Abstract
Purpose
Survival rates for children diagnosed with acute lymphoblastic leukemia (ALL) have increased significantly over recent decades, and thus attention shifted toward understanding the adverse effects of cancer treatment. Chemotherapy has side effects that could affect muscle state and diminish motor performance. This scoping review was conducted to map the breadth of evidence for different tools used in fine motor skills assessment, the extent of upper extremity strength, and fine motor performance, highlighting the potential risk factors that may influence these skills.
Methods
In March 2023, full-text studies that examined fine motor performance and/or upper extremity strength were identified via searches in PubMed, Science Direct, Scopus, Web of Science, and PEDro databases. The titles and abstracts of selected studies were screened according to the inclusion and exclusion criteria.
Results
The search yielded initial 418 citations and 26 peer-reviewed articles were finally included in the review. Considerable heterogeneity was observed regarding the methods of evaluating fine motor skills. The results of this review indicate that children and adolescents with ALL experienced fine motor limitations and upper extremity weakness either during or after cessation of treatment.
Conclusion
This scoping review presents a broad overview of the literature addressing fine motor difficulties in the pediatric population with ALL. Results accentuate the need to incorporate strengthening and occupational therapy training to preserve muscle strength and minimize future fine motor problems along the course of chemotherapeutic treatment. Little evidence was reported regarding the risk factors that may impair muscle strength and motor performance.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acute lymphoblastic leukemia (ALL) is the most common childhood cancer with an annual incidence rate of approximately 3.9 per 100,000 children, which accounts for over 75% of childhood leukemia [1, 2]. The highest incidence of ALL occurs during the first 5 years of life [3] that was more commonly observed in males as compared to females [4, 5].
Survival rates for childhood ALL have increased over the past five decades and reached about 90% [6]. Since the survival rate of leukemia has been improved, attention has shifted toward the understanding and treating of the adverse effects of cancer treatment [7].
Neurotoxicity from chemotherapy drugs affects both the central and peripheral nervous systems. Chemotherapeutic agents used in the management of childhood malignancies have side effects that can lead to a decrease in motor performance [8]. Treatment of ALL in children, mainly with vincristine and methotrexate, causes a long-standing axonal injury throughout the nervous system and demyelination within the spinal cord [9]. Motor and sensory-perceptual deficits can be considered important indicators of a compromised central nervous system. Chemotherapy induces oxidative stress in children with ALL, which in turn increases the risk of fine and visual-motor problems [10].
Generally, motor development is divided into fine and gross motor skills. Fine motor skills are necessary for developing basic self-help skills which involve using small muscles to manipulate objects [11, 12]. Fine motor functions have great biological importance for humans, as reflected by the large cortical representation of hands in the cerebral cortex [13]. During an ordinary school day, children spend between 30 and 60% of their school day performing fine motor tasks that involve the manipulation of writing implements, such as pencils, which are considered the most important skill in academic achievement [14]. Paper and pencil-based activities account for up to 85% of the time spent engaged in fine motor tasks [15].
Motor disability in children or survivors diagnosed with ALL is thought to be due to insufficient muscle activity leading to muscle weakness [16]. The resultant direct muscle fiber damage and hypoplasia adversely affect the function of the musculoskeletal system that subsequently lead to disuse and deconditioning with further weakness and impaired motor performance [8, 17].
Measures of fine motor control require manual dexterity and visual-motor integration [18]. Manual dexterity depends on prehension, coordination, and skills acquired through the practice of activities involving the manipulation of objects. Speed and precision are the commonly used criteria to measure this skill [19].
Few review studies discussed the effects of chemotherapeutic treatment on neuropsychological and neurocognitive functions that include fine motor skills or manual dexterity as a secondary aim [20,21,22,23]. The majority of these reviews focused on ALL survivors. In this review, we aimed to summarize clinical findings regarding the used evaluation tools, upper extremity strength status, and fine motor performance in children with ALL, as well as potential risk factors that may influence these skills.
Methodology
Search strategy
Studies published before December 2022 were searched on PubMed, Science Direct, Scopus, Web of Science, and PEDro databases using the keywords including (“acute lymphoblastic leukemia” OR “leukemia”) AND (“motor skills” OR “fine motor skills”) AND (“children” OR “childhood”) AND “strength” AND “chemotherapy”).
Selection criteria
Papers meeting the following criteria were included: (1) studies considered children and adolescents diagnosed with ALL both during and/or after treatment; (2) articles assessed fine motor skills and/or upper extremity strength as a primary or a secondary aim, (3) full-text studies available online, and (4) articles published in English.
Intervention studies and articles that included mixed diagnoses and/or did not distinguish between fine and gross motor performance (had one global motor score) were excluded. We also excluded case reports, reviews, theses, or books. In addition, studies that used questionnaires to assess fine motor performance or muscular strength were not included in the current review. Selected studies were screened based on the title and abstract using the inclusion and exclusion criteria. Then, we reviewed the full text of potential studies and we also carefully reviewed the reference list of the included studies to find additional papers. In this review, we followed the Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) guidelines [24].
