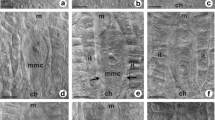
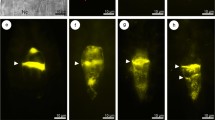
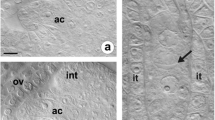
Abstract
Callose (β-1,3-glucan) is one of the cell wall polymers that plays an important role in many biological processes in plants, including reproductive development. In angiosperms, timely deposition and degradation of callose during sporogenesis accompanies the transition of cells from somatic to generative identity. However, knowledge on the regulation of callose biosynthesis at specific sites of the megasporocyte wall remains limited and the data on its distribution are not conclusive. Establishing the callose deposition pattern in a large number of species can contribute to full understanding of its function in reproductive development. Previous studies focused on callose events in sexual species and only a few concerned apomicts. The main goal of our research was to establish and compare the pattern of callose deposition during early sexual and diplosporous processes in the ovules of some Hieracium, Pilosella and Taraxacum (Asteraceae) species; aniline blue staining technique was used for this purpose. Our findings indicate that callose deposition accompanies both meiotic and diplosporous development of the megaspore mother cell. This suggests that it has similar regulatory functions in intercellular communication regardless of the mode of reproduction. Interestingly, callose deposition followed a different pattern in the studied sexual and diplosporous species compared to most angiosperms as it usually began at the micropylar pole of the megasporocyte. Here, it was only in sexually reproducing H. transylvanicum that callose first appeared at the chalazal pole of the megasporocyte. The present paper additionally discusses the occurrence of aposporous initial cells with callose-rich walls in the ovules of diploid species.



Similar content being viewed by others
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
Abou-Saleh RH, Hernandez-Gomez MC, Amsbury S, Paniagua C, Bourdon M, Miyashima S, Helariutta Y, Fuller M, Budtova T, Connell SD, Ries ME, Benitez-Alfonso Y (2018) Interactions between callose and cellulose revealed through the analysis of biopolymer mixtures. Nat Commun 9:4538. https://doi.org/10.1038/s41467-018-06820-y
Asker SE, Jerling L (1992) Apomixis in plants. CRC Press, Boca Raton
Barro-Trastoy D, Gomez MD, Tornero P, Perez-Amador MA (2020) On the way to ovules: the hormonal regulation of ovule development. CRC Crit Rev Plant Sci 39:431–456. https://doi.org/10.1080/07352689.2020.1820203
Bencivenga S, Colombo L, Masiero S (2011) Cross talk between the sporophyte and the megagametophyte during ovule development. Sex Plant Reprod 24:113–121. https://doi.org/10.1007/s00497-011-0162-3
Bicknell RA, Koltunow AM (2004) Understanding apomixis: recent advances and remaining conundrums. Plant Cell 16:228–245. https://doi.org/10.1105/tpc.017921
Bilska-Kos A, Solecka D, Dziewulska A, Ochodzki P, Jończyk M, Bilski H, Sowiński P (2017) Low temperature caused modifications in the arrangement of cell wall pectins due to changes of osmotic potential of cells of maize leaves (Zea mays L.). Protoplasma 254:713–724. https://doi.org/10.1007/s00709-016-0982-y
Bräutigam S, Bräutigam E (1996) Determination of the ploidy level in the genus Hieracium subgenus Pilosella (Hill) S.F. Gray by flow cytometric DNA analysis. Folia Geobot Phytotx 31:315–321. https://doi.org/10.1007/BF02815376
Brukhin V, Osadtchiy JV, Florez-Rueda AM, Smetanin D, Bakin E, Margarida Nobre MS, Grossniklaus U (2019) The Boechera genus as a resource for apomixis research. Front Plant Sci 10:392. https://doi.org/10.3389/fpls.2019.00392
Carman JG (1997) Asynchronous expression of duplicate genes in angiosperms may cause apomixis, bispory, tetraspory, and polyembryony. Biol J Linn Soc Lond 61:51–94. https://doi.org/10.1111/j.1095-8312.1997.tb01778.x
