Abstract
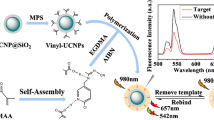
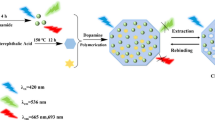
A molecularly imprinted ratiometric fluorescent probe (MIRF probe) was synthesized for the determination of aristolochic acid I (AAI) based on the Schiff-base fluorescent compound N,N′-bis(o-carboxybenzylidene)-p-4,4′-diaminobiphenyl (BDDB). The BDDB was immobilized in the silica nanoparticle (BDDB@SiO2) as an internal standard material. The blue-emitting BDDB@SiO2 and the yellow-emitting carbon quantum dots (y-CDs) were wrapped in the molecularly imprinted polymer (MIP) to provide a reliable reference signal at 440 nm and a fluorescent response signal at 530 nm at the excitation wavelength of 365 nm, respectively. In the preparation of the MIP of the MIRF probe, 4-vinylbenzoic acid as the functional monomer and AAI as the template molecule were used. An imprinting factor of 2.25 was obtained. Under the optimum conditions, the fluorescent response signal at 530 nm was quenched gradually by AAI in the range 1.0 to 120.0 μmol/L, while the reference signal at 440 nm remained unchanged. The limit of detection was 0.45 μmol/L, and the fluorescent color of the MIRF probe changed gradually from yellow to green to blue, which illustrated that the developed probe had a specific AAI recognition ability, a good anti-interference ability, and a sensitively visual determination ability. The probe was successfully applied to the AAI determination in traditional Chinese medicine (TCM) Asarum. The results showed that it had satisfactory recoveries (95.5–107.3%) and low relative standard deviations (2.0%). Furthermore, this method has a potential for the onsite naked eye determination of AAI in TCM samples.
Graphical abstract





Similar content being viewed by others
References
Zhou Q, Pei J, Poon J, Lau AY, Zhang L, Wang Y, Liu C, Huang L (2019) Worldwide research trends on aristolochic acids (1957-2017): suggestions for researchers. PLoS One 14:e0216135
Arlt VM, Stiborova M, Schmeiser HH (2002) Aristolochic acid as a probablehuman cancer hazard in herbal remedies: a review. Mutagenesis 17:265–277
Yang HY, Yang CC, Wu CY, Wang LJ, Lu KL (2019) Aristolochic acid and immunotherapy for urothelial carcinoma: directions for unmet needs. Int J Mol Sci 20:3162
Chen CH, Dickman KG, Huang CY, Moriya M, Shun CT, Tai HC, Huang KH, Wang SM, Lee YJ, Grollman AP, Pu YS (2013) Aristolochic acid-induced upper tract urothelial carcinoma in Taiwan: clinical characteristics and outcomes. Int J Cancer 133:14–20
Ng AWT, Poon SL, Huang MN, Lim JQ, Boot A, Yu W, Suzuki Y, Thangaraju S, Ng CCY, Tan P, Pang ST, Huang HY, Yu MC, Lee PH, Hsieh SY, Chang AY, Teh BT, Rozen SG (2017) Aristolochic acids and their derivatives are widely implicated in liver cancers in Taiwan and throughout Asia. Sci Transl Med 9:eaan6446
Poon SL, Pang ST, McPherson JR, Yu W, Huang KK, Guan P, Weng WH, Siew EY, Liu Y, Heng HL, Chong SC, Gan A, Tay ST, Lim WK, Cutcutache I, Huang D, Ler LD, Nairismagi ML, Lee MH, Chang YH, Yu KJ, Chan-on W, Li BK, Yuan YF, Qian CN, Ng KF, Wu CF, Hsu CL, Bunte RM, Stratton MR, Futreal PA, Sung WK, Chuang CK, Ong CK, Rozen SG, Tan P, Teh BT (2013) Genome-wide mutational signatures of aristolochic acid and its application as a screening tool. Sci Transl Med 5:197ra101
Wu HZ, Guo L, Mak YF, Liu N, Poon WT, Chan YW, Cai Z (2011) Proteomics investigation on aristolochic acid nephropathy: a case study on rat kidney tissues. Anal Bioanal Chem 399:3431–3439
Zhao Y, Chan CK, Chan KKJ, Chan W (2019) Quantitation of N6-formyl-lysine adduct following aristolochic acid exposure in cells and rat tissues by liquid chromatography-tandem mass spectrometry coupled with stable isotope-dilution method. Chem Res Toxicol 32:2086–2094
