Abstract
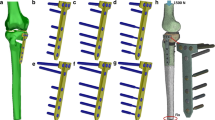
Diaphyseal tibia fractures may require plate fixation for proper healing to occur. Currently, there is no consensus on the number of screws required for proper fixation or the optimal placement of the screws within the plate. Mechanical stability of the construct is a leading criterion for choosing plate and screws configuration. However, number and location of screws have implications on the mechanical environment at the fracture site and, consequently, on bone healing response: The interfragmentary motion attained with a specific plate and screw construct may elicit mechano-transduction signals influencing cell-type differentiation, which in turn affects how well the fracture heals. This study investigated how different screw configurations affect mechanical performance of a tibia plate fixation construct. Three configurations of an eight-hole plate were considered with the fracture in the center of the plate: eight screws—screws at first, fourth, fifth and eighth hole and screws at first, third, sixth and eighth hole. Constructs’ stiffness was compared through biomechanical tests on bone surrogates. A finite element model of tibia diaphyseal fracture was used to conduct a stress analysis on the implanted hardware. Finally, the potential for bone regeneration of each screw configuration was assessed via the computational model through the evaluation of the magnitude of mechano-transduction signals at the bone callus. The results of this study indicate that having screws at fourth and fifth holes represents a preferable configuration since it provides mechanical properties similar to those attained by the stiffest construct (eight screws), and elicits an ideal bone regenerative response.








Similar content being viewed by others
References
Kempf I, Grosse A, Lafforgue D (1978) Combined Kuntscher nailing and screw fixation. Rev Chir Orthop Reparatrice Appar Mot 64(8):635–651
Adam P, Bonnomet F, Ehlinger M (2012) Advantage and limitations of a minimally-invasive approach and early weight bearing in the treatment of tibial shaft fractures with locking plates. Orthop Traumatol Surg Res 98(5):564–569
Sandoval JMC, Rueda JLO, Daza AS (2017) Management of diaphyseal tibial fractures by plate fixation with absolute or relative stability: a retrospective study of 45 patients. Trauma Surg Acute Care Open. https://doi.org/10.1136/tsaco-2016-000029
Egol KA, Kubiak EN, Fulkerson E, Kummer FJ, Koval KJ (2004) Biomechanics of locked plates and screws. J Orthop Trauma 18(8):488–493
Hertel R, Eijer H, Meisser A, Hauke C, Perren SM (2001) Biomechanical and biological considerations relating to the clinical use of the Point Contact-Fixator–evaluation of the device handling test in the treatment of diaphyseal fractures of the radius and/or ulna. Injury 32:B10–B14
Cronier P, Pietu G, Dujardin C, Bigorre N, Ducellier F, Gerard R (2010) The concept of locking plates. Orthop Traumatol Surg Res 96(4):S17–S36
Gautier E, Sommer C (2003) Guidelines for the clinical application of the LCP. Injury 34:B63–B76
Lv H, Chang W, Yuwen P, Yang N, Yan X, Zhang Y (2017) Are there too many screw holes in plates for fracture fixation? BMC Surg 17:46
Perren SM (1979) Physical and biological aspects of fracture healing with special reference to internal fixation. Clin Orthop Relat Res 138:175–196
Zhang L, Richardson M, Mendis P (2012) Role of chemical and mechanical stimuli in mediating bone fracture healing. Clin Exp Pharmacol Physiol 39(8):706–710
Palomares KTS, Gleason RE, Mason ZD, Cullinane DM, Einhorn TA, Gerstenfeld LC, Morgan EF (2009) Mechanical stimulation alters tissue differentiation and molecular expression during bone healing. J Orthop Res 27(9):1123–1132. https://doi.org/10.1002/jor.20863
Morgan EF, Palomares KTS, Gleason RE, Bellin DL, Chien KB, Unnikrishnan GU, Leong PL (2010) Correlations between local strains and tissue phenotypes in an experimental model of skeletal healing. J Biomech 43(12):2418–2424
Qin YX, Hu M (2014) Mechanotransduction in musculoskeletal tissue regeneration: effects of fluid flow, loading, and cellular-molecular pathways. Biomed Res Int. https://doi.org/10.1155/2014/863421
Fritton SP, Weinbaum S (2009) Fluid and solute transport in bone: flow-induced mechanotransduction. Annu Rev Fluid Mech 41:347–374
Hayward LNM, Morgan EF (2009) Assessment of a mechano-regulation theory of skeletal tissue differentiation in an in vivo model of mechanically induced cartilage formation. Biomech Model Mechanobiol. https://doi.org/10.1007/s10237-009-0148-3