Results
A total of 418 articles were identified initially through database searching, and then, they went down to 301 citations after duplicate removal (Supplementary file 1). Titles and abstracts were screened by two independent reviewers using the inclusion criteria, excluding 242 citations. Five additional articles were selected for inclusion through reference list review. The same reviewers then independently assessed the full text of 64 articles for inclusion according to the predefined eligibility criteria. Any disagreements between reviewers were resolved through a group discussion with an additional reviewer until the final consensus was reached.
After reviewing the literature obtained during the search of the previous databases, a total of 26 studies were potentially eligible (Fig. 1). Since the timing of assessment would largely affect the results, we classified the selected papers according to whether they were conducted during or after the end of treatment for children with ALL. Twelve studies were applied to children receiving treatment, eleven studies were conducted after completion of therapy, and three studies included children on and off treatment. A detailed description of these studies is presented in Table 1.
Assessment measures
Handgrip strength had been assessed using different types of dynamometers as Jamar, pediatric Nicolas, and digital hand-held dynamometers [25,26,27], in addition to using a blood pressure cuff [16]. Muratt et al. [28] measured the strength of elbow flexors and extensors by a calibrated isokinetic dynamometer.
With regard to the assessment of fine motor skills, the Purdue Pegboard Test (PPT) was the most frequently used measure to evaluate fine motor skills in previous literature [10, 29,30,31,32,33,34]. It can be used in the pediatric population as a brief, easy-to-administer test to assess fine motor dexterity and gross movement of the upper extremity by placing pegs into small holes with the dominant hand, non-dominant hand, and with both hands. The testing board consists of 4 cups at the top and two vertical rows, each row containing 25 small holes. It consists of four subtests and takes approximately 5 to 10 min to administer [35]. Another common measure used to assess motor speed and dexterity is the Grooved Pegboard Test (GPT) [27, 36,37,38,39,40,41]. The test can be used for children above 5 years of age and requires about 5 min to be completed by inserting pegs into 25 holes using the dominant hand, followed by the non-dominant hand. The score is recorded by the time required to place the pegs and calculated separately for each hand [42].
Five studies [37, 43,44,45,46] used the manual dexterity subtest of Movement Assessment Battery for Children–2 (MABC-2) to evaluate the fine motor performance. The MABC-2 is a standardized measure of motor functioning for children aged 3–16 years, including three tasks in the manual dexterity subtest [47].
Few studies used the finger-tapping test [34, 38, 41]. This test was designed to assess the fine motor speed and lateralized coordination using the index finger of the dominant and non-dominant hands [48]. Vainionpaa [49] simply examined the fine motor movements by tapping the fingertips against the thumb tip in a particular sequence, while Goebel et al. [50] assessed fine motor function only in terms of drawing and handwriting abilities using a Digitizing Tablet. Children were allowed to perform three tasks with the dominant hand. Also, the Nine-Hole Peg Test (9-HPT) was used in two studies conducted by Yildiz Kabak et al. [27, 51]. The participant is instructed to place and remove 9 pegs using the dominant hand then the non-dominant hand. The total time to complete the task was recorded in seconds that starts when the first peg is touched and ends when the last peg is dropped back in the container [52].
The Bruininks-Oseretsky Test of Motor Proficiency, Second Edition (BOT-2), is a standardized norm-referenced measure to measure global and fine motor proficiency in children aged from 4 to 21 years [53]. The fine motor form of BOT-2 was administered only in a single study on children with ALL [54]. It consists of the fine manual control and manual coordination composites that include fine motor precision (seven items), fine motor integration (eight items), manual dexterity (five items), and upper limb coordination subtests (seven items). Yildiz Kabak et al. [51] used the short form of the BOT-2 which consists of 14 items that can be used as a screening tool to achieve rapid and easy scoring, reflecting overall motor proficiency [53].
Upper extremity weakness
Despite the limited number of available studies, distal upper extremity weakness has been recorded early in children with ALL, even within 7 days after diagnosis [25]. By the end of the acute treatment phase and before starting the maintenance phase, Akyay et al. [26] reported a significant reduction of grip strength in children with leukemia when compared to a healthy control group. This weakness tends to continue during the maintenance therapy as children had poor handgrip strength that was 60% of the norm-referenced values [27]. A previous study investigating isokinetic upper limb strength in children receiving maintenance therapy revealed that they had weaker elbow extensor strength than their healthy counterparts [28].
