Abstract
Background
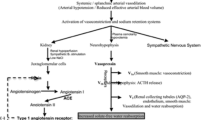
The water channel aquaporin 2 (AQP2) at the apical membrane of renal collecting duct cells mediates water reabsorption. The expression of AQP2 at the apical membrane is tightly regulated by vasopressin and was quantitated by measurement of the urinary form by a recently developed ELISA. Tolvaptan, an antagonist of vasopressin type 2 receptor, inhibits water reabsorption in cirrhosis. The aim of this study was to determine the correlation between the pharmacological effect of tolvaptan and the dynamics of urinary AQP2 levels.
Methods
Tolvaptan was administered to 41 cirrhotic patients with ascites unresponsive to standard diuretic therapy. Urinary excretion of AQP2 and urinary osmolarity were measured at the baseline and at 4, 8, and 24 h after administration of tolvaptan.
Results
At the baseline, urinary AQP2/creatinine ratios were significantly higher in cirrhotic patients with ascites than in healthy controls (P < 0.0001). After administration of tolvaptan, urinary AQP2/creatinine ratios decreased by 45.0 % at 4 h and 77.0 % at 8 h. Similarly, urinary osmolarity decreased by 42.0 % at 4 h and 41.5 % at 8 h. Urinary AQP2 levels and urinary osmolarity significantly correlated at the baseline and at all time points after tolvaptan administration. The degree of the decrease in urinary AQP2 levels and degree of the decrease in urinary osmolarity correlated significantly at 4 h (r = 0.452, P = 0.009) and 8 h (r = 0.384, P = 0.030) after tolvaptan administration.
Conclusions
These results indicate that the vasopressin–AQP2 system plays a major role in fluid retention in cirrhosis and that the pharmacological effect of tolvaptan to inhibit water reabsorption can be monitored by measurement of the dynamics of urinary AQP2 levels.




Similar content being viewed by others
References
Sola E, Watson H, Graupera I, et al. Factors related to quality of life in patients with cirrhosis and ascites: relevance of serum sodium concentration and leg edema. J Hepatol. 2012;57(6):1199–206.
Gines P, Cardenas A, Arroyo V, Rodes J. Management of cirrhosis and ascites. N Engl J Med. 2004;350(16):1646–54.
Gines P, Jimenez W, Arroyo V, et al. Atrial natriuretic factor in cirrhosis with ascites: plasma levels, cardiac release and splanchnic extraction. Hepatology. 1988;8(3):636–42.
La Villa G, Romanelli RG, Casini Raggi V, et al. Plasma levels of brain natriuretic peptide in patients with cirrhosis. Hepatology. 1992;16(1):156–61.
Arroyo V, Bosch J, Mauri M, et al. Renin, aldosterone and renal haemodynamics in cirrhosis with ascites. Eur J Clin Investig. 1979;9(1):69–73.
Bichet D, Szatalowicz V, Chaimovitz C, Schrier RW. Role of vasopressin in abnormal water excretion in cirrhotic patients. Ann Intern Med. 1982;96(4):413–7.
Claria J, Jimenez W, Arroyo V, et al. Blockade of the hydroosmotic effect of vasopressin normalizes water excretion in cirrhotic rats. Gastroenterology. 1989;97(5):1294–9.
Claria J, Jimenez W, Arroyo V, et al. Effect of V1-vasopressin receptor blockade on arterial pressure in conscious rats with cirrhosis and ascites. Gastroenterology. 1991;100(2):494–501.
Asahina Y, Izumi N, Enomoto N, et al. Increased gene expression of water channel in cirrhotic rat kidneys. Hepatology. 1995;21(1):169–73.
Fushimi K, Uchida S, Hara Y, Hirata Y, Marumo F, Sasaki S. Cloning and expression of apical membrane water channel of rat kidney collecting tubule. Nature. 1993;361(6412):549–52.
Sasaki S, Fushimi K, Saito H, et al. Cloning, characterization, and chromosomal mapping of human aquaporin of collecting duct. J Clin Investig. 1994;93(3):1250–6.
Radin MJ, Yu MJ, Stoedkilde L, et al. Aquaporin-2 regulation in health and disease. Vet Clin Pathol. 2012;41(4):455–70.
Sasaki S. Aquaporin 2: from its discovery to molecular structure and medical implications. Mol Aspects Med. 2012;33(5–6):535–46.
Brown D, Hasler U, Nunes P, Bouley R, Lu HA. Phosphorylation events and the modulation of aquaporin 2 cell surface expression. Curr Opin Nephrol Hyperten. 2008;17(5):491–8.
Kanno K, Sasaki S, Hirata Y, et al. Urinary excretion of aquaporin-2 in patients with diabetes insipidus. N Engl J Med. 1995;332(23):1540–5.
Pisitkun T, Shen RF, Knepper MA. Identification and proteomic profiling of exosomes in human urine. Proc Natl Acad Sci U S A. 2004;101(36):13368–73.
Rai T, Sekine K, Kanno K, et al. Urinary excretion of aquaporin-2 water channel protein in human and rat. J Am Soc Nephrol. 1997;8(9):1357–62.
