Abstract
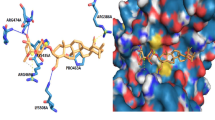
In continuation of our efforts to develop new antiproliferative agents that could be effective and selective in treatment of cancer, we designed and synthesized new hybrid structures containing an arylsulfonamide scaffold linked to a steroidal skeleton through a 1,2,3-triazole ring. Both in vitro cytotoxicity on breast MCF-7 cancer cells and human placental aromatase enzyme (pAROM) inhibition assays were performed on new hybrids. All new hybrids showed marked cytotoxic activity against breast MCF-7 cancer cells (IC50 = 3.56–43.76 μM) in comparison to staurosporine (IC50 = 4.06 μM). Tumor selectivity index was higher for some of the new hybrids on normal fibroblast (F-180) cells (TS = 1.5–25) in comparison to staurosporine (TS = 2.5). The p-nitro derivative exhibited the best inhibitory activity on the pAROM with an IC50 of 64.6 ng/cm3, compared to hybrids unsubstituted derivative, p-bromo derivative, and letrozole (IC50 = 375.14, 269.86, and 132.86 ng/cm3, respectively). Furthermore, the p-nitro hybrid arrested the cell cycle selectively at the G2/M phase, in addition to inducing both early and late apoptotic processes of breast MCF-7 cancer cells. Molecular docking studies were performed within pAROM to explore the binding modes of the new hybrids. Collectively, the antiproliferative profile of new hybrids indicates how good they are as promising leads for developing tumor-specific cytotoxins, and deserve further studies to optimize their structure and in vivo activity.
Graphic abstract












Similar content being viewed by others
References
Hu W, Huang XS, Wu JF, Yang L, Zheng YT, Shen YM, Li ZY, Li X (2018) J Med Chem 61:8947
Erşatır M, Yıldırım M, Giray ES, Yalın S (2020) Monatsh Chem 151:625
Vaidya A, Jain S, Prashantha Kumar B, Singh SK, Kashaw SK, Agrawal RK (2020) Monatsh Chem 151:385
Li Z, Chen Y, Liu Z, Wang Q, Zhao Y, Wei J, Liu M, Wang Z, Li D, Han J (2020) Monatsh Chem 151:353
Acar Cevik U, Saglik BN, Osmaniye D, Levent S, Kaya Cavusoglu B, Karaduman AB, Ozkay Y, Kaplancikli ZA (2020) Arch Pharm 353(5):e2000008
Chia S, Gradishar W, Mauriac L, Bines J, Amant F, Federico M, Fein L, Romieu G, Buzdar A, Robertson JF, Brufsky A, Possinger K, Rennie P, Sapunar F, Lowe E, Piccart M (2008) J Clin Oncol 26:1664
Miller WR, Bartlett JM, Canney P, Verrill M (2007) Breast Cancer Res Treat 103:149
Ghosh D, Griswold J, Erman M, Pangborn W (2010) J Steroid Biochem Mol Biol 118:197
Favia AD, Nicolotti O, Stefanachi A, Leonetti F, Carotti A (2013) Expert Opin Drug Discov 8:395
Macedo LF, Sabnis G, Brodie A (2009) Ann N Y Acad Sci 1155:162
Santen RJ, Brodie H, Simpson ER, Siiteri PK, Brodie A (2009) Endocr Rev 30:343
Espinoza-Fonseca LM (2006) Bioorg Med Chem 14:896
Mencher SK, Wang LG (2005) BMC Clin Pharmacol 5:3
Morphy R, Kay C, Rankovic Z (2004) Drug Discov Today 9:641
Gediya LK, Njar VC (2009) Expert Opin Drug Discov 4:1099
Viegas-Junior CDA, da Silva BV, Barreiro EJ, Fraga CAM (2007) Curr Med Chem 14:1829
Meegan NMO, Ba MJ (2011) Curr Med Chem 18:4722
El-Sheref EM, Aly AA, Ameen MA, Brown AB (2019) Monatsh Chem 150:747
Mahdavi M, Saeedi M, Karimi M, Foroughi N, Hasanshahi F, Alinezhad H, Foroumadi A, Shafiee A, Akbarzadeh T (2016) Monatsh Chem 147:2151
Doiron J, Soultan AH, Richard R, Toure MM, Picot N, Richard R, Cuperlovic-Culf M, Robichaud GA, Touaibia M (2011) Eur J Med Chem 46:4010
Pingaew R, Mandi P, Nantasenamat C, Prachayasittikul S, Ruchirawat S, Prachayasittikul V (2014) Eur J Med Chem 81:192
Pingaew R, Prachayasittikul V, Mandi P, Nantasenamat C, Prachayasittikul S, Ruchirawat S, Prachayasittikul V (2015) Bioorg Med Chem 23:3472
Pingaew R, Mandi P, Prachayasittikul V, Prachayasittikul S, Ruchirawat S, Prachayasittikul V (2018) Eur J Med Chem 143:1604
Ghorab MM, Alsaid MS, Al-Ansary GH, Abdel-Latif GA, Abou El Ella DA (2016) Eur J Med Chem 124:946
Ghorab MM, Alsaid MS, Samir N, Abdel-Latif GA, Soliman AM, Ragab FA, Abou El Ella DA (2017) Eur J Med Chem 134:304
Ghosh D, Lo J, Morton D, Valette D, Xi J, Griswold J, Hubbell S, Egbuta C, Jiang W, An J, Davies HM (2012) J Med Chem 55:8464
Ziemer B, Sauer E (1998) Acta Crystallogr Sect C Cryst Struct Commun 54:IUC9700034
Ueno T, Toriumi M, Nonogaki S, Hashimoto M, Hayashi N, Tadano K, Iwayanagi T (1990) Azidobenzenesulfonates, their preparation, and their use in photosensitive materials for photolithographic imaging. Patent DE 3941757, Jun 21, 1990; (1991) Chem Abstr 114:52928
Mirian M, Zarghi A, Sadeghi S, Tabaraki P, Tavallaee M, Dadrass O, Sadeghi-Aliabadi H (2011) Iran J Pharm Res 10:741
El-Kardocy A, Mustafa M, Ahmed ER, Mohamady S, Mostafa YA (2019) Med Chem Res 28:2088
Woo LW, Bubert C, Purohit A, Potter BV (2011) ACS Med Chem Lett 2:243
Kalder M, Hadji P (2014) Breast Care 9:312
Chan KT, Meng FY, Li Q, Ho CY, Lam TS, To Y, Lee WH, Li M, Chu KH, Toh M (2010) Cancer Lett 294:118
Ghosh D, Egbuta C, Lo J (2018) J Steroid Biochem Mol Biol 181:11
Acknowledgements
Special thanks to Prof. Gamal Abu-Rahmah, Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Minia University for his advises through the research work, also to Dr. Abu-Baker Mostafa, Department of Pharmaceutical Organic Chemistry, Faculty of Pharmacy, Assiut University for his guidance through chemistry part, and finally, for Dr. A. Y. Desoky, Department of Chemistry, Faculty of Science, the University of Waterloo for running mass spectral analysis for our group.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mustafa, M., El-Kardocy, A. & Mostafa, Y.A. Development of new hetero-steroid hybrids with antiproliferative activity against MCF-7 breast cancer cells. Monatsh Chem 152, 137–149 (2021). https://doi.org/10.1007/s00706-020-02716-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-020-02716-0