Abstract
Background
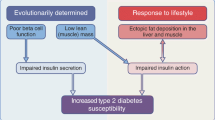
Type 2 diabetes (T2DM) is genetically heterogenous, driven by beta cell dysfunction and insulin resistance. Insulin resistance drives the development of cardiometabolic complications and is typically associated with obesity. A group of common variants at eleven loci are associated with insulin resistance and risk of both type 2 diabetes and coronary artery disease. These variants describe a polygenic correlate of lipodystrophy, with a high metabolic disease risk despite a low BMI.
Objectives
In this cross-sectional study, we sought to investigate the association of a polygenic risk score composed of eleven lipodystrophy variants with anthropometric, glycaemic and metabolic traits in an island population characterised by a high prevalence of both obesity and type 2 diabetes.
Methods
814 unrelated adults (n = 477 controls and n = 337 T2DM cases) of Maltese-Caucasian ethnicity were genotyped and associations with phenotypes explored.
Results
A higher polygenic lipodystrophy risk score was correlated with lower adiposity indices (lower waist circumference and body mass index measurements) and higher HOMA-IR, atherogenic dyslipidaemia and visceral fat dysfunction as assessed by the visceral adiposity index in the DM group. In crude and covariate-adjusted models, individuals in the top quartile of polygenic risk had a higher T2DM risk relative to individuals in the first quartile of the risk score distribution.
Conclusion
This study consolidates the association between polygenic lipodystrophy risk alleles, metabolic syndrome parameters and T2DM risk particularly in normal-weight individuals. Our findings demonstrate that polygenic lipodystrophy risk alleles drive insulin resistance and diabetes risk independent of an increased BMI.


Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Sattar N, Rawshani A, Franzén S et al (2019) Age at diagnosis of type 2 diabetes mellitus and associations with cardiovascular and mortality risks. Circulation 139:2228–2237. https://doi.org/10.1161/CIRCULATIONAHA.118.037885
Wang T, Li M, Zeng T et al (2022) Association between insulin resistance and cardiovascular disease risk varies according to glucose tolerance status: a nationwide prospective cohort study. Diabetes Care 45:1863–1872. https://doi.org/10.2337/dc22-0202
Perng W, Conway R, Mayer-Davis E, Dabelea D (2023) Youth-onset type 2 diabetes: the epidemiology of an awakening epidemic. Diabetes Care 46:490–499. https://doi.org/10.2337/dci22-0046
Mahler RJ, Adler ML (1999) Type 2 diabetes mellitus: update on diagnosis, pathophysiology, and treatment. J Clin Endocrinol Metab 84:1165–1171. https://doi.org/10.1210/jcem.84.4.5612
Li M, Chi X, Wang Y et al (2022) Trends in insulin resistance: insights into mechanisms and therapeutic strategy. Sig Transduct Target Ther 7:1–25. https://doi.org/10.1038/s41392-022-01073-0
James DE, Stöckli J, Birnbaum MJ (2021) The aetiology and molecular landscape of insulin resistance. Nat Rev Mol Cell Biol 22:751–771. https://doi.org/10.1038/s41580-021-00390-6
DeFronzo RA (2010) Insulin resistance, lipotoxicity, type 2 diabetes and atherosclerosis: the missing links. The claude bernard lecture 2009. Diabetologia 53:1270–1287. https://doi.org/10.1007/s00125-010-1684-1
Wondmkun YT (2020) Obesity, insulin resistance, and type 2 diabetes: associations and therapeutic implications. Diabetes Metab Syndr Obes 13:3611–3616. https://doi.org/10.2147/DMSO.S275898
Laakso M, Kuusisto J (2014) Insulin resistance and hyperglycaemia in cardiovascular disease development. Nat Rev Endocrinol 10:293–302. https://doi.org/10.1038/nrendo.2014.29
Scott RA, Lagou V, Welch RP et al (2012) Large-scale association analyses identify new loci influencing glycemic traits and provide insight into the underlying biological pathways. Nat Genet 44:991–1005. https://doi.org/10.1038/ng.2385
Manning AK, Hivert M-F, Scott RA et al (2012) A genome-wide approach accounting for body mass index identifies genetic variants influencing fasting glycemic traits and insulin resistance. Nat Genet 44:659–669. https://doi.org/10.1038/ng.2274
