Abstract
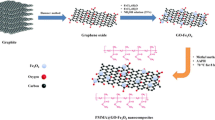
A magnetic sorbent was fabricated by coating the magnetized graphene oxide with polystyrene (PS) to obtain a sorbent of the type GO-Fe3O4@PS. The chemical composition and morphology of the sorbent were characterized. The sorbent was employed for the enrichment of polycyclic aromatic hydrocarbons (PAHs) from water samples. Various parameters affecting the enrichment were investigated. The PAHs were then quantified by gas chromatography with flame ionization detection. Linear responses were found in the range of 0.03–100 ng mL−1 for naphthalene and 2-methylnaphthalene, and of 0.01–100 ng mL−1 for fluorene and anthracene. The detection limits (at an S/N ratio of 3) range between 3 and 10 pg mL−1. The relative standard deviations (RSDs) for five replicates at three concentration levels (0.05, 5 and 50 ng mL−1) of analytes ranged from 4.9 to 7.4%. The method was applied to the analysis of spiked real water samples. Relative recoveries are between 95.8 and 99.5%, and RSD% are <8.4%.

A magnetic sorbent was fabricated by polystyrene coated on the magnetic graphene oxide for the extraction and preconcentration of PAHs in water samples prior to their determination by gas chromatography with flame ionization detection.







Similar content being viewed by others
References
Giakisikli G, Anthemidis AN (2013) Magnetic materials as sorbents for metal/metalloid preconcentration and/or separation. A review. Anal Chim Acta 789:1–16
Wierucka M, Biziuk M (2014) Application of magnetic nanoparticles for magnetic solid-phase extraction in preparing biological, environmental and food samples. TrAC Trends Anal Chem 59:50–58
Hemmati M, Rajabi M, Asghari A (2018) Magnetic nanoparticle based solid-phase extraction of heavy metal ions: a review on recent advances. Microchim Acta 185:160
Amiri A, Zonoz FM, Targhoo A, Saadati-Moshtaghin HR (2017) Enrichment of phenolic compounds from water samples by using magnetic Fe3O4 nanoparticles coated with a Keggin type heteropoly acid of type H6[BFe(OH2)W11O39] as a sorbent. Microchim Acta 184:1093–1101
Amiri A, Saadati-Moshtaghin HR, Zonoz FM, Targhoo A (2017) Preparation and characterization of magnetic wells–Dawson heteropoly acid nanoparticles for magnetic solid-phase extraction of aromatic amines in water samples. J Chromatogr A 1483:64–70
Safari M, Yamini Y, Tahmasebi E, Latifeh F (2015) Extraction and preconcentration of formaldehyde in water by polypyrrole-coated magnetic nanoparticles and determination by high-performance liquid chromatography. J Sep Sci 38:3421–3427
Asgharinezhad AA, Karami S, Ebrahimzadeh H, Shekari N, Jalilian N (2015) Polypyrrole/magnetic nanoparticles composite as an efficient sorbent for dispersive micro-solid-phase extraction of antidepressant drugs from biological fluids. Int J Pharm 494:102–112
Mehdinia A, Einollahi S, Jabbari A (2016) Magnetite nanoparticles surface-modified with a zinc (II)-carboxylate Schiff base ligand as a sorbent for solid-phase extraction of organochlorine pesticides from seawater. Microchim Acta 183:2615–2622
Asgharinezhad AA, Ebrahimzadeh H (2016) A simple and fast method based on mixed hemimicelles coated magnetite nanoparticles for simultaneous extraction of acidic and basic pollutants. Anal Bioanal Chem 408:473–486
Safari M, Yamini Y, Mani-Varnosfaderani A, Asiabi H (2017) Synthesis of Fe3O4@PPy–MWCNT nanocomposite and its application for extraction of ultra-trace amounts of PAHs from various samples. J Iran Chem Soc 14:623–634
Asgharinezhad AA, Ebrahimzadeh H (2015) Coextraction of acidic, basic and amphiprotic pollutants using multiwalled carbon nanotubes/magnetite nanoparticles@ polypyrrole composite. J Chromatogr A 1412:1–11
Li N, Jiang HL, Wang XL, Wang X, Xu GJ, Zhang BB, Wang LJ, Zhao RS, Lin JM (2018) Recent advances in graphene-based magnetic composites for magnetic solid-phase extraction. TrAC Trends Anal Chem 102:60–74
Jalilian N, Ebrahimzadeh H, Asgharinezhad AK, Molaei K (2017) Extraction and determination of trace amounts of gold(III), palladium(II), platinum(II) and silver(I) with the aid of a magnetic nanosorbent made from Fe3O4-decorated and silica-coated graphene oxide modified with a polypyrrole-polythiophene copolymer. Microchim Acta 184:2191–2200
