Abstract
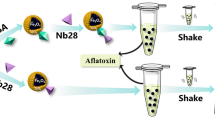
This article describes a method for determination of four aflatoxins by a combination of ultra-high performance liquid chromatography and mass spectrometry (UHPLC-MS/MS). Magnetic dummy molecularly imprinted polymers (md-MIPs) are used for fairly selective preconcentration. The preparation of the md-MIP involves (a) coating of magnetic nanoparticles (with a typical diameter of 200 nm) with a shell of silica, (b) modifying the surface with methacryloxy groups, and (c) creating a MIP on the surface by graft imprinting in aqueous solutions using 5,7-dimethoxycoumarin as the dummy template, and methacrylic acid and 4-vinylpyridine as the functional monomers. After elution of the template, the md-MIPs can be used for fairly selective extraction of the aflatoxins B1, B2, G1 and G2. Following magnetic separation of the particles, the aflatoxins were eluted and quantified by UHPLC-MS/MS. The limit of detection is 0.05 μg·kg−1 for aflatoxins B2 and G2, and 0.1 μg·kg−1 for aflatoxins B1 and G1. False-positive samples were identified by UHPLC-MS/MS in the positive ionization mode with multiple reaction monitoring.

We describe a method for determination of four aflatoxins by ultra-high performance liquid chromatography tandem mass spectrometry (UHPLC-MS/MS). Determination of aflatoxins was accomplished by the magnetic dummy molecularly imprinted polymers, followed by a magnetic separation and subsequent quantification by UHPLC-MS/MS.




Similar content being viewed by others
References
Richard JL (2007) Some major mycotoxins and their mycotoxicoses an overview. Int J Food Microbiol 119:3–10
Shephard GS (2008) Determination of mycotoxins in human food. Chem Soc Rev 37:2468–2477
Kensler TW, Roebuck BD, Wogan GN, Groopman JD (2011) Aflatoxin: a 50-year odyssey of mechanistic and translational toxicology. Toxicol Sci 120:S28–S48
Park JW, Kim Y (2006) Effect of pressure cooking on aflatoxin B1 in rice. J Agric Food Chem 54:2431–2435
Bennett JW, Klich M (2003) Mycotoxins. Clin Microbiol Rev 16:497–516
International agency for research on cancer (IARC), IARC monographs on the evaluation of carcinogenic risk to humans, vol. 56, p. 245.
Wild CP, Gong YY (2011) Mycotoxins and human disease: a largely ignored global health issue. Carcinogenesis 31:71–82
Commission regulation (EU) no. 165/2010 of 26 February 2010 amending regulation (EC) no. 1881/2006 setting maximum levels for certain contaminants in foodstuffs as regards aflatoxins.
Maragos CM (2009) Recent advances in the development of novel materials for mycotoxin analysis. Anal Bioanal Chem 395:1205–1213
Cervino C, Asam S, Knopp D, Rychlik M, Niessner R (2008) Use of isotope-labeled aflatoxins for LC-MS/MS stable isotope dilution analysis of foods. J Agric Food Chem 56:1873–1879
Mccullum C, Tchounwou P, Ding LS, Liao X, Liu YM (2014) Extraction of aflatoxins from liquid foodstuff samples with polydopamine-coated superparamagnetic nanoparticles for HPLCMS/MS analysis. J Agric Food Chem 62:4261–4267
Zhao L, Qin HQ, Wu RA, Zou HF (2012) Recent advances of mesoporous materials in sample preparation. J Chromatogr A 1228:193–204
Do JH, Choi DK (2007) Aflatoxins: detection, toxicity, and biosynthesis. biotechnol. Bioproc E 12:585–593
Haupt K (2010) Biomaterials: plastic antibodies. Nat Mater 9:612–614
Romero-Gonzalez R, Vidal JLM, Aguilera-Luiz MM, Frenich G (2009) Application of conventional solid-phase extraction for multimycotoxin analysis in beers by ultrahigh-performance liquid chromatography-tandem mass spectrometry. J Agric Food Chem 57:9385–9392
Matuszewski BK, Constanzer ML, Chavez-eng CM (2003) Strategies for the assessment of matrix effect in quantitative bioanalytical methods based on HPLC-MS/MS 75:3019–3030.
Gosettti F, Mazzucco E, Zampieri D, Gennaro MC (2010) Signal suppression/enhancement in high-performance liquid chromatography tandem mass spectrometry 1217:3929–3937.