Ge YH, Shu H, Xu XY, Guo PQ, Liu RL, Luo ZM, Chang C, Fu Q (2019) Combined magnetic porous molecularly imprinted polymers and deep eutectic solvents for efficient and selective extraction of aristolochic acid I and II from rat urine. Mater Sci Eng C 97:650–657
Yu FY, Lin YH, Su CC (2006) A sensitive enzyme-linked immunosorbent assay for detecting carcinogenic aristolochic acid in herbal remedies. J Agric Food Chem 54:2496–2501
Li S, Wu X, Song S, Zheng Q, Kuang H (2019) Development of ic-ELISA and an immunochromatographic strip assay for the detection of aristolochic acid I. Food Agric Immunol 30:140–149
Yamani A, Bunel V, Antoine MH, Husson C, Stevigny C, Duez P, Elachouri M, Nortier J (2015) Substitution between Aristolochia and Bryonia genus in north-eastern Morocco: toxicological implications. J Ethnopharmacol 166:250–260
Ji F, Jin R, Luo C, Deng C, Hu Y, Wang L, Wang R, Zhang J, Song G (2020) Fast determination of aristolochic acid I (AAI) in traditional Chinese medicine soup with magnetic solid-phase extraction by high performance liquid chromatography. J Chromatogr A 1609:460455
Zhao YY, Tang DD, Chen H, Mao JR, Bai X, Cheng XH, Xiao XY (2015) Urinary metabolomics and biomarkers of aristolochic acid nephrotoxicity by UPLC-QTOF/HDMS. Bioanalysis 7:685–700
Yan Y, Huang C, Shen X (2019) Electromembrane extraction of aristolochic acids: new insights in separation of bioactive ingredients of traditional Chinese medicines. J Chromatogr A 1608:460424
Wang Y, Chan W (2014) Determination of aristolochic acids by high-performance liquid chromatography with fluorescence detection. J Agric Food Chem 62:5859–5864
Fu X, Liu Y, Li W, Bai Y, Liao Y, Liu H (2011) Determination of dissociation constants of aristolochic acid I and II by capillary electrophoresis with carboxymethyl chitosan-coated capillary. Talanta 85:813–815
Wei F, Feng YQ (2008) Rapid determination of aristolochic acid I and II in medicinal plants with high sensitivity by cucurbit[7]uril-modifier capillary zone electrophoresis. Talanta 74:619–624
Hsieh SC, Huang MF, Lin BS, Chang HT (2006) Determination of aristolochic acid in Chinese herbal medicine by capillary electrophoresis with laser-induced fluorescence detection. J Chromatogr A 1105:127–134
Oraby HF, Alarfaj NA, El-Tohamy MF (2017) Gold nanoparticle-enhanced luminol/ferricyanide chemiluminescence system for aristolochic acid-I detection in medicinal plants and slimming products. Green Chem Lett Rev 10:138–147
Yin HQ, Yang JC, Yin XB (2017) Ratiometric fluorescence sensing and real-time detection of water in organic solvents with one-pot synthesis of Ru@MIL-101(Al)–NH2. Anal Chem 89:13434–13440
Han L, Liu SG, Dong XZ, Liang JY, Li NB, Luo HQ (2019) Construction of an effective ratiometric fluorescent sensing platform for specific and visual detection of mercury ions based on target-triggered the inhibition on inner filter effect. J Hazard Mater 376:170–177
Liu J, Xu C, Yang T, Hu Z, Zhang Z, Feng G (2019) Developed a novel sensor based on fluorescent graft conjugated polymer for the determination of aristolochic acid in traditional Chinese medicine. Spectrochim Acta A Mol Biomol Spectrosc 222:117239
Hu Y, Wu HL, Yin XL, Gu HW, Xiao R, Wang L, Fang H, Yu RQ (2017) Interference-free spectrofluorometric quantification of aristolochic acid I and aristololactam I in five Chinese herbal medicines using chemical derivatization enhancement and second-order calibration methods. Spectrochim Acta A Mol Biomol Spectrosc 175:229–238
Gui R, An X, Huang W (2013) An improved method for ratiometric fluorescence detection of pH and Cd2+ using fluorescein isothiocyanate-quantum dots conjugates. Anal Chim Acta 767:134–140