Regarding ALL survivors, Akyay et al. [26] and Lofstad et al. [38] reported that after cessation of treatment, grip strength was within normal range. On the contrary, Wright et al. [16] showed that the hand grip strength of the ALL survivors was significantly weaker than that of the comparison group. The difference in the results of this study may be attributed to the high proportion of survivors who received cranial irradiation (about 80% of the sample), which probably caused significant muscle weakness. Another possible factor was the difference in the hand grip evaluation tools used.
Fine motor performance
During treatment
Shortly after diagnosis, no significant differences were found regarding fine motor skills between children with ALL and their siblings [32]. After starting the induction or consolidation phase, children with ALL started to develop fine motor impairments compared to typically developing children [51].
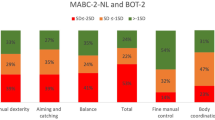
On maintenance therapy, Yildiz Kabak et al. [27] reported that the results of the GPT completion time did not differ from normative values and the 9-HPT completion time was 14% higher than the normative value. In contrast, Sabarre et al. [43] showed that more than 50% of children with ALL scored below average in fine motor skills, with 27% of children having significantly impaired scores. Also, Hanna et al. [54] reported that 67% of children undergoing maintenance therapy had fine motor difficulties in all domains of BOT-2, with 24% falling into the well-below-average category. Such variation of results during the maintenance phase may be attributed to inconsistency in assessment methods used in each study as well as differences in age groups included in these studies.
Because of the long duration of therapy that could reach up to 2 years, longitudinal studies in this period can reflect an in-depth picture of the existence of motor problems. Fine motor speed and dexterity for children had significantly less than the expected values at different phases of treatment [31]. Two longitudinal studies [10, 41] showed a significant improvement in fine motor skills over time that could reach the average range towards the end of therapy while only the performance of the dominant hand remained significantly below expectations [10].
On the contrary, another two longitudinal studies revealed that the incidence of fine motor impairments increased with time [45, 49]. Reinders-Messelink et al. [45] concluded that children with ALL, regardless of treatment phase or post–vincristine therapy, performed more poorly in fine motor skills when compared to a control group. After approximately 1 year of treatment, the percentage of fine motor problems increased to reach about 33%. According to Vainionpaa [49], no fine motor impairments have been detected at the time of diagnosis; however, fine motor impairments started to appear through the course of treatment. After 2–3 years of treatment, the percentage of fine motor impairments remained constant in intermediate and high-risk children receiving cranial irradiation, while the percentage of impairment increased to 33.3% in standard-risk children treated with chemotherapy only.
After completion of treatment
Most cross-sectional studies that investigated the fine motor skills after completion of therapy revealed that ALL survivors had poorer fine motor skills than their matched peers and/or normative values [33, 36,37,38,39, 46, 50]. However, only two studies reported that the fine motor functioning after completion of treatment was in the average range [40, 44]. This may be due to the lack of a control group in these studies, as the results of both studies were only compared to standard values. Regarding the longitudinal analysis, after cessation of chemotherapy, dexterity scores diminished over time and became below the average norm scores [34].
On/off treatment
Few studies have examined fine motor performance (2 studies) in children with ALL during and after completion of their course of treatment. No problems in neuropsychological outcomes have been detected at the time of diagnosis or after 3–6 months of completion of treatment, but children with ALL showed a significant decrease only in complex fine motor functions compared to healthy siblings 4.5 years after diagnosis [30]. Otherwise, Moore et al. [29] reported that fine motor dexterity improved over time but was significantly below the age-adjusted norms either at remission or after 3 years from the baseline evaluation.
Factors that might affect fine motor performance
Cumulative chemotherapeutic doses
Along the course of treatment, no relationship was found between fine motor performance and cumulative doses of vincristine [43, 45, 54], methotrexate, or dexamethasone [54]. A longitudinal study conducted by Dowel et al. [41] revealed that the fine motor improvements significantly corresponded with the decrease in the number of vincristine and L-asparaginase administrations while they found no relation with respect to methotrexate. In addition, among ALL survivors, no relation was detected between fine motor functions and vincristine administration doses [46].
Gender effect
Two studies reported that girls had better manual dexterity scores than boys [30, 46], while only one study demonstrated a significant outperformance of boys in different fine motor domains [54]. Otherwise, most studies did not identify any significant difference in hand strength [16, 26] or fine motor skills in relation to gender in ALL survivors [37, 40, 44, 50]. The variation in results seems reasonable because these studies examined children with ALL at different ages and phases of treatment and most were after the end of treatment. There is only one study [54] that addressed the effect of gender on fine motor skills at an early age during treatment, while the remaining studies included children and adolescents of ALL survivors.
Age at diagnoses/assessment
Oswald and Bo [37] found that the older the age at diagnosis, the lower the performance on the Grooved Pegboard for both hands in ALL survivors. The results of this study should be interpreted with caution because the sample included a wide age range (5–18 years) of participants who were actively receiving maintenance treatment in addition to children who had completed their medical treatment. Goebel et al. [50] did not find any correlation between younger age and impaired motor performance; however, they found that older age at diagnosis was associated with higher variability on the more complex task of digitizing tablet. Other previous studies revealed no significant association between fine motor impairments and age at diagnosis [30, 40, 44] and/or age at assessment [44, 54].