Wulff G (1995) Molecular imprinting in cross-linked materials with the aid of molecular templates - a way towards artificial antibodies. Angew Chem Int Ed 34:1812–1832
Li SJ, Gao SS, Whitcombe MJ, Piletsky SA (2014) Size matters: challenges in imprinting macromolecules. Prog Polym Sci 39:145–163
Tan L, Chen KC, Huang C, Peng RF, Luo XY, Yang R, Cheng YF, Tang YW (2015) A fluorescent turn-on detection scheme for α-fetoprotein using Quantum-dots placed in a boronate-modified molecularly imprinted polymer with high affinity for glycoproteins. Microchim Acta 182:2615–2622
Tan J, Wang HF, Yan XP (2009) Discrimination of saccharides with a fluorescent molecular imprinting sensor array based on phenylboronic acid functionalized mesoporous silica. Anal Chem 81:5273–5280
Shi XZ, Liu JH, Sun AL, Li DX, Chen J (2012) Group-selective enrichment and determination of pyrethroid insecticides in aquaculture seawater via molecularly imprinted solid phase extraction coupled with gas chromatography-electron capture detection. J Chromatog A 1227:60–66
Lenain P, Mavungu JDD, Dubruel P, Robbens J, Saeger SD (2012) Development of suspension polymerized molecularly imprinted beads with metergoline as template and application in a solid-phase extraction procedure toward ergot alkaloids. Anal Chem 84:10411–10418
Urraca J, Aureliano C, Schillinger E, Esselmann H, Wiltfang J, Sllergren B (2011) Polymeric complements to the Alzheimer's disease biomarker β-amyloid isoforms Aβ1–40 and Aβ1–42 for blood serum analysis under denaturing conditions. J Am Chem Soc 133:9220–9223
Rao W, Cai R, Yin YL, Long F, Zhang ZH (2014) Magnetic dummy molecularly imprinted polymers based on multi-walled carbon nanotubes for rapid selective solid-phase extraction of 4-nonylphenol in aqueous samples. Talanta 128:170–176
Moreira FTC, Sharma S, Dutra RAF, Noronha JPC, Cass AEG, Sales MGF (2015) Detection of cardiac biomarker proteins using a disposable based on a molecularly imprinted polymer grafted onto graphite. Microchim Acta 182:975–983
Lv Y, Tan T, Svec F (2013) Molecular imprinting of proteins in polymers attached to the surface of nanomaterials for selective recognition of biomacromolecules 31: 1172–1186.
Li Y, Zhang XM, Deng CH (2013) Functionalized magnetic nanoparticles for sample preparation in proteomics and peptidomics analysis. Chem Soc Rev 42:8517–8539
Zhang Y, Liu Y, Hu YL, Li GK (2009) Microwave heating in preparation of magnetic molecularly imprinted polymer beads for trace triazines analysis in complicated samples. Anal Chem 81:967–976
Azodi-Deilami S, Najafabadi AH, Asadi E, Abdouss M, Kordestani D (2014) Magnetic molecularly imprinted polymer nanoparticles for the solid-phase extraction of paracetamol from plasma samples, followed its determination by HPLC. Microchim Acta 181:1823–1832
Qiao L, Gan N, Hu F, Wang D, Lan H, Li T, Wang H (2014) Magnetic nanospheres with a molecularly imprinted shell for the preconcentration of diethylstilbestrol. Microchim Acta 181:1341–1351
Gao L, Chen L, Li X (2015) Magnetic molecularly imprinted polymers based on carbon nanotubes for extraction of carbamates. Microchim Acta 182:781–787
Tan L, Yu ZR, Zhou XM, Xing D, Luo XY, Peng RF, Tang YW (2015) Antibody-free Ultra-high performance liquid chromatography/tandem mass spectrometry measurement of angiotensin I and II using magnetic epitope-imprinted polymers. J Chromatogr A 1411:69–76
Pardo A, Mespouille A, Dubois P, Blankert B, Duez P (2014) Molecularl imprinted polymers: compromise between flexibility and rigidity for improving capture of template analogues. Chem Eur J 20:3500–3509
Yang HY, Wang F, Wang H, Yang GL (2012) Miniaturized molecularly imprinted matrix solid-phase dispersion coupled with high performance liquid chromatography for rapid determination of auxins in Orange samples. J Chromatogr A 1256:1–8
Acknowledgments
We gratefully acknowledge financial support from the National Natural Science Foundation of China (No: 21505026 and 21275057) and Medical Science and Technology Project of Guangzhou (No: 20141 A010055).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author(s) declare that they have no competing interests
Electronic supplementary material
ESM 1
(PDF 751 kb)
Rights and permissions
About this article
Cite this article
Tan, L., He, R., Chen, K. et al. Ultra-high performance liquid chromatography combined with mass spectrometry for determination of aflatoxins using dummy molecularly imprinted polymers deposited on silica-coated magnetic nanoparticles. Microchim Acta 183, 1469–1477 (2016). https://doi.org/10.1007/s00604-016-1790-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-016-1790-y