Ma F, Sun M, Zhang K, Wang S (2015) A ratiometric fluorescence sensor for highly selective and sensitive detection of mercuric ion. Sensors Actuators B Chem 209:377–383
Hao T, Wei X, Nie Y, Xu Y, Lu K, Yan Y, Zhou Z (2016) Surface modification and ratiometric fluorescence dual function enhancement for visual and fluorescent detection of glucose based on dual-emission quantum dots hybrid. Sensors Actuators B Chem 230:70–76
Wang J, Dai J, Xu Y, Dai X, Zhang Y, Shi W, Sellergren B, Pan G (2019) Molecularly imprinted fluorescent test strip for direct, rapid, and visual dopamine detection in tiny amount of biofluid. Small 15:e1803913
Yao C, Niu C, Na N, He D, Ouyang J (2015) Aggregation-induced emission compounds as new assisted matrices for laser desorption/ionization time-of-flight mass spectrometry. Anal Chim Acta 853:375–383
Zhang X, Zhang X, Yang B, Liu L, Hui J, Liu M, Chen Y, Wei Y (2014) Aggregation-induced emission dye based luminescent silica nanoparticles: facile preparation, biocompatibility evaluation and cell imaging applications. RSC Adv 4:10060
Zhang X, Zhang X, Tao L, Chi Z, Xu J, Wei Y (2014) Aggregation induced emission-based fluorescent nanoparticles: fabrication methodologies and biomedical applications. J Mater Chem B 2:4398–4414
Achadu OJ, Revaprasadu N (2018) Microwave-assisted synthesis of thymine-functionalized graphitic carbon nitride quantum dots as a fluorescent nanoprobe for mercury (II). Microchim Acta 185:461
Qin Z, Wang W, Zhan X, Du X, Zhang Q, Zhang R, Li K, Li J, Xu W (2019) One-pot synthesis of dual carbon dots using only an N and S co-existed dopant for fluorescence detection of Ag+. Spectrochim Acta A Mol Biomol Spectrosc 208:162–171
Wen X, Fan Z (2016) Linear Schiff-base fluorescence probe with aggregation-induced emission characteristics for Al3+ detection and its application in live cell imaging. Anal Chim Acta 945:75–84
Kim S, Pudavar HE, Bonoiu A, Prasad PN (2007) Aggregation-enhanced fluorescence in organically modified silica nanoparticles: a novel approach toward high-signal-output nanoprobes for two-photon fluorescence bioimaging. Adv Mater 19:3791–3795
Ding Y, Zheng J, Wang J, Yang Y, Liu X (2019) Direct blending of multicolor carbon quantum dots into fluorescent films for white light emitting diodes with an adjustable correlated color temperature. J Mater Chem C 7:1502–1509
Xiao Y, Xiao R, Tang J, Zhu Q, Li X, Xiong Y, Wu X (2017) Preparation and adsorption properties of molecularly imprinted polymer via RAFT precipitation polymerization for selective removal of aristolochic acid I. Talanta 162:415–422
Dai Q, Wang Y, Xu W, Liu Y, Zhou Y (2017) Adsorption and specific recognition of DNA by using imprinted polymer layers grafted onto ionic liquid functionalized magnetic microspheres. Microchim Acta 184:4433–4441
Li F, Gao J, Li X, Li Y, He X, Chen L, Zhang Y (2019) Preparation of magnetic molecularly imprinted polymers functionalized carbon nanotubes for highly selective removal of aristolochic acid. J Chromatogr A 1602:168–177
Madhu P, Sivakumar P (2019) Curcumin-based fluorescent chemosensor for selective and efficient detection of picric acid. J Mol Struct 1185:410–415
Funding
This work was supported by the Guangdong Basic and Applied Basic Research Foundation (No. 2019A1515012169) and the National Natural Science Foundation of China (No. 81573678).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 1.30 MB)
Rights and permissions
About this article
Cite this article
Ye, J., Cai, X., Zhou, Q. et al. Molecularly imprinted ratiometric fluorescent probe for visual and fluorescent determination of aristolochic acid I based on a Schiff-base fluorescent compound. Microchim Acta 187, 623 (2020). https://doi.org/10.1007/s00604-020-04598-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-020-04598-9