Carman JG, Crane CF, Riera-Lizarazu O (1991) Comparative histology of cell walls during meiotic and apomeiotic megasporogenesis in two hexaploid Australasian Elymus species. Crop Sci 31:1527–1532. https://doi.org/10.2135/cropsci1991.0011183X003100060029x
Chen XY, Kim JY (2009) Callose synthesis in higher plants. Plant Signal Behav 4:489–492. https://doi.org/10.4161/psb.4.6.8359
Cheng CY, Mathews DE, Schaller GE, Kieber JJ (2013) Cytokinin-dependent specification of the functional megaspore in the Arabidopsis female gametophyte. Plant J 73:929–940. https://doi.org/10.1111/tpj.12084
Cucinotta M, Colombo L, Roig-Villanova I (2014) Ovule development, a new model for lateral organ formation. Front Plant Sci 5:117. https://doi.org/10.3389/fpls.2014.00117
Delgado L, Galdeano F, Sartor ME, Quarin CL, Espinoza F, Ortiz JPA (2014) Analysis of variation for apomictic reproduction in diploid Paspalum rufum. Ann Bot 113:1211–1218. https://doi.org/10.1093/aob/mcu056
Delgado L, Sartor ME, Espinoza F, Soliman M, Galdeano F, Ortiz JPA (2016) Hybridity and autopolyploidy increase the expressivity of apospory in diploid Paspalum rufum. Plant Syst Evol 302:1471–1481. https://doi.org/10.1007/s00606-016-1345-z
Drews GN, Koltunow AMG (2011) The Female Gametophyte Arabidopsis Book 9:e0155. https://doi.org/10.1199/tab.0155
Dusi DMA, Willemse MTM (1999) Apomixis in Brachiaria decumbens Stapf.: gametophytic development and reproductive calendar. Acta Biol Cracov Bot 41:151–162
Ellinger D, Voigt CA (2014) Callose biosynthesis in Arabidopsis with a focus on pathogen response: what we have learned within the last decade. Ann Botany 114:1349–1358. https://doi.org/10.1093/aob/mcu120
Endress PK (2011) Angiosperm ovules: diversity, development, evolution. Ann Botany 107:1465–1489. https://doi.org/10.1093/aob/mcr120
Fagundes NF, de Araujo Mariath JE (2014) Ovule ontogeny in Billbergia nutans in the evolutionary context of Bromeliaceae (Poales). Plant Syst Evol 300:1323–1336. https://doi.org/10.1007/s00606-013-0964-x
Feng X, Zilberman D, Dickinson H (2013) A conversation across generations: soma-germ cell crosstalk in plants. Dev Cell 24:215–225. https://doi.org/10.1016/j.devcel.2013.01.014
Grossniklaus U (2011) Plant germline development: a tale of cross-talk, signaling, and cellular interactions. Sex Plant Reprod 24:91–95. https://doi.org/10.1007/s00497-011-0170-3
Heslop-Harrison J, Mackenzie A (1967) Autoradiography of soluble [2-14C]thymidine derivatives during meiosis and microsporogenesis in Lilium anthers. J Cell Sci 2:387–400. https://doi.org/10.1242/jcs.2.3.387
Houston K, Tucker MR, Chowdhury J, Shirley N, Little A (2016) The plant cell wall: a complex and dynamic structure as revealed by the responses of genes under stress conditions. Front Plant Sci 7:984. https://doi.org/10.3389/fpls.2016.00984
Humphrey TV, Bonetta DT, Goring DR (2007) Sentinels at the wall: cell wall receptors and sensors. New Phytol 176:7–21. https://doi.org/10.1111/j.1469-8137.2007.02192.x
Ilnicki T, Hasterok R, Szeląg Z (2010) Cytogenetic analysis of Hieracium transylvanicum (Asteraceae). Caryologia 63:192–196. https://doi.org/10.1080/00087114.2010.589726
Janas A, Musiał K, Kościńska-Pająk M, Marciniuk P (2016) Insights into developmental processes in anthers, ovaries, and ovules of Taraxacum belorussicum (Asteraceae-Cichorioideae) using DIC optics. Plant Syst Evol 302:617–628. https://doi.org/10.1007/s00606-016-1288-4
Janas AB, Szeląg Z, Musiał K (2021) In search of female sterility causes in the tetraploid and pentaploid cytotype of Pilosella brzovecensis (Asteraceae). J Plant Res 134:803–810. https://doi.org/10.1007/s10265-021-01290-8
