Abstract
Background
In preparation for surgery, patients being treated with disease-modifying antirheumatic drugs (DMARDs) are recommended to either continue or withhold therapy perioperatively. Some of these drugs have known effects against bone healing, hence the importance of adequately managing them before and after surgery.
Objective
We aim to assess the current evidence for managing conventional synthetic and/or biologic DMARDs in the perioperative period for elective spine surgery.
Methods
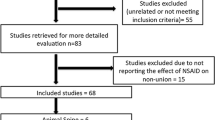
A systematic review of four databases was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. The included manuscripts were methodically scrutinized for quality, postoperative infections, wound healing characteristics, bone fusion rates, and clinical outcomes.
Results
Six studies were identified describing the management of conventional synthetic and/or biologic DMARDs. There were 294 DMARD-treated patients described undergoing various spine surgeries such as craniovertebral junction fusions. Three of the studies involved exclusive continuation of DMARDs in the perioperative window; one study involved exclusive discontinuation of DMARDs in the perioperative window; and two studies involved continuation or discontinuation of DMARDs perioperatively. Of patients that continued DMARDs in the perioperative period, 13/50 patients (26.0%) had postoperative surgical site infections or wound dehiscence, 2/19 patients (10.5%) had delayed wound healing, and 32/213 patients (15.0%) had secondary revision surgeries. A fusion rate of 14/19 (73.6%) was described in only one study for patients continuing DMARDs perioperatively.
Conclusions
The available published data may suggest a higher risk of wound healing concerns and lower than average bone fusion, although this may be under-reported given the current state of the literature.

Similar content being viewed by others
References
Cronstein BN (1997) The mechanism of action of methotrexate. Rheum Dis Clin North Am 23(4):739–55. https://doi.org/10.1016/s0889-857x(05)70358-6
Broen JCA, van Laar JM (2020) Mycophenolate mofetil, azathioprine and tacrolimus: mechanisms in rheumatology. Nat Rev Rheumatol 16(3):167–178. https://doi.org/10.1038/s41584-020-0374-8
Mitoma H, Horiuchi T, Tsukamoto H, Ueda N (2018) Molecular mechanisms of action of anti-TNF-α agents–comparison among therapeutic TNF-α antagonists. Cytokine 101:56–63. https://doi.org/10.1016/j.cyto.2016.08.014
Schett G, Emery P, Tanaka Y et al (2016) Tapering biologic and conventional DMARD therapy in rheumatoid arthritis: current evidence and future directions. Ann Rheum Dis 75(8):1428–37. https://doi.org/10.1136/annrheumdis-2016-209201
Smolen JS, van der Heijde D, Machold KP, Aletaha D, Landewé R (2014) Proposal for a new nomenclature of disease-modifying antirheumatic drugs. Ann Rheum Dis 73(1):3–5. https://doi.org/10.1136/annrheumdis-2013-204317
Smolen JS, Breedveld FC, Burmester GR et al (2016) Treating rheumatoid arthritis to target: 2014 update of the recommendations of an international task force. Ann Rheum Dis 75(1):3–15. https://doi.org/10.1136/annrheumdis-2015-207524
Thong B, Olsen NJ (2017) Systemic lupus erythematosus diagnosis and management. Rheumatology (Oxford) 56(suppl_1):i3–i13. https://doi.org/10.1093/rheumatology/kew401
Ward MM, Deodhar A, Gensler LS et al (2019) 2019 update of the American college of rheumatology/spondylitis association of America/spondyloarthritis research and treatment network recommendations for the treatment of ankylosing spondylitis and nonradiographic axial spondyloarthritis. Arthritis Rheumatol 71(10):1599–1613. https://doi.org/10.1002/art.41042
