Abstract
Purpose
To provide evidence-based recommendations on the management of malignant bowel obstruction (MBO) for patients with advanced cancer.
Methods
The Multinational Association for Supportive Care in Cancer (MASCC) MBO study group conducted a systematic review of databases (inception to March 2021) to identify studies about patients with advanced cancer and MBO that reported on the following outcomes: symptom management, bowel obstruction resolution, prognosis, overall survival, and quality of life. The review was restricted to studies published in English, but no restrictions were placed on publication year, country, and study type. As per the MASCC Guidelines Policy, the findings were synthesized to determine the levels of evidence to support each MBO intervention and, ultimately, the graded recommendations and suggestions.
Results
The systematic review identified 17,656 published studies and 397 selected for the guidelines. The MASCC study group developed a total of 25 evidence-based suggestions and recommendations about the management of MBO-related nausea and vomiting, bowel movements, pain, inflammation, bowel decompression, and nutrition. Expert consensus-based guidance about advanced care planning and psychosocial support is also provided.
Conclusion
This MASCC Guideline provides comprehensive, evidence-based recommendations about MBO management for patients with advanced cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
MBO is a severe complication in advanced cancer [1]. MBO was defined in the International Conference on MBO and Clinical Protocol Committee as (i) clinical evidence of bowel obstruction (via history, physical, and/or radiological examination), (ii) bowel obstruction beyond the ligament of Treitz, and (iii) diagnosis of intra-abdominal cancer with an incurable disease, or a non-intra-abdominal primary cancer with the clear intraperitoneal disease [1]. The algorithm of care for patients with MBO is not uniform and often differs according to clinical factors (e.g., prognosis) and between institutions and countries.
In this article, the MASCC MBO multidisciplinary study group presents comprehensive, evidence-based recommendations about MBO management for patients with advanced cancer and the methodology used to develop these recommendations.
Background
Etiology
MBO most frequently occurs when patients have advanced cancers that originate in the abdomen or pelvis. Though the incidence of MBO is not well established, based on retrospective and autopsy-based studies, MBO is estimated to occur in 10–28% of patients with gastrointestinal cancers and up to 51% of patients with advanced ovarian cancer [2]. Limitations of these estimates include patient population selection and nonhomogeneous criteria to diagnose MBO.
The mechanisms of MBO development are multi-factorial and can be divided into two main groups: mechanical and functional obstruction [2, 3]. Causes of mechanical obstruction include extrinsic obstruction of the lumen by pathology, such as mesenteric and omental masses, adhesions, and fibrosis; intra-luminal obstruction from tumor growth in the bowel; and intra-mural obstruction by tumor within the bowel wall, which impairs motility. Whereas functional obstruction is a result of motility disorders, which can be due to tumor infiltration of mesentery, nerves and/or celiac and enteric plexus, and paraneoplastic syndromes. Further, nonmalignant factors may induce or worsen bowel obstruction (BO) in patients with advanced cancer, including constipation/fecal impaction, pharmacological (i.e., opioids, intra-peritoneal chemotherapy), fibrosis, and adhesions from prior surgery and radiotherapy.
Pathophysiology and symptoms
MBO can occur in the small or large bowel, with small BO being more common [3, 4]. The obstruction can be partial or complete and can occur at single or multiple transition points. MBO causes reduction or absence of movements of the intestinal content and bowel distension [3, 4]. The accumulation of content in the intestinal lumen increases the epithelial surface area and prompts an accumulation of gastric, pancreatic, biliary secretions, water, and salt, which damages the intestinal epithelium and triggers an inflammatory response with intestinal edema, hyperemia, and production of inflammatory mediators (i.e., prostaglandins, vasoactive intestinal polypeptide, and nociceptive mediators). Bacterial overgrowth and translocation are important mechanisms in the development of symptoms. The cumulative impact of these events results in abdominal pain, cramps, distention, nausea, vomiting, absence of gas and stool passage, and, occasionally, overflow diarrhea. MBO symptoms usually start gradually and become more frequent and severe when a complete obstruction occurs. Re-obstruction and malnutrition are common, with malnutrition being an independent predictor of poor survival in this population [5,6,7,8].
Diagnosis
MBO is a clinical diagnosis that is confirmed with radiological imaging. Historically, abdominal x-rays were recommended to be the initial imaging modality, but their sensitivity to detect MBO is moderate and poses challenges in detecting the exact site, cause, or complications derived from MBO [9]. Contrast computed tomography (CT) is more valuable as it provides diagnostic precision [4]. The American College of Radiology recommends the use of CT of the abdomen and pelvis with intravenous (IV) contrast for patients with suspected acute small BO and either a CT abdomen and pelvis with IV contrast or a CT enterography for patients suspected to have intermittent or low-grade small BO [9].
Advanced care planning and goals of care conversations
When patients with advanced cancers are diagnosed with MBO, clinicians should acknowledge that medical decisions are directed at extending life, minimizing distressing symptoms, and maximizing quality of life. Care should be holistic and person-centered with focus on the interrelationship between physical, psychosocial, and spiritual issues [10]. Clinicians should encourage patients to substitute decision-makers with who they can communicate their values and goals to assist them with medical decision-making when they no longer have the capacity. In some jurisdictions, written advance care documents are legislated and may include options of appointing a formal decision-maker on the loss of capacity, statements about values and goals, and even binding refusals of specific interventions.
Before offering interventions for MBO management, clinicians should take into consideration the risks associated with the interventions in addition to the patient’s estimated prognosis, performance status, comorbidities, location of care and ease of further assessment and support (e.g., home parenteral nutrition programs may not be universally available). Financial implications may also impact whether an intervention might be offered or undertaken. Furthermore, patients may have strongly held views about the interventions that they would accept and their decisions can change over time, especially when the benefit of therapy or intervention may no longer exceed the burden, and cessation may be considered appropriate by their clinicians or even desired by the patient. Clinicians are encouraged to discuss these possible scenarios with patients and their families before commencing interventions, such as total parenteral nutrition. As patients approach the end of their lives, their goals of care may shift from disease management and life prolongation to symptom management and quality of life [11].
The interventions that might be offered or considered clinically appropriate may depend on the estimated prognosis of the patient. Physician prediction of survival is known to be inaccurate and often overly optimistic. Objective factors associated with a short prognosis include deteriorating performance status and the presence of other symptoms, potentially indicative of progressive malignant disease in other organs [12]. Laboratory findings associated with systemic inflammatory response (e.g., elevated C-reactive protein), reduced albumin, and leucocytosis are also associated with poor prognosis [13]. Prognostic models incorporating physician prediction of survival and clinical and laboratory factors improve the accuracy of clinical prediction [14].
The treating clinician should offer sensitive discussions with patients and their families about different treatment options that may be possible, what outcomes might be reasonably expected, possible adverse effects or burdens, and how to measure whether such interventions may provide some success in terms of longevity, symptom control or improvement in quality of life. Assessments of a patient’s illness understanding and decision-making wishes around disclosure and acknowledging emotional responses are essential [15]. Exploring these issues requires sensitivity and excellent communication skills. Generally, personal autonomy and the right to be involved in all medical decision-making is a widely held, but not a universal, principle. Some patients may choose not to confront their own mortality or lack the capacity to do so. In certain cultures, patients may choose to defer medical decision-making to their family. Goals of care conversations require careful exploration, always with the patient’s best interest being foremost.
Methods
Literature review
An information specialist (RF) conducted a systematic literature search in Medline ALL (Medline and Epub Ahead of Print and In-Process, In-Data-Review, and Other Non-Indexed Citations), Embase Classic +Embase, Cochrane Central Register of Controlled Trial, Cochrane Database of Systematic Reviews, PsycInfo, all from the OvidSP platform; Scopus from Elsevier, and Global Index Medicus (LILACS, WPRIM, IMEMR, IMSEAR, and AIM) from WHO. The literature search was executed from the inception of each database to March 2021 with no language limitations. Each search strategy comprised a combination of controlled vocabulary terms and text words, adapting the database-specific search syntax. Where applicable, the search was restricted to human studies, adults ≥18 years of age and excluded books, conference abstracts, and dissertations. Appendix A presents the Medline ALL search.
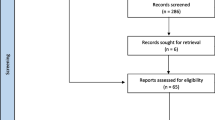
Records obtained were stored on EndNote citation software, following which duplicates were removed and studies uploaded onto Covidence. Abstracts were screened for eligibility, and then full-text articles were assessed. Any discrepancies in study selection were resolved through consensus. Inclusion criteria were studies about people with advanced cancers (any type) with MBO (small and/or large bowel) that examined MBO interventions and reported on outcomes related to symptom management, BO resolution, prognosis, overall survival, and/or quality of life. All types of primary research studies (with or without comparators) were included. Studies about anti-cancer treatments (e.g., chemotherapy, radiation) for MBO management and non-English studies were excluded. In total, 17,565 studies were identified, and 3450 duplicates removed. From 14,115 studies that were screened against title and abstract, 13,561 were excluded. A total of 554 studies were assessed for full-text eligibility, and 397 studies were included for this review. Figure 1 presents the PRISMA flow diagram. Using the 2018 MASCC Guidelines Policy: recommendations for MASCC guideline construction and the endorsement of externally generated guidelines, the level of evidence (Table 1) for each MBO intervention was determined and synthesized into suggestions or recommendations with gradings (Table 2). “Suggestions” are used for statements that are based on level III, IV, or level V evidence. “Recommendations” are reserved for statements that are based on level I or level II evidence. Whereas “no guideline possible” are used when there is insufficient evidence on which to base a guideline. This implies that there is little or no evidence regarding the practice in question or that the panel lacks consensus on the interpretation of existing evidence.