Jane WN, Chiang SHT (1996) Ultrastructure of megasporogenesis and early megagametogenesis in Arundo formosana Hack. (Poaceae). Int J Plant Sci 157:418–431. https://doi.org/10.1086/297358
Keegstra K (2010) Plant cell walls. Plant Physiol 154:483–486. https://doi.org/10.1104/pp.110.161240
Koltunow AM, Johnson SD, Bicknell RA (1998) Sexual and apomictic development in Hieracium. Sex Plant Reprod 11:213–230. https://doi.org/10.1007/s004970050144
Krzesłowska M (2011) The cell wall in plant cell response to trace metals: polysaccharide remodeling and its role in defense strategy. Acta Physiol Plant 33:35–51. https://doi.org/10.1007/s11738-010-0581-z
Lampugnani ER, Khan GA, Somssich M, Persson S (2018) Building a plant cell wall at a glance. J Cell Sci 131:jcs207373. https://doi.org/10.1242/jcs.207373
Leblanc O, Peel MD, Carman JG, Savidan Y (1995) Megasporogenesis and megagametogenesis in several Tripsacum species (Poaceae). Am J Bot 82:57–63. https://doi.org/10.2307/2445787
Li JJ, Liu L, Ouyang YD, Yao JL (2011) Sexual reproduction development in apomictic Eulaliopsis binata (Poaceae). Genet Mol Res 10:2326–2339. https://doi.org/10.4238/2011.October.5.3
Lora J, Herrero M, Tucker MR, Hormaza JI (2017) The transition from somatic to germline identity shows conserved and specialized features during angiosperm evolution. New Phytol 216:495–509. https://doi.org/10.1111/nph.14330
Lora J, Yang X, Tucker MR (2019) Establishing a framework for female germline initiation in the plant ovule. J Exp Bot 70:2937–2949. https://doi.org/10.1093/jxb/erz212
Lovell JT, Aliyu OM, Mau M, Schranz E, Koch M, Kiefer C, Song BH, Mitchell-Olds T, Sharbel TF (2013) On the origin and evolution of apomixis in Boechera. Plant Reprod 26:309–315. https://doi.org/10.1007/s00497-013-0218-7
Marciniuk J, Rerak J, Grabowska-Joachimiak A, Jastrząb I, Musiał K, Joachimiak AJ (2010) Chromosome numbers and stomatal cell length in Taraxacum sect. Palustria from Poland. Acta Biol Cracov Bot 52:117–121. https://doi.org/10.2478/v10182-010-0015-7
Meier M, Zappacosta D, Selva JP, Pessino S, Echenique V (2011) Evaluation of different methods for assessing the reproductive mode of weeping lovegrass plants, Eragrostis curvula (Schrad.) Nees. Aust J Bot 59:253–261. https://doi.org/10.1071/BT10267
Mogie M, Ford H (1988) Sexual and asexual Taraxacum species. Biol J Linn Soc 35:155–168. https://doi.org/10.1111/j.1095-8312.1988.tb00463.x
Musiał K, Kościńska-Pająk M (2017) Pattern of callose deposition during the course of meiotic diplospory in Chondrilla juncea (Asteraceae, Cichorioideae). Protoplasma 254:1499–1505. https://doi.org/10.1007/s00709-016-1039-y
Musiał K, Kościńska-Pająk M (2019) Callose is integral to meiotic diplospory of the Taraxacum type: new evidence from ovules of Chondrilla brevirostris (Asteraceae-Cichorioideae). Bot Lett 166:274–282. https://doi.org/10.1080/23818107.2019.1573378
Musiał K, Szeląg Z (2015) Chromosome numbers in Hieracium (Asteraceae) from Central and Southeastern Europe I. Acta Biol Cracov Bot 57:115–120. https://doi.org/10.1515/abcsb-2015-0020
Musiał K, Kościńska-Pająk M, Antolec R, Joachimiak AJ (2015) Deposition of callose in young ovules of two Taraxacum species varying in the mode of reproduction. Protoplasma 252:135–144. https://doi.org/10.1007/s00709-014-0654-8
Naumova TN, Willemse MTM (1995) Ultrastructural characterization of apospory in Panicum maximum. Sex Plant Reprod 8:192–204. https://doi.org/10.1007/BF00228937
Naumova T, den Nijs APM, Willemse MTM (1993) Quantitative analysis of aposporous parthenogenesis in Poa pratensis genotypes. Acta Bot Neerl 42:299–312. https://doi.org/10.1111/j.1438-8677.1993.tb00707.x
Naumova T, Osadtchiy JV, Sharma VK, Dijkhuis P, Ramulu KS (1999) Apomixis in plants: structural and functional aspects of diplospory in Poa nemoralis and P. palustris. Protoplasma 208:186–195. https://doi.org/10.1007/BF01279089