Risk classification
Few studies addressed the effect of different ALL risk groups on muscle strength and fine motor skills. During the maintenance treatment, Hanna et al. [54] reported that the overall fine motor performance of children treated according to the standard-risk protocol was significantly better than that of the low-risk group. ALL risk groups did not affect manual dexterity along the course of treatment [31] or hand strength after completion of therapy [16].
Discussion
Pediatric cancer treatment has negative sequelae that may impair levels of physical activity and fitness in pediatric cancer survivors compared to their healthy peers [55]. Alterations in motor performance, limitations in daily living activities, and muscle weakness were reported as the main priorities for physical rehabilitation of children and adolescents with cancer [56]. Incorporating the entire rehabilitation team and evaluating these priorities at the beginning, during, and after cancer treatment are beneficial to improve patient care by proactively identifying motor deficits. In addition, early and continuous rehabilitation allows for appropriate motor development as well as improvement of the child’s daily living activities and fine motor skills that may be affected by distal muscle weakness or atrophy [57].
In the current review, we aimed to discuss the state of upper extremity weakness in children diagnosed with ALL, as sufficient muscle strength is crucial to ensure successful fine and executive functions [58] and is directly correlated with functional motor tasks [59]. In this review, though there was no strong evidence that ALL survivors experienced muscle weakness after cessation of therapy, early muscle weakness in the upper extremities was reported along the course of treatment [25,26,27,28]. Therefore, these children may benefit from periodic evaluation of upper extremity strength and physical therapy intervention. Nielsen et al. [60] reported that children with ALL who underwent 3 months of supervised physical activity intervention maintained their baseline levels of handgrip strength. Inappropriate space for rehabilitation and lack of funding or resources are the most commonly identified barriers for physical rehabilitation [56].
Fine motor skills require integration of manual abilities, cognitive skills, spatial organization, and visual-motor coordination, and have been identified as an integral part of elementary school education [14]. Participation in play or school may be affected in children with ALL due to hospitalization and fatigue [43, 56]. Frequent absence from school and difficulties in fine motor activities can decrease school-based participation that may exhibit significant difficulties with academic achievement in ALL survivors [23]. Fine motor skills are not necessarily evaluated only in hospitals and clinics, but can also be addressed at school and the necessary modifications can be made in the classroom by consulting an occupational therapist.
Therefore, evaluation of fine motor abilities is a challenge and requires an aggregate measure rather than separate component skills [61, 62]. A considerable heterogeneity was observed regarding the methods of evaluating children’s fine motor skills that ranged from specific tests to assess motor speed/dexterity (e.g., Finger tapping task, Purdue Pegboard Test, Grooved Pegboard test) to comprehensive tests that measure more than one aspect of fine motor skills (e.g., BOT-2, MABC-2).
According to Knight and colleagues [63], tasks of a timed measure nature (Purdue Pegboard test or Finger tapping test) that assess fine motor coordination fail to control for the impact of psychomotor speed which has been affected in those children. Also, the BOT-2 brief form is a screening tool and may not be as accurate as the complete form to detect the mild impairments that may have an impact on the participation in physical activity [64]. In addition, its final score does not distinguish between gross and fine motor scores, although differences between them are important to consider in clinical assessments and designing rehabilitation interventions [65].
The development of central and peripheral nervous systems of those children may be subtly affected by chemotherapeutic agents (e.g., Vincristine; Methotrexate) which may be reflected in the form of reduced motor performance on functional measures [63]. Vincristine is commonly used as a part of standard treatment of childhood ALL treatment protocols [64]. Peripheral neuropathy is the most frequent side effect of vincristine toxicity that presents usually within the first 3 months of treatment. It causes paresthesia and pain in the hands and feet, followed by muscle weakness, particularly in wrist extensors and dorsiflexors [66]. Accordingly, the impaired fine motor performance may be related to the peripheral neuropathic effects of vincristine [23, 50].
Although most studies in this review confirmed that these children had fine motor impairments throughout the treatment period, we could not confirm the course of progression of these skills over time, as some longitudinal studies indicated their improvement, while others indicated their deterioration. As for post-treatment, the included studies assessed fine motor skills at various times ranging from a few months to several years off-treatment, making it difficult to confirm the extent to which these skills have deteriorated.
Some research gaps were identified in our scoping review. First, there could be some risk of bias in the results of this review due to the lack of a control group in several studies [10, 25, 27, 29, 31, 40, 41, 43, 44, 49]. Indeed, ten of the included studies did not have a control group, but they compared their results with normative values. Gaul and Issartel [67] claimed that during early school age children’s proficiency in fine motor skills did not progress at the rate expected by normative data. Accordingly, further studies with matched age- and sex-control groups are highly recommended.