Suggestions/recommendations
The following sections present each MBO management recommendation and its associated evidence (Table 3). In complete MBO, medication should be administered intravenously or subcutaneously if available.
Anti-emetics
The 2021 MASCC guideline about select pharmacologic management of nausea and vomiting in MBO was released in August 2021 as an update to a previous guideline published in 2017 [16]. The following sections and Appendix B provide suggestions/recommendations for all anti-emetic drug classes reportedly used for MBO management and their associated existing evidence:
Anticholinergics
Suggestion: The benefit of anticholinergics (hyoscine butylbromide) may be inferior to octreotide to reduce vomiting in MBO. (Level of evidence: III; Grade: D).
Hyoscine (scopolamine) butylbromide is an anticholinergic agent that reduces gastrointestinal secretions. A 2016 systematic review [17] identified four randomized clinical trials with high Cochrane risk of bias that found somatostatin analog (e.g., octreotide) were more effective than hyoscine butylbromide in reducing nausea and vomiting [18,19,20,21]. Three case reports reported mixed findings, where two reported hyoscine butylbromide in combination with other drugs (e.g., octreotide, dexamethasone) was effective in reducing vomiting [22, 23], and one that reported that it was ineffective for controlling vomiting [24]. Since the publication of that review, no new randomized clinical trials have been published. An alternative anticholinergic that can reportedly be used is glycopyrrolate. However, this review only identified one case report that reported that glycopyrrolate reduced nausea and vomiting in MBO [25]. Appendix B presents the characteristics of all included studies about anticholinergic use in MBO.
Butyrophenone antipsychotic
Suggestion: Haloperidol, a butyrophenone antipsychotic, may be an effective anti-emetic in MBO, particularly for complete MBO (level of evidence: IV; grade: B),
Butyrophenone antipsychotics are commonly used as anti-psychotics for delirium management but are also used for the management of nausea and vomiting in MBO. The main butyrophenone used is haloperidol. The review identified two cross-sectional studies [26, 27] and two case reports/series that reported on haloperidol’s use in MBO [25, 28]. All studies suggested that haloperidol effectively relieve nausea and vomiting in MBO and is the preferred anti-emetic in complete BO [25,26,27,28].
Dopamine antagonist prokinetic
Suggestion: Dopamine antagonist prokinetic drugs (e.g., metoclopramide, domperidone) may be effective for the management of nausea, vomiting, and restoring bowel transit time in partial MBO. Due to the potential increased risk of bowel perforation, it likely should be avoided in complete MBO. (level of evidence: III; grade B)
Dopamine antagonist prokinetic drugs block dopamine receptors. They increase lower esophageal sphincter pressure, gastric motility, and, therefore, gastric emptying [29]. The main prokinetic drugs used in MBO management are domperidone and metoclopramide. We identified two cross-sectional studies [26, 27] and three case reports/series [24, 30, 31] published between 1985 and 2016, that reported on their use in MBO. Of these studies, three studies found metoclopramide was effective for nausea, vomiting, and restoring intestinal transit time [27, 30, 31], particularly in incomplete BO [27]. However, two studies found that metoclopramide was ineffective for relieving these symptoms [24, 26]. Only one study reported on the use of domperidone and found it ineffective for control of vomiting [26].
Histamine (H 1 ) antagonist
Suggestion: Histamine H 1 antagonists (e.g., dimenhydrinate, cyclizine) may be an effective anti-emetic in complete MBO (level of evidence: IV; grade D).
Histamine H1 antagonists are a drug class that is primarily used for the management of motion sickness from vestibular stimulation. The main histamine H1 antagonist that is used is dimenhydrinate. This review identified only one cross-sectional study published in 1994 that reported histamine H1 antagonist use for nausea management in complete BO [27].
Phenothiazines
Suggestion: Phenothiazines (e.g., chlorpromazine) may reduce nausea and vomiting in MBO (level of evidence: IV; grade: D).
Phenothiazines are first-generation antipsychotic agents that can be used for the prevention and control of nausea and vomiting. Examples of phenothiazines are chlorpromazine, prochlorperazine, and methotrimeprazine (also known as levomepromazine). The review identified one cross-sectional study that found chlorpromazine, methotrimeprazine, and prochlorperazine effectively reduce nausea and vomiting in patients with MBO [26].
Serotonin (5-HT 3 ) antagonists
Suggestion: Granisetron, serotonin (5HT 3 ) antagonist may reduce nausea and the frequency of vomiting in MBO (level of evidence: III; grade D).
Serotonin (5-HT3) antagonists act on receptors located in the chemoreceptor trigger zone to reduce nausea and vomiting. The review identified only one study published in 2009 that examined the use of a 5-HT3 antagonist, granisetron, for MBO management [32]. This study was a phase II clinical trial that found granisetron, in addition to dexamethasone and as-needed haloperidol, significantly reduced the severity of nausea (p < 0.001) and a number of episodes of vomiting (p < 0.001) before and after treatment. However, the reported incidence of constipation associated with granisetron ranges from 3 to 18% [29]. Further studies are required to assess its use in MBO.
Somatostatin analog
Recommendation: Somatostatin analog (octreotide, lanreotide) may reduce vomiting in MBO (levels of evidence: I; grade A).
MBO can cause intestinal secretions to accumulate and contribute to bowel distention, resulting in nausea and vomiting. Octreotide is a somatostatin analog that reduces intestinal and pancreas secretion and gastrointestinal motility, biliary contraction, and intestinal edema [20, 33]. Octreotide may be administered by subcutaneous bolus or continuous subcutaneous infusion. Its duration of activity is approximately 6 to 12 h, with an average half-life elimination of 1.8 h, thus necessitating multiple daily dosing schedules. Given it has a short half-life, a long-acting depot formulation is available to be administered intramuscularly once a month. Furthermore, another somatostatin analog, lanreotide, is available as a long-acting depot formulation [34, 35].
A 2016 systematic review of randomized control trials and quasi-randomized control trials, published between 1979 and 2016, identified seven studies that compared the effect of somatostatin analog with placebo and/or other pharmacologic agents (e.g., hyoscine butylbromide) on vomiting [17]. A meta-analysis was not possible given the heterogeneity in study design, outcomes, and timing of endpoints [17]. Of these seven studies, five studies with high Cochrane risk of bias found the somatostatin analog were more effective than hyoscine butylbromide [18,19,20,21] and placebo [36] for reducing vomiting. Whereas two studies with low Cochrane risk of bias found no significant difference in vomiting between somatostatin analog and placebo in their primary end points [34, 37]. A secondary analysis of a randomized control trial originally published in 2015 examined the health-related quality of life in patients with inoperable MBO and found no difference in quality-of-life scores [38].
Thienobenzodiazepene (second-generation) antipsychotic
Suggestion: Thienobenzodiazepene antipsychotic (e.g., olanzapine) may reduce nausea and vomiting in MBO (level of evidence: IV; grade: D).
Olanzapine is a second-generation thienobenzodiazepine antipsychotic that antagonizes serotonin 5-HT3 and 5-HT2c and dopamine D2 receptors, which may be responsible for its anti-emetic effects. We identified one cross-sectional study, published in 2012, that found olanzapine reduced the average nausea scores and frequency of vomiting in patients with advanced cancer and partial BO [39].
Laxatives
Suggestion: Oral osmotic laxatives should be considered in the management of impaired bowel movements in partial bowel obstruction but should be avoided in complete MBO (level of evidence: V; grade: D).
Patients with MBO present with reduced or no bowel movements. The review did not identify any studies that specifically examined the use of laxatives in this context. If patients have complete MBO, the use of laxatives is not recommended. Whereas, if patients have partial BO, the cautious use of oral osmotic laxatives (e.g., polyethylene glycol 3350, also known as macrogol) can be used. Osmotic laxatives draw water into the lumen of the bowel to soften stool and stimulate peristalsis [40]. The use of bulk-forming laxatives (e.g., psyllium) is not advised as they will increase stool consistency and potentially worsen BO [40]. If a digital rectal examination identifies a full rectum or fecal impaction, suppositories and fecal disimpaction can be considered in partial MBO [41]. However, enemas should be used with caution and are generally not recommended in MBO due to the risk of bowel perforation. Stool softeners (e.g., docusate) may be used, but their effect on bowel movement frequency is not well established.