Singh JA, Saag KG, Bridges SL Jr et al (2016) 2015 American college of rheumatology guideline for the treatment of rheumatoid arthritis. Arthritis Care Res (Hoboken) 68(1):1–25. https://doi.org/10.1002/acr.22783
Aletaha D, Smolen JS (2018) Diagnosis and management of rheumatoid arthritis: a review. JAMA 320(13):1360–1372. https://doi.org/10.1001/jama.2018.13103
Mertelsmann-Voss C, Lyman S, Pan TJ, Goodman S, Figgie MP, Mandl LA (2014) Arthroplasty rates are increased among US patients with systemic lupus erythematosus: 1991–2005. J Rheumatol 41(5):867–74. https://doi.org/10.3899/jrheum.130617
Krauss WE, Bledsoe JM, Clarke MJ, Nottmeier EW, Pichelmann MA (2010) Rheumatoid arthritis of the craniovertebral junction. Neurosurgery 66(Suppl_3):83–95. https://doi.org/10.1227/01.Neu.0000365854.13997.B0
Goodman SM, Springer B, Guyatt G et al (2017) 2017 American college of rheumatology/American association of hip and knee surgeons guideline for the perioperative management of antirheumatic medication in patients with rheumatic diseases undergoing elective total hip or total knee arthroplasty. Arthritis Care Res (Hoboken) 69(8):1111–1124. https://doi.org/10.1002/acr.23274
Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A (2016) Rayyan-a web and mobile app for systematic reviews. Syst Rev 5(1):210. https://doi.org/10.1186/s13643-016-0384-4
Khan KS, Kunz R, Kleijnen J, Antes G (2003) Five steps to conducting a systematic review. J R Soc Med 96(3):118–21. https://doi.org/10.1258/jrsm.96.3.118
Khanna R, Dlouhy BJ, Smith ZA, Lam SK, Koski TR, Dahdaleh NS (2015) The impact of steroids, methotrexate, and biologics on clinical and radiographic outcomes in patients with rheumatoid arthritis undergoing fusions at the craniovertebral junction. J Craniovertebr Junction Spine 6(2):60–4. https://doi.org/10.4103/0974-8237.156044
Koyama K, Ohba T, Ebata S, Haro H (2016) Postoperative surgical infection after spinal surgery in rheumatoid arthritis. Orthopedics 39(3):e430-3. https://doi.org/10.3928/01477447-20160404-05
Dalle Ore CL, Ames CP, Deviren V, Lau D (2019) Perioperative outcomes associated with thoracolumbar 3-column osteotomies for adult spinal deformity patients with rheumatoid arthritis. J Neurosurg Spine 30(6):822–832. https://doi.org/10.3171/2018.11.Spine18927
Elia CJ, Brazdzionis J, Toor H et al (2020) Impact of chronic DMARD therapy in patients with rheumatoid arthritis undergoing surgery of the craniovertebral junction: a multi-center retrospective study. Spine (Phila Pa 1976) 45(13):930–936. https://doi.org/10.1097/brs.0000000000003402
Gaudiani MA, Winkelman RD, Ravishankar P, Rabah NM, Mroz TE, Coughlin DJ (2021) The association of preoperative TNF-alpha inhibitor use and reoperation rates in spinal fusion surgery. Spine J 21(6):972–979. https://doi.org/10.1016/j.spinee.2021.01.020
Ogihara S, Murase S, Oguchi F, Saita K (2020) Deep surgical site infection after posterior instrumented fusion for rheumatoid upper cervical subluxation treated with antibiotic-loaded bone cement: three case reports. Medicine (Baltimore) 99(26):e20892. https://doi.org/10.1097/md.0000000000020892
Zhou J, Wang R, Huo X, Xiong W, Kang L, Xue Y (2020) Incidence of surgical site infection after spine surgery: a systematic review and meta-analysis. Spine (Phila Pa 1976) 45(3):208–216. https://doi.org/10.1097/brs.0000000000003218
Youssef JA, Heiner AD, Montgomery JR et al (2019) Outcomes of posterior cervical fusion and decompression: a systematic review and meta-analysis. Spine J 19(10):1714–1729. https://doi.org/10.1016/j.spinee.2019.04.019