Noyes RD (2007) Apomixis in the Asteraceae: diamonds in the rough. Funct Plant Sci Biotechnol 1:207–222
Ochoa-Villarreal M, Aispuro-Hernández E, Vargas-Aispuro I, Martínez-Téllez MÁ (2012) Plant cell wall polymers: function, structure and biological activity of their derivatives. In: De Souza Gomes A (ed) Polymerization. IntechOpen, London, United Kingdom, pp 63–86. https://doi.org/10.5772/46094
Okada T, Catanach AS, Johnson SD, Bicknell RA, Koltunow AM (2007) An Hieracium mutant, loss of apomeiosis 1 (loa1) is defective in the initiation of apomixis. Sex Plant Reprod 20:199–211. https://doi.org/10.1007/s00497-007-0057-5
Pagnussat GC, Alandete-Saez M, Bowman JL, Sundaresan V (2009) Auxin-dependent patterning and gamete specification in the Arabidopsis female gametophyte. Science 324:1684–1689. https://doi.org/10.1126/science.1167324
Peel MD, Carman JG, Leblanc O (1997) Megasporocyte callose in apomictic buffelgrass, Kentucky bluegrass, Pennisetum squamulatum Fresen, Tripsacum L., and weeping lovegrass. Crop Sci 37:724–732. https://doi.org/10.2135/cropsci1997.0011183X003700030006x
Pereira EJ, Bittencourt NS (2016) Comparative ovule ontogeny in some members of the Tabebuia alliance (Bignoniaceae). Int J Plant Sci 177:481–497. https://doi.org/10.1086/686794
Piršelová B, Matušíková I (2013) Callose: the plant cell wall polysaccharide with multiple biological functions. Acta Physiol Plant 35:635–644. https://doi.org/10.1007/s11738-012-1103-y
Pogan E, Wcisło H (1989) Cytological investigations on Hieracium pilosella L. from Poland. I. Karyological Studies Acta Biol Cracov Bot 31:19–28
Richmond TA, Somerville CR (2000) The cellulose synthase superfamily. Plant Physiol 124:495–498. https://doi.org/10.1104/pp.124.2.495
Robert HS, Crhak Khaitova L, Mroue S, Benková E (2015) The importance of localized auxin production for morphogenesis of reproductive organs and embryos in Arabidopsis. J Exp Bot 66:5029–5042. https://doi.org/10.1093/jxb/erv256
Rodkiewicz B (1970) Callose in cell wall during megasporogenesis in Angiosperms. Planta 93:39–47. https://doi.org/10.1007/BF00387650
Rodkiewicz B (1973) Callose walls in megaspores in Fuchsia and Epilobium. Caryologia 25(sup1):59–66. https://doi.org/10.1080/00087114.1973.10797113
Rodkiewicz B, Górska-Brylass A (1968) Callose in the walls of the developing megasporocyte and megaspores in the orchid ovule. Acta Soc Bot Pol 37:19–28. https://doi.org/10.5586/asbp.1968.003
Rojek J, Kapusta M, Kozieradzka-Kiszkurno M, Majcher D, Górniak M, Sliwinska E, Sharbel TF, Bohdanowicz J (2018) Establishing the cell biology of apomictic reproduction in diploid Boechera stricta (Brassicaceae). Ann Bot 122:513–539. https://doi.org/10.1093/aob/mcy114
Sak D, Janas A, Musiał K, Płachno BJ (2016) Sexual reproductive traits in tetraploid Pilosella officinarum (Asteraceae, Cichorioideae): DIC microscope study of cleared whole-mount tissue. Acta Biol Cracov Bot 58 suppl:1:90. https://abcbot.pl/pdf/suppl58_1/posters.pdf. Accessed 15 March 2022
Sarkar P, Bosneaga E, Auer M (2009) Plant cell walls throughout evolution: towards a molecular understanding of their design principles. J Exp Bot 60:3615–3635. https://doi.org/10.1093/jxb/erp245
Schinkel CCF, Kirchheimer B, Dellinger AS, Klatt S, Winkler M, Dullinger S, Hörandl E (2016) Correlations of polyploidy and apomixis with elevation and associated environmental gradients in an alpine plant. AoB Plants 8: plw064. https://doi.org/10.1093/aobpla/plw064
Schmidt A, Schmid MW, Grossniklaus U (2015) Plant germline formation: common concepts and developmental flexibility in sexual and asexual reproduction. Development 142:229–241. https://doi.org/10.1242/dev.102103
Schneider R, Hanak T, Persson S, Voight CA (2016) Cellulose and callose synthesis and organization in focus, what’s new? Curr Opin Plant Biol 34:9–16. https://doi.org/10.1016/j.pbi.2016.07.007