Dupuis J, Langenberg C, Prokopenko I et al (2010) New genetic loci implicated in fasting glucose homeostasis and their impact on type 2 diabetes risk. Nat Genet 42:105–116. https://doi.org/10.1038/ng.520
Ladha FA, Stitzel ML, Hinson JT (2020) From GWAS association to function: candidate gene screening within insulin resistance-associated genomic loci using a preadipocyte differentiation model. Circ Res 126:347–349. https://doi.org/10.1161/CIRCRESAHA.119.316405
Yaghootkar H, Scott RA, White CC et al (2014) Genetic evidence for a normal-weight “metabolically obese” phenotype linking insulin resistance, hypertension, coronary artery disease, and type 2 diabetes. Diabetes 63:4369–4377. https://doi.org/10.2337/db14-0318
Srinivasan S, Jablonski KA, Knowler WC et al (2019) A polygenic lipodystrophy genetic risk score characterizes risk independent of BMI in the diabetes prevention program. J Endocr Soc 3:1663–1677. https://doi.org/10.1210/js.2019-00069
Pace NP, Grech CA, Vella B et al (2022) Frequency and spectrum of glucokinase mutations in an adult Maltese population. Acta Diabetol 59:339–348. https://doi.org/10.1007/s00592-021-01814-7
Agius R, Pace NP, Fava S (2022) Sex differences in cardiometabolic abnormalities in a middle-aged Maltese population. Can J Public Health 25:1–17. https://doi.org/10.17269/s41997-021-00592-7
Agius R, Pace NP, Fava S (2021) Characterisation of body size phenotypes in a middle-aged Maltese population. J Nutr Sci 10:74. https://doi.org/10.1017/jns.2021.74
Lohman TJ, Roache AF, Martorell R (1992) Anthropometric standardization reference manual In: Medicine & science in sports & exercise, p 952
Zhu X, Yu L, Zhou H et al (2018) Atherogenic index of plasma is a novel and better biomarker associated with obesity: a population-based cross-sectional study in China. Lipids Health Dis 17:37. https://doi.org/10.1186/s12944-018-0686-8
Amato MC, Giordano C, Galia M et al (2010) Visceral Adiposity Index: a reliable indicator of visceral fat function associated with cardiometabolic risk. Diabetes Care 33:920–922. https://doi.org/10.2337/dc09-1825
Gurdasani D, Barroso I, Zeggini E, Sandhu MS (2019) Genomics of disease risk in globally diverse populations. Nat Rev Genet 20:520–535. https://doi.org/10.1038/s41576-019-0144-0
Peterson RE, Kuchenbaecker K, Walters RK et al (2019) Genome-wide association studies in ancestrally diverse populations: opportunities, methods, pitfalls, and recommendations. Cell 179:589–603. https://doi.org/10.1016/j.cell.2019.08.051
Duncan L, Shen H, Gelaye B et al (2019) Analysis of polygenic risk score usage and performance in diverse human populations. Nat Commun 10:3328. https://doi.org/10.1038/s41467-019-11112-0
Martin AR, Kanai M, Kamatani Y et al (2019) Clinical use of current polygenic risk scores may exacerbate health disparities. Nat Genet 51:584–591. https://doi.org/10.1038/s41588-019-0379-x
Azzopardi Muscat N, Calleja N, Calleja A, Cylus J (2014) Malta: health system review. Health Syst Transit 16:1–97
Cauchi D, Rutter H, Knai C (2015) An obesogenic island in the Mediterranean: mapping potential drivers of obesity in Malta. Public Health Nutrit First View 51:1–13. https://doi.org/10.1017/S1368980015000476
Gilbert E, Shanmugam A, Cavalleri GL (2022) Revealing the recent demographic history of Europe via haplotype sharing in the UK Biobank. Proc Natl Acad Sci 119:e2119281119. https://doi.org/10.1073/pnas.2119281119
Borg R, Farrugia Wismayer M, Bonavia K et al (2021) Genetic analysis of ALS cases in the isolated island population of Malta. Eur J Hum Genet 29:604–614. https://doi.org/10.1038/s41431-020-00767-9
Mintoff D, Pace NP, Borg I (2023) NCSTN in-frame deletion in maltese patients with hidradenitis suppurativa. JAMA Dermatol. https://doi.org/10.1001/jamadermatol.2023.2227
Hivert M-F, Christophi CA, Franks PW et al (2016) Lifestyle and metformin ameliorate insulin sensitivity independently of the genetic burden of established insulin resistance variants in diabetes prevention program participants. Diabetes 65:520–526. https://doi.org/10.2337/db15-0950
Khera AV, Chaffin M, Aragam KG et al (2018) Genome-wide polygenic scores for common diseases identify individuals with risk equivalent to monogenic mutations. Nat Genet 50:1219–1224. https://doi.org/10.1038/s41588-018-0183-z