Second, the articles included in this review encompassed a wide range of different assessment measures that precluded the interpretation and generalization of results. Third, the relatively broad age range of the included studies including children and adolescents added further to the complexity of interpretation and analysis. Another possible limitation is the great variation of the evaluation time across reviewed studies, either during or after treatment. In addition, language bias might exist as the review was restricted to articles published in English. Moreover, one study in this review included ALL patients/survivors who were either actively receiving maintenance therapy or off-medical treatment.
Recommendations
The findings from this scoping review highlight some important areas for future research in children diagnosed with ALL. Regular evaluation of fine motor performance along the course of chemotherapy could identify early impairments. The multidisciplinary cancer team, including physical and occupational therapists, has a crucial role in detecting and providing the optimal intervention program. Upper extremity strengthening and occupational therapy training are prompted to preserve muscle strength and to minimize future fine motor problems along the course of chemotherapeutic treatment. More work is needed to identify comprehensive, clinically relevant measurement tools that can truly reflect a real picture of all aspects of fine motor performance, including school-related fine motor tasks where children spend a large portion of their time. Evaluation of neuropathy induced by chemotherapeutic agents in children and adolescents with ALL during and after completion of treatment needs to be addressed in upcoming studies. In addition to the main potential risk factors such as age, gender, and risk classification, some other external factors may negatively impact the development of fine motor skills, such as the duration of hospitalization, the effect of fatigue, and time away from school. Consequently, addressing the causal relationship between the potential risk factors that may influence fine motor performance in future studies is recommended.
Conclusion
In conclusion, this study provides an updated summary of fine motor difficulties in the pediatric population with ALL. Results from this review indicate that those children experienced fine motor limitations and upper extremity weakness either during or after cessation of treatment. However, there is little evidence regarding the risk factors that may impair muscle strength and motor performance.
Overall, early physical rehabilitation is recommended once these children are admitted in an attempt to minimize the complications of therapeutic agents.
Data Availability
The data supporting the findings of this study are available within the article or its supplementary information.
References
Ness KK, Armenian SH, Kadan-Lottick N, Gurney JG (2011) Adverse effects of treatment in childhood acute lymphoblastic leukemia: general overview and implications for long-term cardiac health. Expert Rev Hematol 4:185–197. https://doi.org/10.1586/ehm.11.8
Coebergh JWW, Reedijk AMJ, de Vries E et al (2006) Leukaemia incidence and survival in children and adolescents in Europe during 1978–1997. Report from the Automated Childhood Cancer Information System project. Eur J Cancer 42:2019–2036. https://doi.org/10.1016/j.ejca.2006.06.005
Plasschaert SLA, Kamps WA, Vellenga E et al (2004) Prognosis in childhood and adult acute lymphoblastic leukaemia: a question of maturation? Cancer Treat Rev 30:37–51. https://doi.org/10.1016/S0305-7372(03)00140-3
Williams LA, Richardson M, Marcotte EL et al (2019) Sex ratio among childhood cancers by single year of age. Pediatr Blood Cancer 66:e27620. https://doi.org/10.1002/pbc.27620
Li SY, Ye JY, Meng FY et al (2015) Clinical characteristics of acute lymphoblastic leukemia in male and female patients: a retrospective analysis of 705 patients. Oncol Lett 10:453–458. https://doi.org/10.3892/ol.2015.3202
Hunger SP, Lu X, Devidas M et al (2012) Improved survival for children and adolescents with acute lymphoblastic leukemia between 1990 and 2005: a report from the children’s oncology group. J Clin Oncol 30:1663–1669. https://doi.org/10.1200/JCO.2011.37.8018
Varedi M, McKenna R, Lamberg EM (2017) Balance in children with acute lymphoblastic leukemia. Pediatr Int 59:293–302. https://doi.org/10.1111/ped.13141