Grennan DM, Gray J, Loudon J, Fear S (2001) Methotrexate and early postoperative complications in patients with rheumatoid arthritis undergoing elective orthopaedic surgery. Ann Rheum Dis 60(3):214–7. https://doi.org/10.1136/ard.60.3.214
Kasdan ML, June L (1993) Postoperative results of rheumatoid arthritis patients on methotrexate at the time of reconstructive surgery of the hand. Orthopedics 16(11):1233–1235
Jain A, Witbreuk M, Ball C, Nanchahal J (2002) Influence of steroids and methotrexate on wound complications after elective rheumatoid hand and wrist surgery. J Hand Surg Am 27(3):449–55. https://doi.org/10.1053/jhsu.2002.32958
Murata K, Yasuda T, Ito H, Yoshida M, Shimizu M, Nakamura T (2006) Lack of increase in postoperative complications with low-dose methotrexate therapy in patients with rheumatoid arthritis undergoing elective orthopedic surgery. Mod Rheumatol 16(1):14–9. https://doi.org/10.1007/s10165-005-0444-4
Tanaka N, Sakahashi H, Sato E, Hirose K, Ishima T, Ishii S (2003) Examination of the risk of continuous leflunomide treatment on the incidence of infectious complications after joint arthroplasty in patients with rheumatoid arthritis. J Clin Rheumatol 9(2):115–8. https://doi.org/10.1097/01.RHU.0000062514.54375.bd
Fuerst M, Möhl H, Baumgärtel K, Rüther W (2006) Leflunomide increases the risk of early healing complications in patients with rheumatoid arthritis undergoing elective orthopedic surgery. Rheumatol Int 26(12):1138–42. https://doi.org/10.1007/s00296-006-0138-z
Goëb V, Ardizzone M, Arnaud L et al (2013) Recommendations for using TNFα antagonists and French clinical practice guidelines endorsed by the French national authority for health. Joint Bone Spine 80(6):574–81. https://doi.org/10.1016/j.jbspin.2013.09.001
Scherrer CB, Mannion AF, Kyburz D, Vogt M, Kramers-de Quervain IA (2013) Infection risk after orthopedic surgery in patients with inflammatory rheumatic diseases treated with immunosuppressive drugs. Arthritis Care Res (Hoboken) 65(12):2032–40. https://doi.org/10.1002/acr.22077
Latourte A, Gottenberg JE, Luxembourger C et al (2017) Safety of surgery in patients with rheumatoid arthritis treated by abatacept: data from the French orencia in rheumatoid arthritis registry. Rheumatology (Oxford) 56(4):629–637. https://doi.org/10.1093/rheumatology/kew476
Godot S, Gottenberg JE, Paternotte S et al (2013) Safety of surgery after rituximab therapy in 133 patients with rheumatoid arthritis: data from the autoimmunity and rituximab registry. Arthritis Care Res (Hoboken) 65(11):1874–9. https://doi.org/10.1002/acr.22056
Bovid KM, Moore MD (2019) Juvenile idiopathic arthritis for the pediatric orthopedic surgeon. Orthop Clin North Am 50(4):471–488. https://doi.org/10.1016/j.ocl.2019.06.003
Krause ML, Matteson EL (2014) Perioperative management of the patient with rheumatoid arthritis. World J Orthop 5(3):283–91. https://doi.org/10.5312/wjo.v5.i3.283
Gualtierotti R, Parisi M, Ingegnoli F (2018) Perioperative management of patients with inflammatory rheumatic diseases undergoing major orthopaedic surgery: a practical overview. Adv Ther 35(4):439–456. https://doi.org/10.1007/s12325-018-0686-0
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There is no actual or potential conflict of interest in relation to this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mamaril-Davis, J.C., Aguilar-Salinas, P., Avila, M.J. et al. Perioperative management of disease-modifying antirheumatic drugs for patients undergoing elective spine surgery: a systematic review. Eur Spine J 31, 815–829 (2022). https://doi.org/10.1007/s00586-021-07080-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-021-07080-z