Analgesics
Pain is experienced by 70 to 90% of patients with MBO [42]. Causes include abdominal distention, bowel spasms, and, in some cases, perforation [42]. Pain can be intermittent, cramping, or continuous in nature [4]. The WHO Guidelines for the Pharmacologic and Radiotherapeutic Management of Cancer Pain in Adults and Adolescents recommend that analgesics should be given by mouth whenever possible [43]. However, patients with MBO often have significant nausea and vomiting and malfunctioning gastrointestinal tracts that prevent ingestion and absorption of oral analgesics. Consequently, the parenteral (subcutaneous or intravenous) and/or transdermal routes of administration should be considered for this population to deliver effective analgesia. A comparison of subcutaneous and intravenous routes found no differences, confirming both routes as feasible, effective, and safe [44].
Opioids
Suggestion: Opioids are commonly used to treat pain associated with MBO, but there is no evidence to support their use (level of evidence: V; grade D).
Opioids are the mainstay analgesic for the management of moderate and severe cancer-related pain, including in the context of MBO. The review did not identify any studies that specifically examined the use of opioids for pain management in MBO. Further study is warranted, especially given that opioids impair gastrointestinal motility and can cause nausea, vomiting, and constipation [45].
Anticholinergics
Suggestion: The benefit of anticholinergics (hyoscine butylbromide) may be effective to reduce abdominal pain in MBO (level of evidence: III; grade: D).
Hyoscine (scopolamine) butylbromide is an anticholinergic agent that is widely used to treat spasmodic abdominal pain. In addition to reducing gastrointestinal secretions, it slows propulsive peristalsis and relaxes smooth muscles of the gut. Hyoscine butylbromide is commonly recommended for the management of the inoperable MBO but scarce evidence supports its use. A 2016 systematic review [17] identified two randomized clinical trials with high Cochrane risk of bias that found somatostatin analog (e.g., octreotide) were more effective than hyoscine butylbromide in reducing continuous pain [20, 21]. but one trial did not report this finding [22]. Whereas, all three trials did not find a significant difference in colicky pain between octreotide and hyscine butylbromide [20,21,22]. Two case reports reported hyoscine butylbromide in combination with other drugs (e.g., morphine, octreotide, and dexamethasone) was effective in reducing abdominal pain [22, 23].
Corticosteroids
Suggestion: The use of steroids may help with the acute symptoms of MBO and can be used for short-term benefit (level of evidence: III; grade: B).
The role of corticosteroids in MBO is complex, and the mechanism of action is not completely understood. Corticosteroids likely have an anti-inflammatory and anti-secretory effect, which may help decrease intestinal wall edema, promote salt and water absorption in MBO and, therefore, help with acute management of pain, nausea, and vomiting [46]. Dexamethasone is generally the preferred corticosteroid given its potent anti-inflammatory effect and lack of sodium-retaining properties [47]. The optimal dose of corticosteroids for MBO management is not well established. A dose between 4 and 16 mg of dexamethasone daily may be considered [34, 48]. In cases of no symptomatic improvement in 3 to 5 days, discontinuation should be considered [48].
Unselected and uncontrolled case series have reported a benefit of corticosteroids for the management of BO, yet it is challenging to attribute whether the resolution of MBO related symptoms is due to the medication or potential spontaneous resolution [30, 49, 50]. Moreover, some of these reports look at combination strategies of corticosteroids with other therapies, which makes it challenging to attribute the resolution of MBO to the corticosteroid therapy per se [34]. A retrospective cohort study retrieved information from a Japanese national medical claims database that included 3,090 adult patients with MBO treated with octreotide [51]. Octreotide alone, in combination with H2 antagonists, proton pump inhibitor, or corticosteroids, was administered in 53%, 14%, 11%, and 12% of patients, respectively. A secondary endpoint of the study was the assessment of nasogastric tube removal at 4 days. Of the 1595 patients who underwent a nasogastric tube insertion, those receiving corticosteroids with octreotide had a higher odds ratio (OR) of nasogastric tube removal within four days of insertion compared to those who did not receive corticosteroids (OR 1.16; 95% confidence interval [CI] 1.08–1.23).
A Cochrane systematic review and meta-analysis (updated search performed in 2017, with stable review) assessed the role of corticosteroids for MBO in patients with advanced gynecologic or gastrointestinal tumors [48]. Three double-blind placebo-controlled clinical trials involving 89 patients were included in the analysis. Two of the studies included in the meta-analysis were unpublished. In two of the trials, patients with a history of gastrointestinal hemorrhage, active peptic ulceration, or signs of peritonitis were excluded, and intravenous dexamethasone at 16 mg for 5 days was used [48, 52]. The third trial used methylprednisolone 40 and 240 mg intravenously for 3 days (dexamethasone equivalent 8 and 48 mg; dosage was not considered in the analysis) [48]. The primary outcome of the study was the resolution of BO within 10 days of symptom onset. There was a trend that corticosteroids may be beneficial, with a point estimate of 0.51 (95% CI 0.19–1.43). The number needed to treat with corticosteroids to resolve a BO was six. There was no effect of corticosteroids on mortality at one month. The study also concluded that the incidence of adverse events related to corticosteroids is very low; however, the use of the lowest effective dose for the shortest period should be considered to avoid long-term toxicity [48]. Concerns of long-term use include oral candidiasis, muscle proximal weakness, cushingoid habitus, gastric ulceration, infection risk, mood swings, and sleep disturbances, among others.
In a multi-center randomized trial assessing the role of octreotide in control of vomiting, dexamethasone (8 mg daily, intravenously), along with ranitidine and parenteral hydration, was considered the standardized supportive therapy for acute management of MBO [34]. There were no significant differences in the number of days free of vomiting between the octreotide and placebo groups. However, there was a significant drop in the mean number of vomiting episodes in both groups, suggesting that the standardized supportive therapy was helpful. The relative contribution of dexamethasone and/or ranitidine in the reduction of vomiting could not be addressed given the study design. No other randomized controlled trials assessing the role of corticosteroids have been detected in our review following the updated search of the Cochrane systematic review.
Oral water-soluble contrasts
No guideline possible. There are insufficient data to determine the efficacy of oral water-soluble contrasts in MBO (level of evidence: V; grade D).
Oral water-soluble contrasts are iodinated contrasts that are opaque on plain x-rays [53]. The most common form used is gastrografin, a hyperosmolar solution that is a combination of sodium diatrizoate and meglumine diatrizoate. They can be used in the management of adhesive BO as a tool to predict the resolution of adhesive small BO with conservative management, and it may also decrease the need for surgery and hospital stay [53]. A Cochrane systematic review assessed the role of oral water-soluble contrast in MBO [53] and identified only one randomized double-blinded placebo-controlled feasibility study with a high risk of bias that enrolled nine patients [54]. The study assessed the role of gastrografin compared to placebo in patients with MBO and no indication for surgery or endoscopic interventions. The rate of resolution of MBO or ability to predict resolution of MBO was not reported. No safety signals were detected. A retrospective study assessed the role of the gastrografin in the management of BO [55]. The administration of gastrografin challenge was considered safe. In the subgroup analysis of patients with active malignancy (n = 63), there were no differences in the need for surgical exploration or length of hospital stay between those who did and did not receive gastrografin. No other reports have been detected in our search following the publication of the Cochrane review.
Bowel decompression
Nasogastric tube and percutaneous gastrostomy insertion, either endoscopically or radiologically guided, are established techniques to provide enteral feeding. In the case of MBO, these measures are used as a venting/decompression procedure. The following sections provide suggestions/recommendations for all bowel decompression interventions and their associated existing evidence:
Nasogastric tube
Suggestion: Nasogastric tube may be used for temporary decompression in acute MBO (level of evidence: V; grade: D).
Temporary decompression via a wide bore nasogastric tube can evacuate a large amount of pooled gastric secretions, particularly during acute MBO episodes, and may improve symptoms [56]. Based on clinical expertise and existing guidelines, it is not suggested for chronic MBO management because the nasogastric tubes are not well tolerated when placed for a prolonged time [57, 58]. Nasogastric tubes can become occluded or displaced, which may require flushing or replacement. Other potential complications include nasal cartilage erosion, otitis media, aspiration pneumonia, esophagitis, and bleeding [59].
Endoscopic or percutaneous gastrostomy tube
Suggestion: Endoscopic or percutaneous gastrostomy tube may be used for gastric decompression in MBO (level of evidence: IV; grade B evidence).
A more permanent decompression of gastric contents can be achieved through the placement of an endoscopic or percutaneous gastrostomy tube, also known as “venting gastrostomy”. This review identified 29 studies that suggest gastrostomy insertion is generally feasible [60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87], with a high reduction in symptoms of nausea and vomiting. Major complications were rare, with most complications classified as minor wound infections or leakage of fluid around the tube. The presence of ascites is not an absolute contraindication to the insertion of percutaneous venting gastrostomy in patients with MBO [61, 68, 78]; however, it is reasonable to suggest that ascitic drainage with paracentesis or placement of an intraperitoneal catheter is performed to reduce potential complications.