Asgharinezhad AK, Ebrahimzadeh H (2016) Poly(2-aminobenzothiazole)-coated graphene oxide/magnetite nanoparticles composite as an efficient sorbent for determination of non-steroidal anti-inflammatory drugs in urine sample. J Chromatogr A 1435:18–29
Bashtani E, Amiri A, Baghayeri M (2018) A nanocomposite consisting of poly (methyl methacrylate), graphene oxide and Fe3O4 nanoparticles as a sorbent for magnetic solid-phase extraction of aromatic amines. Microchim Acta 185:14
Mehdinia A, Esfandiarnejad R, Jabbari A (2015) Magnetic nanocomposite of self-doped polyaniline–graphene as a novel sorbent for solid-phase extraction. J Sep Sci 38:141–147
Wang X, Liu B, Lu Q, Qu Q (2014) Graphene-based materials: fabrication and application for adsorption in analytical chemistry. J Chromatogr A 1362:1–15
Zali S, Jalali F, Es-Haghi A, Shamsipur M (2015) Electrospun nanostructured polystyrene as a new coating material for solid-phase microextraction: application to separation of multipesticides from honey samples. J Chromatogr B 1002:387–393
Yu X, Sun Y, Jiang C, Sun X, Gao Y, Wang Y, Zhang H, Song D (2012) Magnetic solid-phase extraction of five pyrethroids from environmental water samples followed by ultrafast liquid chromatography analysis. Talanta 98:257–264
Yu X, Yang H (2017) Pyrethroid residue determination in organic and conventional vegetables using liquid-solid extraction coupled with magnetic solid phase extraction based on polystyrene-coated magnetic nanoparticles. Food Chem 217:303–310
Marcano DC, Kosynkin DV, Berlin JM, Sinitskii A, Sun Z, Slesarev A, Alemany LB, Lu W, Tour JM (2010) Improved synthesis of graphene oxide. ACS Nano 4:4806–4814
Kassaee MZ, Motamedi E, Majdi M (2011) Magnetic Fe3O4-graphene oxide/polystyrene: fabrication and characterization of a promising nanocomposite. Chem Eng J 172:540–549
Kwon SH, Liu YD, Choi HJ (2015) Monodisperse poly (2-methylaniline) coated polystyrene core–shell microspheres fabricated by controlled releasing process and their electrorheological stimuli-response under electric fields. J Colloid Interface Sci 440:9–15
Kekez B, Gojgić-Cvijović G, Jakovljević D, Pavlović V, Beškoski V, Popović A, Vrvić MM, Nikolić V (2016) Synthesis and characterization of a new type of Levan-graft-polystyrene copolymer. Carbohydr Polym 154:20–29
Amiri A, Baghayeri M, Kashmari M (2016) Magnetic nanoparticles modified with polyfuran for the extraction of polycyclic aromatic hydrocarbons prior to their determination by gas chromatography. Microchim Acta 183:149–156
Amiri A, Ghaemi F (2016) Thermally stable carbon nanofibers functionalized with poly (dimethylsiloxane) for solid-phase microextraction of polycyclic aromatic hydrocarbons prior to GC analysis. Microchim Acta 183:1917–1924
Han Q, Wang Z, Xia J, Chen S, Zhang X, Ding M (2012) Facile and tunable fabrication of Fe3O4/graphene oxide nanocomposites and their application in the magnetic solid-phase extraction of polycyclic aromatic hydrocarbons from environmental water samples. Talanta 101:388–395
Tahmasebi E, Yamini Y (2012) Facile synthesis of new nano sorbent for magnetic solid-phase extraction by self assembling of bis-(2, 4, 4-trimethyl pentyl)-dithiophosphinic acid on Fe3O4@ ag core@ shell nanoparticles: characterization and application. Anal Chim Acta 756:13–22
Wang W, Li Z, Wang W, Zhang L, Zhang S, Wang C, Wang Z (2018) Microextraction of polycyclic aromatic hydrocarbons by using a stainless steel fiber coated with nanoparticles made from a porous aromatic framework. Microchim Acta 185:20
Amiri A, Ghaemi F (2017) Graphene grown on stainless steel mesh as a highly efficient sorbent for sorptive microextraction of polycyclic aromatic hydrocarbons from water samples. Anal Chim Acta 994:29–37
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author(s) declare that they have no competing interests.
Electronic supplementary material
ESM 1
(DOCX 1088 kb)
Rights and permissions
About this article
Cite this article
Amiri, A., Baghayeri, M. & Sedighi, M. Magnetic solid-phase extraction of polycyclic aromatic hydrocarbons using a graphene oxide/Fe3O4@polystyrene nanocomposite. Microchim Acta 185, 393 (2018). https://doi.org/10.1007/s00604-018-2928-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-018-2928-x