Lee HY, Bae JH, Chang SH (2015) Mechano-regulation theory-based finite element analysis on the effects of driving strain history on cellular differentiation. Int J Precis Eng Manuf 16(8):1851–1858
Isaksson H, Wilson W, van Donkelaar CC, Huiskes R, Ito K (2006) Comparison of biophysical stimuli for mechano-regulation of tissue differentiation during fracture healing. J Biomech 39:1507–1516
Kumar A, Prasad T (2017) Computer modelling of bone’s adaptation: the role of normal strain, shear strain and fluid flow. Biomech Model Mechanobiol 16(2):395–410
Isaksson H (2012) Recent advances in mechanobiological modeling of bone regeneration. Mech Res Commun 42:22–31
Wolf S, Augat P, Eckert-Hübner K, Laule A, Krischak GD, Claes LE (2001) Effects of high-frequency, low-magnitude mechanical stimulus on bone healing. Clin Orthop Relat Res 385:192–198
Aoyama K, Sueyoshi Y, Tomita K, Sawaguchi T, Sakai Y, Sakamoto J, Oda J (1994) Effect of mechanical stimuli on healing of bone defects in the rabbit tibia. In: Clinical biomechanics and related research, pp 34–43
Hoerth RM, Seidt BM, Shah M, Schwarz C, Willie BM, Duda GN, Fratzl P, Wagermaier W (2014) Mechanical and structural properties of bone in non-critical and critical healing in rat. Acta Biomater 10(9):4009–4019
Ghiasi MS, Chen J, Vaziri A, Rodriguez EK, Nazarian A (2017) Bone fracture healing in mechanobiological modeling: a review of principles and methods. Bone Rep 6:87–100
Prendergast PJ, Huiskes R, Søballe K (1997) Biophysical stimuli on cells during tissue differentiation at implant interfaces. J Biomech 30(6):539–548
Morrison JB (1970) The mechanics of the knee joint in relation to normal walking. J Biomech 3(1):51–61
Taylor WR, Heller MO, Bergmann G, Duda GN (2004) Tibio-femoral loading during human gait and stair climbing. J Orthop Res 22(3):625–632
Maas SA, Ellis BJ, Ateshian GA, Weiss JA (2012) FEBio: finite elements for biomechanics. J Biomech Eng 134(1):011005
Miramini S, Zhang L, Richardson M, Mendis P, Oloyede A, Ebeling PR (2016) The relationship between interfragmentary movement and cell differentiation in early fracture healing under locking plate fixation. Australas Phys Eng Sci Med 39(1):123–133
Miramini S, Zhang L, Richardson M, Mendis P, Ebeling PR (2016) Influence of fracture geometry on bone healing under locking plate fixations: a comparison between oblique and transverse tibial fractures. Med Eng Phys 38(10):1100–1108
Lacroix D, Prendergast PJ (2002) A mechano-regulation model for tissue differentiation during fracture healing: analysis of gap size and loading. J Biomech 35(9):1163–1171
Steiner M, Claes L, Ignatius A, Simon U, Wehner T (2014) Disadvantages of interfragmentary shear on fracture healing—mechanical insights through numerical simulation. J Orthop Res 32(7):865–872
Klein P, Schell H, Streitparth F, Heller M, Kassi JP, Kandziora F, Bragulla H, Haas NP, Duda GN (2003) The initial phase of fracture healing is specifically sensitive to mechanical conditions. J Orthop Res 21(4):662–669
Grover P, Albert C, Wang M, Harris GF (2011) Mechanical characterization of fourth generation composite humerus. J Eng Med 225(12):1169–1176
Smit TH, Huyghe JM, Cowin SC (2002) Estimation of the poroelastic parameters of cortical bone. J Biomech 35:829–835
Malachanne EDD, Cañadas P, Jourdan F (2008) Experimental and numerical identification of cortical bone permeability. J Biomech 41:721–725
Welsch G, Boyer R, Collings EW (1993) Materials properties handbook: titanium alloys. ASM International, Materials Park
Funding
This study was partially supported by Miami CORE (Center for Orthopaedic Research and Education), University of Miami, Department of Orthopaedics (information that explains whether and by whom the research was supported).
Author information
Authors and Affiliations
Contributions
FT, LTB, EM and LL conceived and designed the study, performed experiments, participated to data interpretation and drafted the manuscript. FT developed the computational model.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Travascio, F., Buller, L.T., Milne, E. et al. Mechanical performance and implications on bone healing of different screw configurations for plate fixation of diaphyseal tibia fractures: a computational study. Eur J Orthop Surg Traumatol 31, 121–130 (2021). https://doi.org/10.1007/s00590-020-02749-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00590-020-02749-5