Percutaneous transesophageal gastro-tubing
Suggestion: Percutaneous transesophageal gastro-tubing may be used for gastric decompression in MBO (level of evidence: IV; grade: C).
The placement of percutaneous transesophageal gastro-tubing (PTEG) may be used to decompress gastric contents. This review identified four studies with a limited number of patients with MBO that are not candidates for surgical decompression or percutaneous/endoscopic gastrostomy tubes [88,89,90,91]. A Japanese randomized controlled trial assessed PTEG or nasogastric (NG) tube in 40 patients with inoperable MBO. The PTEG resulted in fewer symptoms and higher quality of life compared with NG tube [88]. The primary endpoint was symptom palliation for two weeks (non-validated questionnaire), and showed lower symptom burden in those patients with PTEG. Secondary endpoints included quality of life measures (EQ-5D and SF-8 scores), which were significantly higher in the PTEG group. No differences in overall survival were detected between the two groups.
Palliative surgery and stent
Recommendation and suggestions
-
Self-expanding metallic stents are the preferred alternative for the management of single-level large bowel obstruction when technically feasible and in the absence of colonic perforation (level of evidence: II; grade: B). Surgical palliation may be considered an alternative in selected cases.
-
In the case of a multi-level obstruction, palliative surgical intervention may be considered in a highly selected population (level of evidence: IV; grade: B).
-
Patients with advanced cancer that undergo palliative surgery for MBO are at high risk of surgical complications, and less invasive surgical interventions should be considered (level of evidence: IV; grade: B).
Interventions in single-level obstruction may include decompression methods or open surgery with bypass or stoma [92]. Obtaining a surgical opinion should be considered at the time of MBO diagnosis. Stoma formation is preferred if the length of the proximal intestine ensures gastrointestinal autonomy (low risk of short bowel syndrome) or if the risk of complications related to intra-abdominal anastomosis is too high [93]. The large bowel decompression options may include colonic decompression tubes, ablative methods, and the use of self-expanding metal stents (SEMS) [94]. The SEMS are generally the preferred decompression method as they have shown higher success rates than decompression tubes [95, 96]. Decompression methods are not indicated in patients with a bowel perforation [94].
Most studies assessing the role of SEMS in MBO have included mixed populations (curative intent with the stent placement as a bridge-to-surgery and palliative) [97,98,99,100,101,102]. The role of the stent as bridge-to-surgery is out of the scope of this guideline. In a meta-analysis of patients with large BO caused by colorectal cancer undergoing SEMS placement or emergency surgery with palliative intent, eighteen studies (randomized controlled trials and comparative observational studies) with 1518 patients were included. Results showed that 30-day mortality was higher in those patients undergoing a surgical procedure (OR 0.4; 95% CI 0.28–0.69) [103]. Early complications were more frequent in the surgical group, whereas late complications occurred more frequently after SEMS, mainly re-obstructions. Another meta-analysis in the same setting only including randomized controlled trials (four studies, 125 patients), showed no differences in 30-day mortality or mean survival between those patients undergoing SEMS or emergency surgery, with a shorter hospital stay in the SEMS group [104]. Another randomized controlled trial assessed the role of stent insertion or surgical decompression for non-curable large BO [105]. Results showed that the combined costs were lower in those patients treated with a stent. In terms of quality of life, measured by European quality of life five dimension (EQ-5D) scores, no differences were detected at 4-weeks between the two groups.
Most of the studies assessing the role of palliative surgery for multi-focal or small bowel MBO are retrospective in nature. A Cochrane systematic review of studies that assessed the role of surgery for MBO in advanced gynecologic or colorectal cancer identified 43 studies with a total of 4265 participants [2]. Most studies were retrospective, of low methodological quality and high risk of bias. Additionally, some studies included mixed populations with the benign and malignant causes of BO. Re-obstruction rates post-surgical laparotomy, when reported, ranged from 10 to 63%, with limited time to re-obstruction data. The ability to feed orally ranged between 30 and 100%. When reported, postoperative mortality within 30 days was 0 to 32%, and postoperative morbidity ranged from 22 to 87%. It was not possible to conclude whether there was the benefit of using surgery in this setting. Following the publication of the Cochrane review, no randomized trials have been detected in our review. However, other systematic reviews have focused on studies assessing the role of surgery in specific populations with MBO, including patients with small BO or peritoneal carcinomatosis [93, 106]. Similarly, it was not possible to conclude if there is a benefit on surgery in this setting, as most studies were retrospective, of low methodological quality, and high selection bias in those deemed appropriate for surgical intervention. It may be reasonable to select patients that are more likely to benefit from the procedure, including good performance status, and to focus the surgical intervention on relieving symptoms.
Nutrition
Oral intake is significantly impaired in MBO and raises important questions about hydration and nutrition, which should be carefully considered and discussed with the patient and their substitute decision-makers.
Diet
Suggestions:
-
When a patient is initially diagnosed with MBO, they should be made Nil Per Os (NPO; nothing by mouth), and then when the acute MBO resolves fully or partially, a symptom led, slow and graded reintroduction to oral diet is recommended. This may include clear fluids, free or full fluids, texture modified low fiber diet (soft, minced, and pureed), and if tolerated, back to normal textured low fiber diet (level of evidence: IV; grade: B).
-
Nutrition interventions should be initiated in patients with advanced cancers only where the benefits of these interventions on quality of life and survival outweigh the risks, with clear expectations discussed by a multidisciplinary team with patients and families (level of evidence: IV; grade: B).
Patients admitted with acute onset of an MBO are made nil by mouth with the addition of a decompressive procedures to control symptoms such as nausea and vomiting [2, 107, 108]. Managing the nutrition requirements of patients presenting with advanced cancers and MBO is controversial and ethically challenging as there is no consensus due to scant published evidence [5, 109, 110]. Nutrition interventions must be considered in the context of the patient’s prognosis with the focus on prioritising the patient’s wishes [111]. Awareness of the sociocultural meaning of food and nutrition for many people is an important consideration and may contribute to increases in quality of life [111, 112].
As a patient’s acute MBO resolves, either fully or partially, a symptom led, slow, and a graded reintroduction to oral diet is recommended. This may include clear fluids, free or full fluids, texture modified low fiber diet (soft, minced, and pureed) and back to normal textured low fiber diet. A low fiber diet is one that contains a maximum of 10 g of fiber per day. A low fiber diet is thought to be beneficial due to a reduction in stool bulk which may lead to reduced pain, abdominal cramps, gas or feeling of fullness, particularly in those people with ongoing subacute BO [110]. General recommendations also include a grazing eating pattern, with small volumes of food and fluid consumed at any one time. Nutrition education to assist the patient and family to modify their diet according to symptoms is recommended to enhance autonomy and self-management.
Hydration
Suggestions
-
Parenteral hydration does not prevent or improve symptoms, such as thirst or dry mouth, nor does it increase survival, and in excessive amounts, it may bring on fluid overload, peripheral and pulmonary edema (level of evidence: III; grade: B).
-
Parenteral hydration should not be initiated routinely in the last days of life (level of evidence: III; grade: B).
Hydration is an element of parenteral nutrition,42 yet hydration may be delivered without nutrition, and it should be considered separately as a means of palliative care to relieve symptoms, not prolonging life [113]. Parenteral hydration (PH) means providing fluids infusion by the route other than oral or enteral (i.e., intravenously or subcutaneously; hypodermoclysis) [111]. Hypodermoclysis is an effective and safe route of hydrating a patient up to 1500 mL/day, with few local adverse effects [114]; with attention to water and electrolytes balance. PH is an element of palliative care and should follow predefined realistic treatment goals.
In a small prospective randomized trial of patients with inoperable MBO, the amount of parenterally supplied fluids was not associated with thirst, dry mouth intensity, or abdominal distention, but a volume of >500 mL/day might reduce nausea and drowsiness [20]. However, a high level of PH may result in more gastrointestinal secretions [3].
A Cochrane review found an insufficient number of low risk of bias studies, which does not allow the formulation of any clinical practice recommendations about medically assisted PH in palliative care patients [115]. All the studies included in the Cochrane review only had participants with advanced cancer, but it was not reported whether they had MBO. Our review did not detect specific studies assessing PH at the end of life, specifically in patients with MBO. At the end of life, PH may not be beneficial [116]. In a systematic review of practices in the last week of the life of patients with cancer, PH did not improve symptoms such as thirst and delirium, although data to support or discourage it is scarce [117]. Dehydration is a frequent cause of delirium, and PH may appear effective, except in the dying phase when even moderate amounts of artificial hydration may be harmful. PH in the dying phase may increase the risk of fluid overload, peripheral swelling, ascites, and pulmonary edema [116]. In a multi-center, double-blind placebo-controlled randomized study, subcutaneous infusion of 1000 mL daily did not improve symptoms of dehydration (fatigue, myoclonus, sedation, and hallucinations), quality of life, or survival at the end of life [118].