Maamari DJ, Brockman DG, Aragam K et al (2022) Clinical implementation of combined monogenic and polygenic risk disclosure for coronary artery disease. JACC Adv 1:100068. https://doi.org/10.1016/j.jacadv.2022.100068
Kullo IJ, Lewis CM, Inouye M et al (2022) Polygenic scores in biomedical research. Nat Rev Genet 23:524–532. https://doi.org/10.1038/s41576-022-00470-z
Ahlqvist E, Storm P, Käräjämäki A et al (2018) Novel subgroups of adult-onset diabetes and their association with outcomes: a data-driven cluster analysis of six variables. Lancet Diabetes Endocrinol 6:361–369. https://doi.org/10.1016/S2213-8587(18)30051-2
Mansour Aly D, Dwivedi OP, Prasad RB et al (2021) Genome-wide association analyses highlight etiological differences underlying newly defined subtypes of diabetes. Nat Genet 53:1534–1542. https://doi.org/10.1038/s41588-021-00948-2
Udler MS, McCarthy MI, Florez JC, Mahajan A (2019) Genetic risk scores for diabetes diagnosis and precision medicine. Endocr Rev 40:1500–1520. https://doi.org/10.1210/er.2019-00088
Udler MS, Kim J, von Grotthuss M et al (2018) Type 2 diabetes genetic loci informed by multi-trait associations point to disease mechanisms and subtypes: a soft clustering analysis. PLoS Med 15:e1002654. https://doi.org/10.1371/journal.pmed.1002654
Gujral UP, Narayan KMV (2019) Diabetes in normal-weight individuals: high susceptibility in nonwhite populations. Diabetes Care 42:2164–2166. https://doi.org/10.2337/dci19-0046
Lontchi-Yimagou E, Dasgupta R, Anoop S et al (2022) An atypical form of diabetes among individuals with low BMI. Diabetes Care 45:1428–1437. https://doi.org/10.2337/dc21-1957
Amato MC, Giordano C (2014) Visceral adiposity index: an indicator of adipose tissue dysfunction. Int J Endocrinol 2014:730827. https://doi.org/10.1155/2014/730827
Bellary S, Kyrou I, Brown JE, Bailey CJ (2021) Type 2 diabetes mellitus in older adults: clinical considerations and management. Nat Rev Endocrinol 17:534–548. https://doi.org/10.1038/s41574-021-00512-2
Buchanan TA, Watanabe RM, Xiang AH (2010) Limitations in surrogate measures of insulin resistance. J Clin Endocrinol Metab 95:4874–4876. https://doi.org/10.1210/jc.2010-2167
Pou SA, Diaz MDP, Velázquez GA, Aballay LR (2022) Sociodemographic disparities and contextual factors in obesity: updated evidence from a national survey of risk factors for chronic diseases. Public Health Nutr 25:3377–3389. https://doi.org/10.1017/S1368980021004924
Keramati AR, Fathzadeh M, Go G-W et al (2014) A form of the metabolic syndrome associated with mutations in DYRK1B. N Engl J Med 370:1909–1919. https://doi.org/10.1056/NEJMoa1301824
Zembic A, Eckel N, Stefan N et al (2021) An empirically derived definition of metabolically healthy obesity based on risk of cardiovascular and total mortality. JAMA Netw Open 4:e218505. https://doi.org/10.1001/jamanetworkopen.2021.8505
Funding
This work was supported by institutional funds from the Faculty of Medicine and Surgery, University of Malta.
Author information
Authors and Affiliations
Contributions
NPP, SF, JV and RA collected data. RA, MZ and NPP performed the analyses. NPP and MZ drafted the manuscript. SF and JV reviewed the manuscript. All authors contributed to study conception and design and have read and approved the submitted manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical standard statement
This study was approved by the institutional ethics review board of the University of Malta (FRECMDS_1819_049), and the study protocol was in compliance with the Declaration of Helsinki.
Informed consent
All subjects gave written informed consent for their participation in the study and for genetic analysis.
Additional information
Communicated by Fabrizio Barbetti.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zammit, M., Agius, R., Fava, S. et al. Association between a polygenic lipodystrophy genetic risk score and diabetes risk in the high prevalence Maltese population. Acta Diabetol 61, 555–564 (2024). https://doi.org/10.1007/s00592-023-02230-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-023-02230-9