Peterson J, Darling T (2018) Childhood cancer and treatment effects on motor performance. Int J Exerc Sci 11:657–668
Harila-Saari AH, Vainionpää LK, Kovala TT et al (1998) Nerve lesions after therapy for childhood acute lymphoblastic leukemia. Cancer 82:200–207. https://doi.org/10.1002/(SICI)1097-0142(19980101)82:1%3c200::AID-CNCR25%3e3.0.CO;2-5
Hockenberry MJ, Krull KR, Insel KC et al (2015) Oxidative stress, motor abilities, and behavioral adjustment in children treated for acute lymphoblastic leukemia. Oncol Nurs Forum 42:542–549. https://doi.org/10.1188/15.ONF.542-549
Payne VG, Chang SH (2020) Fine motor development. In: Hupp S, Jewell J (eds) The Encyclopedia of Child and Adolescent Development. Wiley, pp 1–10
Cools W, De MK, Samaey C, Andries C (2009) Movement skill assessment of typically developing preschool children: a review of seven movement skill assessment tools. J Sports Sci Med 8:154–168
Comuk-Balci N, Bayoglu B, Tekindal A et al (2016) Screening preschool children for fine motor skills: environmental influence. J Phys Ther Sci 28:1026–1031. https://doi.org/10.1589/jpts.28.1026
McHale K, Cermak SA (1992) Fine motor activities in elementary school: preliminary findings and provisional implications for children with fine motor problems. Am J Occup Ther 46:898–903. https://doi.org/10.5014/ajot.46.10.898
Marr D, Cermak S, Cohn ES, Henderson A (2003) Fine motor activities in head start and kindergarten classrooms. Am J Occup Ther 57:550–557. https://doi.org/10.5014/ajot.57.5.550
Wright MJ, Halton JM, Martin RF, Barr RD (1998) Long-term gross motor performance following treatment for acute lymphoblastic leukemia. Med Pediatr Oncol 31:86–90. https://doi.org/10.1002/(SICI)1096-911X(199808)31:2%3c86::AID-MPO7%3e3.0.CO;2-V
Warner JT (2008) Body composition, exercise and energy expenditure in survivors of acute lymphoblastic leukaemia. Pediatr Blood Cancer 50:456–461
Brookman A, McDonald S, McDonald D, Bishop DVM (2013) Fine motor deficits in reading disability and language impairment: same or different? PeerJ 1:1–20. https://doi.org/10.7717/peerj.217
Desrosiers J, Bravo G, Hébert R et al (1994) Validation of the Box and Block Test as a measure of dexterity of elderly people: Reliability, validity, and norms studies. Arch Phys Med Rehabil 75:751–755. https://doi.org/10.1016/0003-9993(94)90130-9
Iyer NS, Balsamo LM, Bracken MB, Kadan-Lottick NS (2015) Chemotherapy-only treatment effects on long-term neurocognitive functioning in childhood ALL survivors: a review and meta-analysis. Blood 126:346–353. https://doi.org/10.1182/blood-2015-02-627414
Green JL, Knight SJ, McCarthy M, De Luca CR (2013) Motor functioning during and following treatment with chemotherapy for pediatric acute lymphoblastic leukemia. Pediatr Blood Cancer 60:1261–1266. https://doi.org/10.1002/pbc.24537
Peterson CC, Johnson CE, Ramirez LY et al (2008) A meta-analysis of the neuropsychological sequelae of chemotherapy-only treatment for pediatric acute lymphoblastic leukemia. Pediatr Blood Cancer 51:99–104. https://doi.org/10.1002/pbc.21544
Moleski M (2000) Neuropsychological, neuroanatomical, and neurophysiological consequences of CNS chemotherapy for acute lymphoblastic leukemia. Arch Clin Neuropsychol 15:603–630. https://doi.org/10.1016/S0887-6177(99)00050-5
Tricco AC, Lillie E, Zarin W et al (2018) prisma extension for scoping reviews (prisma-scr): checklist and explanation. Ann Intern Med 169:467–473. https://doi.org/10.7326/M18-0850
Ness KK, Kaste SC, Zhu L et al (2015) Skeletal, neuromuscular and fitness impairments among children with newly diagnosed acute lymphoblastic leukemia. Leuk Lymphoma 56:1004–1011. https://doi.org/10.3109/10428194.2014.944519
Akyay A, Olcay L, Sezer N, Sönmez ČA (2014) Muscle strength, motor performance, cardiac and muscle biomarkers in detection of muscle side effects during and after acute lymphoblastic leukemia treatment in children. J Pediatr Hematol Oncol 36:594–598. https://doi.org/10.1097/MPH.0000000000000067
Yildiz Kabak V, Ipek F, Unal S et al (2021) An evaluation of participation restrictions and associated factors via the ICF-CY framework in children with acute lymphoblastic leukemia receiving maintenance chemotherapy. Eur J Pediatr 180:1081–1088. https://doi.org/10.1007/s00431-020-03833-y
Muratt MD, Perondi MB, Greve JMDA et al (2011) Strength capacity in young patients who are receiving maintenance therapy for acute lymphoblastic leukemia: a case-control study. Clinics 66:1277–1281. https://doi.org/10.1590/S1807-59322011000700026