Parenteral nutrition
Suggestions:
-
HPN may be beneficial and maintain the quality of life in a very selected group of patients with MBO (level of evidence: IV; grade: D).
-
Central venous access is preferred for HPN delivery (level III, grade: B).
-
In end-of-life HPN, should be discontinued (or not initiated) as it raises the risk of complications and may prolong suffering (level of evidence: V; grade: D).
The following terms are used to describe parenteral nutrition: “total parenteral nutrition” (TPN): nutrition administered exclusively intravenously; “supplemental parenteral nutrition (SPN)”: nutrition administered intravenously and enterally, regardless of proportion, orally or by artificial access; and “home parenteral nutrition” (HPN): parenteral nutrition (regardless TPN or SPN) administered at home.
Feeding through the peripheral veins may cause phlebitis and the need for the reintroduction of vein access. It may cause patient suffering and require additional medical procedures. Feeding through the central veins (e.g., Broviac catheter, vascular access port, peripherally inserted central catheter) is advised, as it ensures long-term safe access upon the strict aseptic protocol is followed [119, 120].
Most studies assessing the role of the parenteral nutrition are retrospective. A Cochrane systematic review assessed the effectiveness of HPN in survival and quality of life in patients with inoperable MBO [121]. Thirteen studies (all observational with a high risk of bias), including 721 participants were included. The median survival intervals were variable, between 15 to 155 days [121]. Only three studies reported validated quality of life measures, showing equivocal results (one study reported improvements during the first three months and two studies showed an equal number of patients with improved and deteriorated quality of life) [122,123,124].
HPN can be associated with the risk of complications (metabolic and/or catheter-related). In the Cochrane systematic review, adverse events were measured in nine studies, showing that between 6 and 21% of patients developed a central venous catheter infection or were hospitalized because of complications related to parenteral nutrition [121]. The use of HPN also involves financial, personnel, and infrastructure resources. A meta-analysis included a health economic evaluation of HPN in this setting [125].
There is a need to weigh the benefits and risks when recommending HPN, including the predicted cancer-related survival [125,126,127]. Potential prognostic criteria for survival and benefit of HPN may include (i) histopathological type of the tumor—slow-growing and chemo-sensitive cancer [128,129,130,131], (ii) performance status—ECOG <2, (iii) no fluid retention (peripheral edema, pleural or peritoneal effusion), (iv) no anemia, and (v) no hypoalbuminemia [132]. This evaluation should be reassessed over time.
Most patients may not benefit from HPN due to clinical deterioration resulting from cancer progression, chronic cancer treatment [133], latent infections [134], depression (up to 24%) [135, 136], and suboptimal nutritional treatment [137]. Malnutrition and cachexia in patients with advanced cancer and MBO are usually irreversible and treatment-resistant [138]. Therefore, the goal of the treatment is to maintain the nutritional status and achieve functional (through physiotherapy and nutrition), symptomatic, associative and mental function improvement.
The European Society for Clinical Nutrition and Metabolism (ESPEN) practical guideline for clinical nutrition in patients with cancer suggests that in dying patients, treatment recommendations should be based on comfort [109]. Parenteral hydration and nutrition are unlikely to provide any benefit for most patients at the end of life [109].
Psychosocial support
The course of MBO can be unpredictable with a range of complex and challenging symptoms, which can be difficult to manage for both the patient and their family. The importance of family and supportive care is firmly established within palliative and end-of-life care literature [139, 140]. Although the need for emotional and psychological support for both patient and family in the context of MBO is equally acknowledged, high-quality studies which focus specifically on the context of MBO are limited [141, 142]. Evidence has been based on clinical case reviews, small scale qualitative descriptive studies or current best practice opinions.
The unmet psychosocial need has been identified as a significant issue in patients with ovarian cancer and MBO, as it is often poorly addressed. The need for a multidisciplinary approach to care is promoted [143]. A qualitative study described the benefits of a model of supported self-management for women with advanced gynecological cancers and MBO attending out-patient clinics. Clear communication, counselling and referral to early palliative care were identified as important considerations [144]. A separate qualitative study with oncologists treating gynecological cancers identified that early palliative care referral was beneficial, particularly in situations where patients were not candidates for surgery [145]. However, patients’ expectations of their oncologists were not always met, suggesting strengthening of communication strategies and protocols is needed.
Given the nature and range of complex symptoms associated with MBO, early palliative care has been suggested. Optimal treatment requires a realistic assessment of goals of care with important communication about prognosis, management of symptoms, and end-of-life care. A recent review of the surgical management of MBO suggests careful decision-making with patient and family to ensure the most appropriate outcome [146]. A 2018 systematic review focused on the burden of care placed on family caregivers as a result of home-based parenteral nutrition for women with advanced ovarian cancer and MBO found that caregivers described an experience of vulnerability and family disruption caused by the decision to care for the patient at home. Conversely, in acknowledging the challenges placed on the family, patients recognized the lifeline given to them in terms of spending quality time with their family by this option for home care [121].
Conclusion
MBO is a significant complication for patients living with cancer. The approach should be multi and inter-disciplinary to improve the management of these patients and support their families. Studies indicate the need to improve the quality of research and subsequent interventions in this domain (refer to Table 3 for a summary of suggestions and recommendations). Communication around changing goals of care is essential to foster clear and decisive clinical decision making in partnership with patients and families. Further prospective and innovative studies are needed to improve the care for patients with MBO.
Data Availability
The review search strategy and data are available upon reasonable request.
References
Anthony T, Baron T, Mercadante S et al (2007) Report of the clinical protocol committee: development of randomized trials for malignant bowel obstruction. J Pain Symptom Manage 34:S49-59
Cousins SE, Tempest E, Feuer DJ (2016) Surgery for the resolution of symptoms in malignant bowel obstruction in advanced gynaecological and gastrointestinal cancer. Cochrane Database Syst Rev 2016(1):CD002764. https://doi.org/10.1002/14651858.CD002764.pub2
Ripamonti CI, Easson AM, Gerdes H (2008) Management of malignant bowel obstruction. Eur J Cancer 44:1105–15
Lee YC, Jivraj N, O’Brien C et al (2018) Malignant bowel obstruction in advanced gynecologic cancers: an updated review from a multidisciplinary perspective. Obstet Gynecol Int 2018:1867238
National Audit of Small Bowel Obstruction Steering Group & National Audit of Small Bowel Obstruction Collaborators (2019) Outcomes following small bowel obstruction due to malignancy in the national audit of small bowel obstruction. Eur J Surg Oncol 45(12):2319–2324. https://doi.org/10.1016/j.ejso.2019.07.014
Patel PS, Fragkos KC, Keane N et al (2021) Clinical and nutritional care pathways of patients with malignant bowel obstruction: a retrospective analysis in a tertiary UK center. Nutr Cancer 73:572–587
Yim GW, Eoh KJ, Kim SW et al (2016) Malnutrition identified by the nutritional risk index and poor prognosis in advanced epithelial ovarian carcinoma. Nutr Cancer 68:772–9
Marshall KM, Loeliger J, Nolte L et al (2019) Prevalence of malnutrition and impact on clinical outcomes in cancer services: a comparison of two time points. Clin Nutr 38:644–651
Chang KJ, Marin D, Kim DH et al (2020) ACR appropriateness criteria® Suspected small-bowel obstruction. J Am Coll Radiol 17:S305-s314
Puchalski CM, Sbrana A, Ferrell B, et al. (2019) Interprofessional spiritual care in oncology: a literature review. ESMO Open 4(1):e000465. https://doi.org/10.1136/esmoopen-2018-000465