Shirley NJ, Aubert MK, Wilkinson LG, Bird DC, Lora J, Yang X, Tucker MR (2019) Translating auxin responses into ovules, seeds and yield: Insight from Arabidopsis and the cereals. J Integr Plant Biol 61:310–336. https://doi.org/10.1111/jipb.12747
Skinner DJ, Hill TA, Gasser CS (2004) Regulation of ovule development. Plant Cell 16:S32–S45. https://doi.org/10.1105/tpc.015933
Szeląg Z, Ilnicki T (2011) Diploid chromosome numbers in Hieracium and Pilosella (Asteraceae) from Macedonia and Montenegro. Acta Biol Cracov Bot 53:124–126. https://doi.org/10.2478/v10182-011-0023-2
Tucker MR, Koltunow AMG (2009) Sexual and asexual (apomictic) seed development in flowering plants: molecular, morphological and evolutionary relationships. Funct Plant Biol 36:490–504. https://doi.org/10.1071/FP09078
Tucker MR, Koltunow AMG (2014) Traffic monitors at the cell periphery: the role of cell walls during female reproductive cell differentiation in plants. Curr Opin Plant Biol 17:137–145. https://doi.org/10.1016/j.pbi.2013.11.015
Tucker MR, Paech NA, Willemse MTM, Koltunow AM (2001) Dynamics of callose deposition and ß-1,3-glucanase expression during reproductive events in sexual and apomictic Hieracium. Planta 212:487–498. https://doi.org/10.1007/s004250000445
Tucker MR, Lou H, Aubert MK, Wilkinson KG, Little A, Houston K, Pinto SC, Shirley NJ (2018) Exploring the role of cell wall-related genes and polysaccharides during plant development. Plants 7:42. https://doi.org/10.3390/plants7020042
Verma DPS, Hong Z (2001) Plant callose synthase complexes. Plant Mol Biol 47:693–701. https://doi.org/10.1023/A:1013679111111
Wędzony M (1996) Fluorescence microscopy for botanists (in Polish). Department of Plant Physiology Monographs 5, Kraków, Poland, pp 64–65
Willemse MTM, Bednara J (1979) Polarity during megasporogenesis in Gasteria verrucosa. Phytomorphology 29:156–165
Wojtaszek P (2000) Genes and plant cell walls: a difficult relationship. Biol Rev Camb Philos Soc 75:437–475. https://doi.org/10.1017/s0006323100005545
Yurukova-Grancharova P, Robeva-Davidova P, Vladimirov V (2006) On the embryology and mode of reproduction of selected diploid species of Hieracium s.l. (Asteraceae) from Bulgaria. Flora 201:668–675. https://doi.org/10.1016/j.flora.2006.01.003
Záveská Drábková L, Honys D (2017) Evolutionary history of callose synthases in terrestrial plants with emphasis on proteins involved in male gametophyte development. PLoS ONE 12:e0187331. https://doi.org/10.1371/journal.pone.018733
Funding
This research was supported by the statutory research funds DS/MND/WBiNoZ/IB/12/2016, DS/MND/WBiNoZ/IB/7/2017, K/ZDS/008057 of the Institute of Botany, Faculty of Biology, Jagiellonian University in Cracow, Poland and partly co-funded by Siedlce University of Natural Sciences and Humanities (grant no. 78/20/B).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. ABJ – design of the study, material preparation, embryological analysis, interpretation of results, preparation of figures, data collection and writing the manuscript; JM – identification and sampling of plant material and proofreading of the manuscript; ZS – identification and sampling of plant material and proofreading of the manuscript; KM – design of the study, interpretation of results and proofreading of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Handling Editor: Dorota Kwiatkowska
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Janas, A.B., Marciniuk, J., Szeląg, Z. et al. New facts about callose events in the young ovules of some sexual and apomictic species of the Asteraceae family. Protoplasma 259, 1553–1565 (2022). https://doi.org/10.1007/s00709-022-01755-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-022-01755-0