Moore IM, Lupo PJ, Insel K et al (2016) Neurocognitive predictors of academic outcomes among childhood leukemia survivors. Cancer Nurs 39:255–262. https://doi.org/10.1097/NCC.0000000000000293
Jansen NCAJ, Kingma A, Schuitema A et al (2008) Neuropsychological outcome in chemotherapy-only-treated children with acute lymphoblastic leukemia. J Clin Oncol 26:3025–3030. https://doi.org/10.1200/JCO.2007.12.4149
Hockenberry M, Krull K, Moore K et al (2007) Longitudinal evaluation of fine motor skills in children with leukemia. J Pediatr Hematol Oncol 29:535–539. https://doi.org/10.1097/MPH.0b013e3180f61b92
Jansen NC, Kingma A, Tellegen P et al (2005) Feasibility of neuropsychological assessment in leukaemia patients shortly after diagnosis: directions for future prospective research. Arch Dis Child 90:301–304. https://doi.org/10.1136/adc.2004.051839
Kaemingk KL, Carey ME, Moore IM et al (2004) Math weaknesses in survivors of acute lymphoblastic leukemia compared to healthy children. Child Neuropsychol 10:14–23. https://doi.org/10.1076/chin.10.1.14.26240
Kingma A, Van Dommelen RI, Mooyaart EL et al (2001) Slight cognitive impairment and magnetic resonance imaging abnormalities but normal school levels in children treated for acute lymphoblastic leukemia with chemotherapy only. J Pediatr 139:413–420. https://doi.org/10.1067/mpd.2001.117066
Gardner RA, Broman M (1979) The Purdue pegboard: normative data on 1334 school children. J Clin Child Psychol 8:156–162. https://doi.org/10.1080/15374417909532912
van der Plas E, Spencer Noakes TL, Butcher DT et al (2021) Cognitive and behavioral risk factors for low quality of life in survivors of childhood acute lymphoblastic leukemia. Pediatr Res 90:419–426. https://doi.org/10.1038/s41390-020-01230-7
Oswald KA, Bo J (2020) Motor functioning and associated cognitive outcomes in pediatric survivors of acute lymphoblastic leukemia. Child Neuropsychol 26:597–611. https://doi.org/10.1080/09297049.2019.1676406
Lofstad GE, Reinfjell T, Weider S et al (2019) Neurocognitive outcome and compensating possibilities in children and adolescents treated for acute lymphoblastic leukemia with chemotherapy only. Front Psychol 10:1–12. https://doi.org/10.3389/fpsyg.2019.01027
Van Der Plas E, Erdman L, Nieman BJ et al (2018) Characterizing neurocognitive late effects in childhood leukemia survivors using a combination of neuropsychological and cognitive neuroscience measures. Child Neuropsychol 24:999–1014. https://doi.org/10.1080/09297049.2017.1386170
Balsamo LM, Sint KJ, Neglia JP et al (2016) The association between motor skills and academic achievement among pediatric survivors of acute lymphoblastic leukemia. J Pediatr Psychol 41:319–328. https://doi.org/10.1093/jpepsy/jsv103
Dowell RE, Copeland DR, Judd BW (1989) Neuropsychological effects of chemotherapeutic agents. Dev Neuropsychol 5:17–24. https://doi.org/10.1080/87565648909540419
IInstrument L (2002) Grooved pegboard test user instructions. Lafayette, IN: Lafayette Instrument Company
Sabarre CL, Rassekh SR, Zwicker JG (2014) Vincristine and fine motor function of children with acute lymphoblastic leukemia. Can J Occup Ther 81:256–264. https://doi.org/10.1177/0008417414539926
De Luca CR, McCarthy M, Galvin J et al (2013) Gross and fine motor skills in children treated for acute lymphoblastic leukaemia. Dev Neurorehabil 16:180–187. https://doi.org/10.3109/17518423.2013.771221
Reinders-Messelink H, Schoemaker M, Snijders T et al (1999) Motor performance of children during treatment for acute lymphoblastic leukemia. Med Pediatr Oncol 33:545–550. https://doi.org/10.1002/(SICI)1096-911X(199912)33:6%3c545::AID-MPO4%3e3.0.CO;2-Y
Reinders-Messelink HA, Schoemaker MM, Hofte M et al (1996) Fine motor and handwriting problems after treatment for childhood acute lymphoblastic leukemia. Med Pediatr Oncol 27:551–555. https://doi.org/10.1002/(SICI)1096-911X(199612)27:6%3c551::AID-MPO8%3e3.0.CO;2-K
Henderson SE, Sugden DA, Barnett A (2007) Movement assessment battery for children–second edition: examiner’s manual, 2nd edn. Pearson, San Antonio, TX
Reitan RM, Wolfson D (1985) The Halstead-Reitan neuropsychological test battery. Neuropsychology Press, Tucson, AZ
Vainionpää L (1993) Clinical neurological findings of children with acute lymphoblastic leukaemia at diagnosis and during treatment. Eur J Pediatr 152:115–119. https://doi.org/10.1007/BF02072486
Goebel AM, Koustenis E, Rueckriegel SM et al (2019) Motor function in survivors of pediatric acute lymphoblastic leukemia treated with chemotherapy-only. Eur J Paediatr Neurol 23:304–316. https://doi.org/10.1016/j.ejpn.2018.12.005