Crawford GB, Dzierżanowski T, Hauser K, et al. (2021) Care of the adult cancer patient at the end of life: ESMO clinical practice guidelines. ESMO Open 6(4):100225. https://doi.org/10.1016/j.esmoop.2021.100225
Hui D, Hess K, dos Santos R et al (2015) A diagnostic model for impending death in cancer patients: preliminary report. Cancer 121:3914–21
Dolan RD, McSorley ST, Horgan PG et al (2017) The role of the systemic inflammatory response in predicting outcomes in patients with advanced inoperable cancer: systematic review and meta-analysis. Crit Rev Oncol Hematol 116:134–146
Simmons CPL, McMillan DC, McWilliams K et al (2017) Prognostic tools in patients with advanced cancer: a systematic review. J Pain Symptom Manage 53:962-970.e10
Gilligan T, Coyle N, Frankel RM et al (2017) Patient-clinician communication: American Society of Clinical Oncology consensus guideline. J Clin Oncol 35:3618–3632
Davis M, Hui D, Davies A, et al. (2021) Medical management of malignant bowel obstruction in patients with advanced cancer: 2021 MASCC guideline update. Support Care Cancer 29(12):8089–8096. https://doi.org/10.1007/s00520-021-06438-9
Obita GP, Boland EG, Currow DC et al (2016) Somatostatin analogues compared with placebo and other pharmacologic agents in the management of symptoms of inoperable malignant bowel obstruction: a systematic review. J Pain Symptom Manage 52:901-919.e1
Mercadante S, Ripamonti C, Casuccio A et al (2000) Comparison of octreotide and hyoscine butylbromide in controlling gastrointestinal symptoms due to malignant inoperable bowel obstruction. Support Care Cancer 8:188–91
Peng X, Wang P, Li S, et al. (2015) Randomized clinical trial comparing octreotide and scopolamine butylbromide in symptom control of patients with inoperable bowel obstruction due to advanced ovarian cancer. World J Surg Oncol 13:50. https://doi.org/10.1186/s12957-015-0455-3
Ripamonti C, Mercadante S, Groff L et al (2000) Role of octreotide, scopolamine butylbromide, and hydration in symptom control of patients with inoperable bowel obstruction and nasogastric tubes: a prospective randomized trial. J Pain Symptom Manage 19:23–34
Mystakidou K, Tsilika E, Kalaidopoulou O et al (2002) Comparison of octreotide administration vs conservative treatment in the management of inoperable bowel obstruction in patients with far advanced cancer: a randomized, double- blind, controlled clinical trial. Anticancer Res 22:1187–92
De Conno F, Caraceni A, Zecca E et al (1991) Continuous subcutaneous infusion of hyoscine butylbromide reduces secretions in patients with gastrointestinal obstruction. J Pain Symptom Manage 6:484–6
Weber C, Zulian GB (2009) Malignant irreversible intestinal obstruction: the powerful association of octreotide to corticosteroids, antiemetics, and analgesics. Am J Hosp Palliat Care 26:84–8
Mercadante S (1998) Scopolamine butylbromide plus octreotide in unresponsive bowel obstruction. J Pain Symptom Manage 16:278–80
Davis MP, Furste A (1999) Glycopyrrolate: a useful drug in the palliation of mechanical bowel obstruction. J Pain Symptom Manage 18:153–4
Baines M, Oliver DJ, Carter RL (1985) Medical management of intestinal obstruction in patients with advanced malignant disease. A clinical and pathological study. Lancet 2:990–3
Fainsinger RL, Spachynski K, Hanson J et al (1994) Symptom control in terminally ill patients with malignant bowel obstruction (MBO). J Pain Symptom Manage 9:12–8
Muir JC, von Gunten CF (2001) Abdominal cancer, nausea, and vomiting. J Palliat Med 4:391–4
Lexi-drugs online [database on the Internet]. Hudson (OH): Lexicomp, Inc.; 2021 [updated 23 Sept 2021; cited 26 Sept 2021]. Available from: http://online.lexi.com. Subscription required to view. Accessed 1 July 2021
Berger J, Lester P, Rodrigues L (2016) Medical therapy of malignant bowel obstruction with octreotide, dexamethasone, and metoclopramide. Am J Hosp Palliat Care 33:407–10
Mercadante S, Ferrera P, Villari P et al (2004) Aggressive pharmacological treatment for reversing malignant bowel obstruction. J Pain Symptom Manage 28:412–6
Tuca A, Roca R, Sala C et al (2009) Efficacy of granisetron in the antiemetic control of nonsurgical intestinal obstruction in advanced cancer: a phase II clinical trial. J Pain Symptom Manage 37:259–70
Clare C, Bennett M (2002) Tolerance issues in the use of somatostatin analogs in palliative care [1]. J Pain Symptom Manag 23:87–88
Currow DC, Quinn S, Agar M et al (2015) Double-blind, placebo-controlled, randomized trial of octreotide in malignant bowel obstruction. J Pain Symptom Manage 49:814–21
Matulonis UA, Seiden MV, Roche M et al (2005) Long-acting octreotide for the treatment and symptomatic relief of bowel obstruction in advanced ovarian cancer. J Pain Symptom Manage 30:563–9
Laval G, Rousselot H, Toussaint-Martel S et al (2012) SALTO: a randomized, multicenter study assessing octreotide LAR in inoperable bowel obstruction. Bull Cancer 99:E1–E9
Mariani P, Blumberg J, Landau A et al (2012) Symptomatic treatment with lanreotide microparticles in inoperable bowel obstruction resulting from peritoneal carcinomatosis: a randomized, double-blind, placebo-controlled phase III study. J Clin Oncol 30:4337–4343
McCaffrey N, Asser T, Fazekas B et al (2020) Health-related quality of life in patients with inoperable malignant bowel obstruction: secondary outcome from a double-blind, parallel, placebo-controlled randomised trial of octreotide. BMC Cancer 20:1050
Kaneishi K, Kawabata M, Morita T (2012) Olanzapine for the relief of nausea in patients with advanced cancer and incomplete bowel obstruction. J Pain Symptom Manage 44:604–7
Bashir A, Sizar O. Laxatives. [Updated 2021 Sep 4]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 Jan. Available from: https://www.ncbi.nlm.nih.gov/books/NBK537246/ Accessed on: 20th July, 2021.
Larkin PJ, Cherny NI, La Carpia D, et al. (2018) Diagnosis, assessment and management of constipation in advanced cancer: ESMO clinical practice guidelines. Ann Oncol 29:iv111–iv125
Tuca A, Guell E, Martinez-Losada E et al (2012) Malignant bowel obstruction in advanced cancer patients: epidemiology, management, and factors influencing spontaneous resolution. Cancer Manag Res 4:159–69
WHO (2019) Guidelines for the pharmacological and radiotherapeutic management of cancer pain in adults and adolescents. https://www.who.int/publications/i/item/9789241550390 Accessed on: 1st June 2021.
Radbruch L, Trottenberg P, Elsner F et al (2011) Systematic review of the role of alternative application routes for opioid treatment for moderate to severe cancer pain: an EPCRC opioid guidelines project. Palliat Med 25:578–96
Kraichely RE, Arora AS, Murray JA (2010) Opiate-induced oesophageal dysmotility. Aliment Pharmacol Ther 31:601–6
Hsu K, Prommer E, Murphy MC et al (2019) Pharmacologic management of malignant bowel obstruction: when surgery is not an option. J Hosp Med 14:367–373
TABLETS DECADRON® (DEXAMETHASONE TABLETS, USP). U.S. Food and Drug Administration website. https://www.accessdata.fda.gov/drugsatfda_docs/label/2004/11664slr062_decadron_lbl.pdf Accessed on 15th August 2021.
Feuer DJ, Broadley KE (2000) Corticosteroids for the resolution of malignant bowel obstruction in advanced gynaecological and gastrointestinal cancer. Cochrane Database Syst Rev 2000:Cd001219
Laval G, Arvieux C, Stefani L et al (2006) Protocol for the treatment of malignant inoperable bowel obstruction: a prospective study of 80 cases at Grenoble University Hospital Center. J Pain Symptom Manage 31:502–12
Philip J, Lickiss N, Grant PT et al (1999) Corticosteroids in the management of bowel obstruction on a gynecological oncology unit. Gynecol Oncol 74:68–73
Minoura T, Takeuchi M, Morita T et al (2018) Practice patterns of medications for patients with malignant bowel obstruction using a nationwide claims database and the association between treatment outcomes and concomitant use of h<sub>2</sub>-blockers/proton pump inhibitors and corticosteroids with. J Pain Symptom Manage 55:413-419.e2
Hardy J, Ling J, Mansi J et al (1998) Pitfalls in placebo-controlled trials in palliative care: dexamethasone for the palliation of malignant bowel obstruction. Palliat Med 12:437–42
Syrmis W, Richard R, Jenkins-Marsh S, et al. (2018) Oral water soluble contrast for malignant bowel obstruction. Cochrane Database Syst Rev 3(3):CD012014. https://doi.org/10.1002/14651858.CD012014.pub2
Lee C, Vather R, O’Callaghan A et al (2013) Validation of the phase II feasibility study in a palliative care setting: gastrografin in malignant bowel obstruction. Am J Hosp Palliat Care 30:752–8
Khasawneh MA, Eiken PW, Srvantstyan B, et al. (2013) Use of the Gastrografin challenge in patients with a history of abdominal or pelvic malignancy. Surgery 154:769–75 discussion 775