Sasaki S, Ohmoto Y, Mori T, Iwata F, Muraguchi M. Daily variance of urinary excretion of AQP2 determined by sandwich ELISA method. Clin Exp Nephrol. 2012;16(3):406–10.
Ivarsen P, Frokiaer J, Aagaard NK, et al. Increased urinary excretion of aquaporin 2 in patients with liver cirrhosis. Gut. 2003;52(8):1194–9.
Elliot S, Goldsmith P, Knepper M, Haughey M, Olson B. Urinary excretion of aquaporin-2 in humans: a potential marker of collecting duct responsiveness to vasopressin. J Am Soc Nephrol. 1996;7(3):403–9.
Matsuzaki M, Hori M, Izumi T, Fukunami M. Efficacy and safety of tolvaptan in heart failure patients with volume overload despite the standard treatment with conventional diuretics: a phase III, randomized, double-blind, placebo-controlled study (QUEST study). Cardiovasc Drugs Ther. 2011;25(Suppl 1):S33–45.
Konstam MA, Gheorghiade M, Burnett JC Jr, et al. Effects of oral tolvaptan in patients hospitalized for worsening heart failure: the EVEREST outcome trial. JAMA. 2007;297(12):1319–31.
Gheorghiade M, Konstam MA, Burnett JC Jr, et al. Short-term clinical effects of tolvaptan, an oral vasopressin antagonist, in patients hospitalized for heart failure: the EVEREST clinical status trials. JAMA. 2007;297(12):1332–43.
Hori M. Tolvaptan for heart failure patients with volume overload. Cardiovasc Drug Ther. 2011;25(Suppl 1):S1–4.
Schrier RW, Gross P, Gheorghiade M, et al. Tolvaptan, a selective oral vasopressin V2-receptor antagonist, for hyponatremia. N Engl J Med. 2006;355(20):2099–112.
Sakaida I, Kawazoe S, Kajimura K, et al. Tolvaptan for improvement of hepatic edema: a phase 3, multicenter, randomized, double-blind, placebo-controlled trial. Hepatol Res. 2014;44(1):73–82.
Zhang X, Wang SZ, Zheng JF, et al. Clinical efficacy of tolvaptan for treatment of refractory ascites in liver cirrhosis patients. World J Gastroenterol. 2014;20(32):11400–5.
Gaglio P, Marfo K, Chiodo J 3rd. Hyponatremia in cirrhosis and end-stage liver disease: treatment with the vasopressin V2-receptor antagonist tolvaptan. Dig Dis Sci. 2012;57(11):2774–85.
Okita K, Kawazoe S, Hasebe C, et al. Dose-finding trial of tolvaptan in liver cirrhosis patients with hepatic edema: a randomized, double-blind, placebo-controlled trial. Hepatol Res. 2014;44(1):83–91.
Kogiso T, Tokushige K, Hashimoto E, et al. Safety and efficacy of long-term tolvaptan therapy for decompensated liver cirrhosis. Hepatol Res. 2015. doi:10.1111/hepr.12547.
Ohki T, Sato K, Yamada T, et al. Efficacy of tolvaptan in patients with refractory ascites in a clinical setting. World J Hepatol. 2015;7(12):1685–93.
European Association for the Study of the Liver. EASL clinical practice guidelines on the management of ascites, spontaneous bacterial peritonitis, and hepatorenal syndrome in cirrhosis. J Hepatol. 2010;53(3):397–417.
Hernandez-Guerra M, Garcia-Pagan JC, Bosch J. Increased hepatic resistance: a new target in the pharmacologic therapy of portal hypertension. J Clin Gastroenterol. 2005;39(4 Suppl 2):S131–7.
Epstein FH. Underfilling versus overflow in hepatic ascites. N Engl J Med. 1982;307(25):1577–8.
Esteva-Font C, Baccaro ME, Fernandez-Llama P, et al. Aquaporin-1 and aquaporin-2 urinary excretion in cirrhosis: relationship with ascites and hepatorenal syndrome. Hepatology. 2006;44(6):1555–63.
Chung SH, Jun DW, Kim KT, et al. Aquaporin-2 urinary excretion in cirrhosis: relationship to vasopressin and nitric oxide. Dig Dis Sci. 2010;55(4):1135–41.
Acknowledgment
This study was supported by a Grant-in-Aid from the Japan Agency for Medical Research and Development.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Namiki Izumi received grants from the Japanese Ministry of Welfare, Health, and Labor and the Japanese Ministry of Education, Culture, Sports, and Science during the conduct of this study, and has received lecture fees from MSD, Chugai Pharmaceutical Company, Ltd.; Daiichi-Sankyo Company, Ltd.; Bayer AG; and Janssen Pharmaceutical K.K. Masayuki Kurosaki has received lecture fees from Chugai Pharmaceutical Company, Ltd; and Janssen Pharmaceutical K.K.
Additional information
H. Nakanishi and M. Kurosaki contributed equally to this work.
Rights and permissions
About this article
Cite this article
Nakanishi, H., Kurosaki, M., Hosokawa, T. et al. Urinary excretion of the water channel aquaporin 2 correlated with the pharmacological effect of tolvaptan in cirrhotic patients with ascites. J Gastroenterol 51, 620–627 (2016). https://doi.org/10.1007/s00535-015-1143-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-015-1143-3