of palms: a review. Palms 47:79–74
Paiva EAS (2021) Do calcium oxalate crystals protect against herbivory? Sci Nat 108:24. https://doi.org/10.1007/s00114-021-01735-z
Paiva EAS, Pinho SZ, Oliveira DMT (2011) Large plant samples: how to process for GMA embedding? In: Melo RCN (ed) Choarini-Garcia H. Methods and Protocols. Humana Press Totowa, Light Microscopy, pp 37–49
Panza V, Láinez V, Maldonado S (2004) Seed structure and histochemistry in the palm Euterpe edulis. Bot J Linn Soc 145:445–453. https://doi.org/10.1111/j.1095-8339.2004.00293.x
Reis SB, Mello ACMP, Oliveira DMT (2017) Pericarp formation in early divergent species of Arecaceae (Calamoideae, Mauritiinae) and its ecological and phylogenetic importance. Plant Syst Evol 303:675–687. https://doi.org/10.1007/s00606-017-1399-6
Reynolds ES (1963) The use of lead citrate at high ph as an electron-opaque stain in electron microscopy. J Cell Biol 17:208–212. https://doi.org/10.1083/jcb.17.1.208
Ribeiro LM, Souza PP, Rodrigues AG Jr et al (2011) Overcoming dormancy in macaw palm diaspores, a tropical species with potential for use as bio-fuel. Seed Sci Technol 39:303–317. https://doi.org/10.15258/sst.2011.39.2.04
Ribeiro LM, Oliveira DMT, de Garcia Q, S, (2012) Structural evaluations of zygotic embryos and seedlings of the macaw palm (Acrocomia aculeata, Arecaceae) during in vitro germination. Trees 26:851–863. https://doi.org/10.1007/s00468-011-0659-2
Ribeiro LM, Garcia QS, Müller M, Munné-Bosch S (2015) Tissue-specific hormonal profiling during dormancy release in macaw palm seeds. Physiol Plantarum 153:627–642. https://doi.org/10.1111/ppl.12269
Robards AW (1978) An introduction to techniques for scanning electron micoscopy of plant cells. In: Hall JL (ed) Electron microscopy and cytochemistry of plant cells. Elsevier, New York, pp 343–403
Rocha, (2022) Crescimento geotrópico positivo do pecíolo cotiledonar em Butia capitata e implicações ecológicas e filogenéticas em palmeiras. Universidade Estadual de Montes Claros, Dissertação
Roland AM (1978) General preparations and staining of thin sections. In: Hall JL (ed) Electron microscopy and cytochemistry of plant cells. Elsevier, New York, pp 1–62
Salvador HF, Mazzottini-dos-Santos HC, Dias DS et al (2022) The dynamics of Mauritia flexuosa (Arecaceae) recalcitrant seed banks reveal control of their persistence in marsh environments. For Ecol Manage 511:120155. https://doi.org/10.1016/j.foreco.2022.120155
Silva RS, Ribeiro LM, Mercadante-Simões MO et al (2014) Seed structure and germination in buriti (Mauritia flexuosa), the Swamp palm. Flora 209:674–685. https://doi.org/10.1016/j.flora.2014.08.012
Smith N (2015) Mauritiella armata. Palms and people in the Amazon. Springer International Publishing, Cham, pp 383–389
Souza JN, Ribeiro LM, Mercadante-Simões MO (2016) Ontogenesis and functions of saxophone stem in Acrocomia aculeata (Arecaceae). Ann Bot 119:353–365. https://doi.org/10.1093/aob/mcw215
Souza AT, Pereira Junio RF, de Neuba L, M, et al (2020) Caranan fiber from Mauritiella armata palm tree as novel reinforcement for epoxy composites. Polymers 12:2037. https://doi.org/10.3390/polym12092037
Souza FG, De Araújo FF, Orlando EA et al (2022) Characterization of buritirana (Mauritiella armata) fruits from the Brazilian Cerrado: biometric and physicochemical attributes, chemical composition and antioxidant and antibacterial potential. Foods 11:786. https://doi.org/10.3390/foods11060786
Steinbrecher T, Leubner-Metzger G (2017) The biomechanics of seed germination. J Exp Bot 68:765e783. https://doi.org/10.1093/jxb/erw428
Tweddle JC, Dickie JB, Baskin CC, Baskin JM (2003) Ecological aspects of seed desiccation sensitivity. J Ecol 91:294–304. https://doi.org/10.1046/j.1365-2745.2003.00760.x
Veloso VHS, Ribeiro LM, Mercadante-Simões MO, Nunes YRF (2016) Cytological aspects of recalcitrance in dormant seeds of Mauritia flexuosa (Arecaceae). Acta Physiol Plant 38:171. https://doi.org/10.1007/s11738-016-2194-7
Vidal BC (1970) Dichroism in collagen bundles stained with Xylidine-Ponceau 2R. Ann Histochim 15:289–296
Visscher AM, Castillo-Lorenzo E, Toorop PE, Junio Da Silva L, Yeo M, Pritchard HW (2020) Pseudophoenix ekmanii (Arecaceae) seeds at suboptimal temperature show reduced imbibition rates and enhanced expression of genes related to germination inhibition. Plant Biol J 22(6):1041–1051. https://doi.org/10.1111/plb.13156
Watson ML (1958) Staining of tissue sections for electron microscopywith heavy metals II. Application of solutions containing lead andbarium. J Biophys Biochem Cytol 4:727–730
Weis KG, Polito VS, Labavitch JM (1988) Microfluorometry of pectic materials in the dehiscence zone of almond (Prunus dulcis [Mill.] DA Webb) fruits. J Histochem Cytochem 36:1037–1041. https://doi.org/10.1177/36.8.3392393
Zona S (2004) Raphides in palm embryos and their systematic distribution. Ann Bot 93:415–421. https://doi.org/10.1093/aob/mch060
Acknowledgements
The authors thank Dr. Sarah Barbosa Reis for reviewing the text and the Centro de Microscopia da Universidade Federal de Minas Gerais - CM/UFMG for making its infrastructure available for the electron microscopy analyses.
Funding
This research was supported by the Long Term Ecological Research Network (Programa de Pesquisa Ecológica de Longa Duração – PELD-VERE) of the National Council for Scientific and Technological Development – CNPq (Edital 093/2021); the Fundação de Amparo à Pesquisa do Estado de Minas Gerais – FAPEMIG (APQ-03371-21); and the Ministry of Science, Technology and Innovations – MCTI, Brazil. The authors are grateful to FAPEMIG and CNPq for the master’s scholarship granted to PPF, and to CNPq for the research productivity scholarships granted to LMR and YRFN.
Author information
Authors and Affiliations
Contributions
LMR, HCM-S, IFPA, and YRFN idealized and planned the research. PPF collected and processed the plant material, prepared anatomical slides and electronic microscopy samples, and wrote the initial text. LMR and PPF performed the anatomical analyses. HCM-S analyzed histochemical and electron microscopy data. LMR, PPF, and HCM-S elaborated the final text. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Peter Nick.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fonseca, P.P., Mazzottini-dos-Santos, H.C., de Azevedo, I.F.P. et al. Histogenesis and reserve dynamics during the maintenance of dormancy and germination in seeds of the basal palm Mauritiella armata. Protoplasma 261, 425–445 (2024). https://doi.org/10.1007/s00709-023-01905-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-023-01905-y