Dolan EA (2011) Malignant bowel obstruction: a review of current treatment strategies. Am J Hosp Palliat Care 28:576–82
Laval G, Marcelin-Benazech B, Guirimand F et al (2014) Recommendations for bowel obstruction with peritoneal carcinomatosis. J Pain Symptom Manag 48:75–91
Ripamonti C, Twycross R, Baines M et al (2001) Clinical-practice recommendations for the management of bowel obstruction in patients with end-stage cancer. Support Care Cancer 9:223–33
Prabhakaran S, Doraiswamy VA, Nagaraja V et al (2012) Nasoenteric tube complications. Scand J Surg 101:147–55
Malone JM Jr, Koonce T, Larson DM et al (1986) Palliation of small bowel obstruction by percutaneous gastrostomy in patients with progressive ovarian carcinoma. Obstet Gynecol 68:431–3
Lee MJ, Saini S, Brink JA et al (1991) Malignant small bowel obstruction and ascites: not a contraindication to percutaneous gastrostomy. Clin Radiol 44:332–4
Herman LL, Hoskins WJ, Shike M (1992) Percutaneous endoscopic gastrostomy for decompression of the stomach and small bowel. Gastrointest Endosc 38:314–8
Adelson MD, Kasowitz MH (1993) Percutaneous endoscopic drainage gastrostomy in the treatment of gastrointestinal obstruction from intraperitoneal malignancy. Obstet Gynecol 81:467–71
Marks WH, Perkal MF, Schwartz PE (1993) Percutaneous endoscopic gastrostomy for gastric decompression in metastatic gynecologic malignancies. Surg Gynecol Obstet 177:573–6
Cannizzaro R, Bortoluzzi F, Valentini M et al (1995) Percutaneous endoscopic gastrostomy as a decompressive technique in bowel obstruction due to abdominal carcinomatosis. Endoscopy 27:317–20
Cunningham MJ, Bromberg C, Kredentser DC et al (1995) Percutaneous gastrostomy for decompression in patients with advanced gynecologic malignancies. Gynecol Oncol 59:273–6
Campagnutta E, Cannizzaro R, Gallo A et al (1996) Palliative treatment of upper intestinal obstruction by gynecological malignancy: the usefulness of percutaneous endoscopic gastrostomy. Gynecol Oncol 62:103–105
Ryan JM, Hahn PF, Mueller PR (1998) Performing radiologic gastrostomy or gastrojejunostomy in patients with malignant ascites. AJR Am J Roentgenol 171:1003–6
Scheidbach H, Horbach T, Groitl H, et al. (1999) Percutaneous endoscopic gastrostomy/jejunostomy (PEG/PEJ) for decompression in the upper gastrointestinal tract. Initial experience with palliative treatment of gastrointestinal obstruction in terminally ill patients with advanced carcinomas. Surg Endosc 13:1103–1105
Brooksbank MA, Game PA, Ashby MA (2002) Palliative venting gastrostomy in malignant intestinal obstruction. Palliat Med 16:520–6
Jolicoeur L, Faught W (2003) Managing bowel obstruction in ovarian cancer using a percutaneous endoscopic gastrostomy (PEG) tube. Can Oncol Nurs J 13:212–9
Piccinni G, Angrisano A, Testini M et al (2005) Venting direct percutaneous jejunostomy (DPEJ) for drainage of malignant bowel obstruction in patients operated on for gastric cancer. Support Care Cancer 13:535–9
Pothuri B, Montemarano M, Gerardi M et al (2005) Percutaneous endoscopic gastrostomy tube placement in patients with malignant bowel obstruction due to ovarian carcinoma. Gynecol Oncol 96:330–4
Chakraborty A, Selby D, Gardiner K et al (2011) Malignant bowel obstruction: natural history of a heterogeneous patient population followed prospectively over two years. J Pain Symptom Manage 41:412–20
Vashi PG, Dahlk S, Vashi RP et al (2011) Percutaneous endoscopic gastrostomy tube occlusion in malignant peritoneal carcinomatosis-induced bowel obstruction. Eur J Gastroenterol Hepatol 23:1069–73
Rath KS, Loseth D, Muscarella P et al (2013) Outcomes following percutaneous upper gastrointestinal decompressive tube placement for malignant bowel obstruction in ovarian cancer. Gynecol Oncol 129:103–6
Teriaky A, Gregor J, Chande N (2012) Percutaneous endoscopic gastrostomy tube placement for end-stage palliation of malignant gastrointestinal obstructions. Saudi J Gastroenterol. 18(2):95–8. https://doi.org/10.4103/1319-3767.93808
Shaw C, Bassett RL, Fox PS et al (2013) Palliative venting gastrostomy in patients with malignant bowel obstruction and ascites. Ann Surg Oncol 20:497–505
Issaka RB, Shapiro DM, Parikh ND et al (2014) Palliative venting percutaneous endoscopic gastrostomy tube is safe and effective in patients with malignant obstruction. Surg Endosc 28:1668–73
Kawata N, Kakushima N, Tanaka M et al (2014) Percutaneous endoscopic gastrostomy for decompression of malignant bowel obstruction. Dig 26:208–13
DeEulis TG, Yennurajalingam S (2015) Venting gastrostomy at home for symptomatic management of bowel obstruction in advanced/recurrent ovarian malignancy: a case series. J Palliat Med 18:722–8
Zucchi E, Fornasarig M, Martella L et al (2016) Decompressive percutaneous endoscopic gastrostomy in advanced cancer patients with small-bowel obstruction is feasible and effective: a large prospective study. Support Care Cancer 24:2877–82
Dittrich A, Schubert B, Kramer M et al (2017) Benefits and risks of a percutaneous endoscopic gastrostomy (PEG) for decompression in patients with malignant gastrointestinal obstruction. Support Care Cancer 25:2849–2856
Singh Curry R, Evans E, Raftery AM et al (2019) Percutaneous venting gastrostomy/gastrojejunostomy for malignant bowel obstruction: a qualitative study. BMJ Support 9:381–388
Pinard KA, Goring TN, Egan BC et al (2017) Drainage percutaneous endoscopic gastrostomy for malignant bowel obstruction in gastrointestinal cancers: prognosis and implications for timing of palliative intervention. J Palliat Med 20:774–778
Kurita Y, Koide T, Watanabe S et al (2013) Postpyloric decompression tube placement through a gastrostomy for malignant bowel obstruction. BMC Res Notes 6:217
Gauvin G, Do-Nguyen CC, Lou J, et al. (2021) Gastrostomy tube for nutrition and malignant bowel obstruction in patients with cancer. J Natl Compr Canc Netw 19(1):48–56. https://doi.org/10.6004/jnccn.2020.7604
Aramaki T, Arai Y, Takeuchi Y et al (2020) A randomized, controlled trial of the efficacy of percutaneous transesophageal gastro-tubing (PTEG) as palliative care for patients with malignant bowel obstruction: the JIVROSG0805 trial. Support Care Cancer 28:2563–2569
Aramaki T, Arai Y, Inaba Y et al (2013) Phase II study of percutaneous transesophageal gastrotubing for patients with malignant gastrointestinal obstruction; JIVROSG-0205. J Vasc Interv Radiol 24:1011–7
Selby D, Nolen A, Sittambalam C, et al. (2019) Percutaneous transesophageal gastrostomy (PTEG): a safe and well-tolerated procedure for palliation of end-stage malignant bowel obstruction. J Pain Symptom Manage 58:306–310
Mackey R, Chand B, Oishi H et al (2005) Percutaneous transesophageal gastrostomy tube for decompression of malignant obstruction: report of the first case and our series in the US. J Am Coll Surg 201:695–700
Ferguson HJ, Ferguson CI, Speakman J et al (2015) Management of intestinal obstruction in advanced malignancy. Ann Med Surg (Lond) 4:264–70
Paul Olson TJ, Pinkerton C, Brasel KJ et al (2014) Palliative surgery for malignant bowel obstruction from carcinomatosis: a systematic review. JAMA Surg 149:383–92
Adler DG (2005) Management of malignant colonic obstruction. Curr Treat Options Gastroenterol 8:231–237
Van Hooft JE, Van Halsema EE, Vanbiervliet G et al (2014) Self-expandable metal stents for obstructing colonic and extracolonic cancer: European society of gastrointestinal endoscopy (ESGE) Clinical Guideline. Gastrointest Endosc 80:747-761.e7
Wang FG, Bai RX, Yan M, et al. (2020) Short-term outcomes of self-expandable metallic stent versus decompression tube for malignant colorectal obstruction: a meta-analysis of clinical data. J Investig Surg 33:762–770
Atukorale YN, Church JL, Hoggan BL et al (2016) Self-expanding metallic stents for the management of emergency malignant large bowel obstruction: a systematic review. J Gastrointest Surg 20:455–62
Cennamo V, Luigiano C, Coccolini F et al (2013) Meta-analysis of randomized trials comparing endoscopic stenting and surgical decompression for colorectal cancer obstruction. Int J Color Dis 28:855–863
Jain SR, Yaow CYL, Ng CH et al (2020) Comparison of colonic stents, stomas and resection for obstructive left colon cancer: a meta-analysis. Tech Coloproctol 24:1121–1136
Khot UP, Lang AW, Murali K et al (2002) Systematic review of the efficacy and safety of colorectal stents. Br J Surg 89:1096–102
Neo VSQ, Jain SR, Yeo JW et al (2021) Controversies of colonic stenting in obstructive left colorectal cancer: a critical analysis with meta-analysis and meta-regression. Int J Colorectal Dis 36:689–700
Watt AM, Faragher IG, Griffin TT et al (2007) Self-expanding metallic stents for relieving malignant colorectal obstruction: a systematic review. Ann Surg 246:24–30