Yildiz Kabak V, Ekinci Y, Atasavun Uysal S et al (2021) Motor and basic cognitive functions in children with acute lymphoblastic leukemia undergoing induction or consolidation chemotherapy. Percept Mot Skills 128:1091–1106. https://doi.org/10.1177/00315125211002065
Smith YA, Hong E, Presson C (2000) Normative and validation studies of the nine-hole peg test with children. Percept Mot Skills 90:823–843. https://doi.org/10.2466/pms.2000.90.3.823
Bruininks R, Bruininks B (2005) Bruininks-Oseretsky test of motor proficiency, 2nd edn. NCS Pearson, Minneapolis
Hanna S, Elshennawy S, El-Ayadi M, Abdelazeim F (2020) Investigating fine motor deficits during maintenance therapy in children with acute lymphoblastic leukemia. Pediatr Blood Cancer 67:1–9. https://doi.org/10.1002/pbc.28385
Antwi GO, Jayawardene W, Lohrmann DK, Mueller EL (2019) Physical activity and fitness among pediatric cancer survivors: a meta-analysis of observational studies. Support Care Cancer 27:3183–3194. https://doi.org/10.1007/s00520-019-04788-z
Ospina PA, Wiart L, Eisenstat DD, McNeely ML (2020) Physical rehabilitation practices for children and adolescents with cancer in Canada. Physiother Canada 72:207–216. https://doi.org/10.3138/ptc-2018-0077
Lenker H (2021) Multidisciplinary rehabilitation within pediatric cancer care: a holistic approach. Rehabil Oncol 39:E12–E14. https://doi.org/10.1097/01.REO.0000000000000263
Hammed AI, Agbonlahor EI (2017) Relationship between anthropometrics and handgrip strength among Nigerian school children. Biomed Hum Kinet 9:51–56. https://doi.org/10.1515/bhk-2017-0008
Alaniz ML, Galit E, Necesito CI, Rosario ER (2015) Hand strength, handwriting, and functional skills in children with autism. Am J Occup Ther 69:6904220030p1-6904220030p9. https://doi.org/10.5014/ajot.2015.016022
Nielsen MKF, Christensen JF, Frandsen TL et al (2018) Testing physical function in children undergoing intense cancer treatment—a RESPECT feasibility study. Pediatr Blood Cancer 65:1–9. https://doi.org/10.1002/pbc.27100
Cameron CE, Brock LL, Murrah WM et al (2012) Fine motor skills and executive function both contribute to kindergarten achievement. Child Dev 83:1229–1244. https://doi.org/10.1111/j.1467-8624.2012.01768.x
Bos AF, Van Braeckel KNJA, Hitzert MM et al (2013) Development of fine motor skills in preterm infants. Dev Med Child Neurol 55:1–4. https://doi.org/10.1111/dmcn.12297
Knight S, McCarthy M, Anderson V et al (2014) Visuomotor function in children treated for acute lymphoblastic leukaemia with chemotherapy only. Dev Neuropsychol 39:101–112. https://doi.org/10.1080/87565641.2013.860980
Tay CG, Lee VWM, Ong LC et al (2017) Vincristine-induced peripheral neuropathy in survivors of childhood acute lymphoblastic leukaemia. Pediatr Blood Cancer 64:1–7. https://doi.org/10.1002/pbc.26471
Hung SH, Rankin A, Virji-Babul N et al (2017) Associating physical activity levels with motor performance and physical function in childhood survivors of acute lymphoblastic leukemia. Physiother Canada 69:57–64. https://doi.org/10.3138/ptc.2015-67LHC
Soffietti R, Trevisan E, Rudà R (2014) Neurologic complications of chemotherapy and other newer and experimental approaches. Handb Clin Neurol 121:1199–1218. https://doi.org/10.1016/B978-0-7020-4088-7.00080-8
Gaul D, Issartel J (2016) Fine motor skill proficiency in typically developing children: on or off the maturation track? Hum Mov Sci 46:78–85. https://doi.org/10.1016/j.humov.2015.12.011
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
F.A. and M.E. contributed to review conception and design. The literature search, screening, and filtering were conducted by M.E. and S.H., while F.A. was the third reviewer of conflict articles. The first draft of the manuscript was written by S.H. and all authors commented on previous versions of it. All authors made substantial contributions to the interpretation of the data, read, and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
No ethics approval was needed for this study.
Consent to participate
N/A.
Consent for publication
N/A.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hanna, S., El-Ayadi, M. & Abdelazeim, F. Identifying fine motor difficulties in children with acute lymphoblastic leukemia: a scoping review. Support Care Cancer 32, 488 (2024). https://doi.org/10.1007/s00520-024-08667-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00520-024-08667-0