Veld J, Umans D, van Halsema E, et al. (2020) Self-expandable metal stent (SEMS) placement or emergency surgery as palliative treatment for obstructive colorectal cancer: a systematic review and meta-analysis. Crit Rev Oncol Hematol 155:103110. https://doi.org/10.1016/j.critrevonc.2020.103110
Ribeiro IB, Bernardo WM, Martins BDC et al (2018) Colonic stent versus emergency surgery as treatment of malignant colonic obstruction in the palliative setting: a systematic review and meta-analysis. Endosc Int Open 6:E558–E567
Young CJ, Zahid A (2018) Randomized controlled trial of colonic stent insertion in non-curable large bowel obstruction: a post hoc cost analysis. Colorectal Dis 20:288–295
Banting SP, Waters PS, Peacock O et al (2021) Management of primary and metastatic malignant small bowel obstruction, operate or palliate. A systematic review. ANZ J Surg 91:282–290
Gemlo B, Rayner AA, Lewis B et al (1986) Home support of patients with end-stage malignant bowel obstruction using hydration and venting gastrostomy. Am J Surg 152:100–4
Bozzetti F (2019) The role of parenteral nutrition in patients with malignant bowel obstruction. Support Care Cancer 27:4393–4399
Muscaritoli M, Arends J, Bachmann P et al (2021) ESPEN practical guideline: clinical nutrition in cancer. Clin Nutr 40:2898–2913
NHS Scotland (2020) Scottish palliative care guidelines. Bowel Obstruction. https://www.palliativecareguidelines.scot.nhs.uk/guidelines/symptom-control/bowel-obstruction.aspx Accessed on: 15th July 2021
Druml C, Ballmer PE, Druml W et al (2016) ESPEN guideline on ethical aspects of artificial nutrition and hydration. Clin Nutr 35:545–56
Vanhauwaert E, Matthys C, Verdonck L et al (2015) Low-residue and low-fiber diets in gastrointestinal disease management. Adv Nutr 6:820–7
Bozzetti F (2015) Nutrition, hydration, and patient’s preferences at the end of life. Support Care Cancer 23:1487–8
Coelho TA, Wainstein AJA, Drummond-Lage AP (2020) Hypodermoclysis as a strategy for patients with end-of-life cancer in home care settings. Am J Hosp Palliat Care 37:675–682
Good P, Richard R, Syrmis W, et al. (2014) Medically assisted hydration for adult palliative care patients. Cochrane Database Syst Rev 23;(4):CD006273. https://doi.org/10.1002/14651858.CD006273
Lokker ME, van der Heide A, Oldenmenger WH et al (2021) Hydration and symptoms in the last days of life. BMJ Support Palliat Care 11:335–343
Raijmakers NJH, van Zuylen L, Costantini M, et al. (2011) Artificial nutrition and hydration in the last week of life in cancer patients. A systematic literature review of practices and effects. Ann Oncol 22:1478–1486
Bruera E, Hui D, Dalal S et al (2013) Parenteral hydration in patients with advanced cancer: a multicenter, double-blind, placebo-controlled randomized trial. J Clin Oncol 31:111–8
Dreesen M, Foulon V, Spriet I et al (2013) Epidemiology of catheter-related infections in adult patients receiving home parenteral nutrition: a systematic review. Clin Nutr 32:16–26
Pittiruti M, Hamilton H, Biffi R et al (2009) ESPEN guidelines on parenteral nutrition: central venous catheters (access, care, diagnosis and therapy of complications). Clin Nutr 28:365–77
Sowerbutts AM, Lal S, Sremanakova J, et al. (2018) Home parenteral nutrition for people with inoperable malignant bowel obstruction. Cochrane Database Syst Rev 8:CD012812
Bozzetti F, Cozzaglio L, Biganzoli E et al (2002) Quality of life and length of survival in advanced cancer patients on home parenteral nutrition. Clin Nutr 21:281–8
Cotogni P, De Carli L, Passera R et al (2017) Longitudinal study of quality of life in advanced cancer patients on home parenteral nutrition. Cancer Med 6:1799–1806
Finocchiaro C, Gervasio S, Agnello E et al (2002) Multicentric study on home parenteral nutrition in advanced cancer patients. Riv Ital Nutrizione Parenterale Enterale 20:98–107
Naghibi M, Smith TR, Elia M (2015) A systematic review with meta-analysis of survival, quality of life and cost-effectiveness of home parenteral nutrition in patients with inoperable malignant bowel obstruction. Clin Nutr 34:825–37
Bozzetti F, Cotogni P, Lo Vullo S et al (2015) Development and validation of a nomogram to predict survival in incurable cachectic cancer patients on home parenteral nutrition. Ann Oncol 26:2335–40
Messing B, Lémann M, Landais P et al (1995) Prognosis of patients with nonmalignant chronic intestinal failure receiving long-term home parenteral nutrition. Gastroenterology 108:1005–10
Ruggeri E, Giannantonio M, Agostini F et al (2020) Home artificial nutrition in palliative care cancer patients: Impact on survival and performance status. Clin Nutr 39:3346–3353
Brard L, Weitzen S, Strubel-Lagan SL et al (2006) The effect of total parenteral nutrition on the survival of terminally ill ovarian cancer patients. Gynecol Oncol 103:176–80
Chouhan J, Gupta R, Ensor J et al (2016) Retrospective analysis of systemic chemotherapy and total parenteral nutrition for the treatment of malignant small bowel obstruction. Cancer Med 5:239–47
Santarpia L, Alfonsi L, Pasanisi F et al (2006) Predictive factors of survival in patients with peritoneal carcinomatosis on home parenteral nutrition. Nutrition 22:355–60
Dzierzanowski T, Sobocki J (2021) Survival of patients with multi-level malignant bowel obstruction on total parenteral nutrition at home. Nutrients 13:1–12
Jairam V, Lee V, Park HS et al (2019) Treatment-related complications of systemic therapy and radiotherapy. JAMA Oncol 5:1028–1035
Shim J, Seo TS, Song MG et al (2014) Incidence and risk factors of infectious complications related to implantable venous-access ports. Korean J Radiol 15:494–500
Niedzwiedz CL, Knifton L, Robb KA et al (2019) Depression and anxiety among people living with and beyond cancer: a growing clinical and research priority. BMC Cancer 19:943
Krebber AM, Buffart LM, Kleijn G et al (2014) Prevalence of depression in cancer patients: a meta-analysis of diagnostic interviews and self-report instruments. Psychooncology 23:121–30
Caillet P, Liuu E, Raynaud Simon A et al (2017) Association between cachexia, chemotherapy and outcomes in older cancer patients: a systematic review. Clin Nutr 36:1473–1482
Ni J, Zhang L (2020) Cancer Cachexia: Definition, staging, and emerging treatments. Cancer Manag Res 12:5597–5605
Vachon ML, Kristjanson L, Higginson I (1995) Psychosocial issues in palliative care: the patient, the family, and the process and outcome of care. J Pain Symptom Manage 10:142–50
Möllerberg ML, Årestedt K, Sandgren A et al (2020) Adaptation and psychometric evaluation of the short version of family sense of coherence scale in a sample of persons with cancer in the palliative stage and their family members. Palliat Support Care 18:24–32
Daines P, Stilos K, Moura S et al (2013) Nurses’ experiences caring for patients and families dealing with malignant bowel obstruction. Int J Palliat Nurs 19:593–8
Platt V (2001) Malignant bowel obstruction: so much more than symptom control. Int J Palliat Nurs 7:547–54
Chase DM, Monk BJ, Wenzel LB et al (2008) Supportive care for women with gynecologic cancers. Expert Rev Anticancer Ther 8:227–41
Cusimano MC, Sajewycz K, Nelson M et al (2020) Supported self-management as a model for end-of-life care in the setting of malignant bowel obstruction: a qualitative study. Gynecol Oncol 157:745–753
Hoppenot C, Hlubocky FJ, Chor J et al (2020) Approach to palliative care consultation for patients with malignant bowel obstruction in gynecologic oncology: a qualitative analysis of physician perspectives. JCO Oncol Pract 16:483–489
Fallon EA, Miner TJ (2020) Malignant bowel obstruction in the time of the COVID-19 pandemic. Am Surg 86:1473–1477
Acknowledgements
The authors would like to thank MASCC for their in-kind support. The authors would also like to acknowledge Caleb Nault for his help in the article retrieval. Ainhoa Madariaga has performed this work as part of her PhD program at the Autonomous University of Barcelona (Spain). She has received support from the American Society of Clinical Oncology (ASCO) Conquer Cancer Young Investigator Award for research in malignant bowel obstruction.
Author information
Authors and Affiliations
Contributions
All authors were involved in the conception and design, data analysis and interpretation, and manuscript drafting. AM, JL, and SL were involved in data acquisition and final editing.
Corresponding author
Ethics declarations
Ethics approval
Ethics approval was not required for this study.
Consent to participate
This study did not directly involve any human subjects and, therefore, consent was not applicable for this study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Madariaga, A., Lau, J., Ghoshal, A. et al. MASCC multidisciplinary evidence-based recommendations for the management of malignant bowel obstruction in advanced cancer. Support Care Cancer 30, 4711–4728 (2022). https://doi.org/10.1007/s00520-022-06889-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-022-06889-8