Summary
Background
This study evaluated the prevalence of spreading pathways in nontuberculous mycobacteria (NTM) pulmonary infections according to nodular distribution patterns seen on chest computed tomography (CT).
Methods
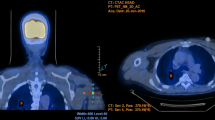
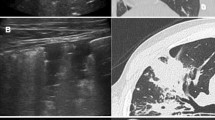
This study included 63 patients diagnosed with NTM lung infections who underwent CT at our institution. A retrospective analysis of CT images focused on the presence and distribution of nodules, presence of intrathoracic lymphadenopathy and the predominant side of infection in the lungs. The findings were classified into five groups; centrilobular (bronchogenic spread), perilymphatic (lymphangitic spread), random (hematogenous spread), combined pattern and no nodules present. The groups were then compared according to other CT findings.
Results
Among 51 (81%) patients identified with a nodular pattern on chest CT, 25 (39.8%) presented with centrilobular, 7 (11.1%) with perilymphatic, 6 (9.5%) with random and 13 (20.6%) with combined nodular patterns but located in different areas of the lungs. The right side of the lungs was predominant in 38 cases (60.3%). Intrathoracic lymphadenopathy was evident in 20 patients (31.7%). Significant differences in distributions of nodular patterns were seen in patients infected with Mycoplasma avium complex (MAC) associated with centrilobular pattern (p = 0.0019) and M. fortuitum associated with random pattern (p = 0.0004). Some of the findings were related to perilymphatic nodules between other isolated species of NTM (p = 0.0379).
Conclusion
The results of this study showed a high proportion of perilymphatic nodules and right-sided predominance in the upper lobe, which, combined with intrathoracic lymphadenopathy is highly suggestive of the lymphangitic spread of lung NTM infections.



Similar content being viewed by others
Abbreviations
- CT:
-
Computed tomography
- HRCT:
-
High-resolution computed tomography
- M:
-
Mycobacteria
- MAC:
-
Mycobacterium avium complex
- NTM:
-
Nontuberculous mycobacteria
- TB:
-
Tuberculosis
References
Verschakelen JA, De Wever W. Computed tomography of the lung. Berlin, Heidelberg, New York: Springer; 2018.
Remy-Jardin M, Beuscart R, Sault M, Marquette C, Remy J. Subpleural micronodules in diffuse infiltrative lung diseases: evaluation with thin section CT scans. Radiology. 1990;177:133–9.
Raoof S, Amchentsev A, Vlahos I, Goud A, Naidich DP. Pictorial essay: multinodular disease—a high-resolution CT scan diagnostic algorithm. Chest. 2006;129:805–15.
Collins J, Blankenbaker D, Stern EJ. CT patterns of bronchiolar disease: what is tree-in-bud? AJR Am J Roentgenol. 1998;171:365–70.
Gallardo X, Castaner E, Mata J, et al. Nodular pattern at lung computed tomography in fat embolism syndrome. A helpful finding. J Comput Assist Tomogr. 2006;30:254–7.
Colby TV, Swensen SJ. Anatomic distribution and histopathologic patterns in diffuse lung disease: correlation with HRCT. J Thorac Imaging. 1996;11:1–26.
Voloudaki AE, Tritou IN, Magkanas EG, et al. HRCT in miliary lung disease. Acta Radiol. 1999;40:451–6.
Kim TS, Koh WJ, Han J, Chung MJ, Lee JH, Lee KS, et al. Hypothesis on the evolution of cavitary lesions in nontuberculous mycobacterial pulmonary infection: thin-section CT and histopathologic correlation. AJR Am J Roentgenol. 2005;184(4):1247–52.
McEvoy S, Lavelle L, Kilcoyne A, McCarthy C, deJong PA, Loeve M, et al. High-resolution CT of nontuberculous mycobacterium infection in adult CF patients: diagnostic accuracy. Eur Radiol. 2012;22(12):2736–42.
Polverosi R, Guarise A, Balestro E, Carloni A, Dalpiaz G, Feragalli B. High-resolution CT of nontuberculous mycobacteria pulmonary infection in immunocompetent, non-HIV-positive patients. Radiol Med. 2010;115(2):191–204.
Kwak N, Lee CH, Lee HJ, Kang YA, Lee JH, Han SK, et al. Non-tuberculous mycobacterial lung disease: diagnosis based on computed tomography of the chest. Eur Radiol. 2016;26(12):4449–56.
Jeong YJ, Lee KS, Koh WJ, Han J, Kim TS, Kwon OJ. Nontuberculous mycobacterial pulmonary infection in immunocompetent patients: comparison of thin-section CT and histopathologic findings. Radiology. 2004;231(3):880–6.
Lee KS, Kim TS, Han J, Hwang JH, Yoon JH, Kim Y, et al. Diffuse micronodular lung disease: HRCT and pathologic findings. J Comput Assist Tomogr. 1999;23(1):99–106.
Ko JM, Park HJ, Cho DG, Kim CH. CT differentiation of tuberculous and non-tuberculous pleural infection, with emphasis on pulmonary changes. Int J Tuberc Lung Dis. 2015;19(11):1361–8.
Klein E. Contributions to the normal and pathological anatomy of the lymphatic system of the lungs. Proc R Soc Lond. 1874;22:133–45.
Basaraba RJ, Smith EE, Shanley CA, Orme IM. Pulmonary lymphatics are primary sites of Mycobacterium tuberculosis infection in guinea pigs infected by aerosol. Infect Immun. 2006;74(9):5397–401.
Behr MA, Waters WR. Is tuberculosis a lymphatic disease with a pulmonary portal? Lancet Infect Dis. 2014;14(3):250–5.
Pereira M, Gazzoni FF, Marchiori E, Irion K, Moreira J, Giacomelli IL, et al. High-resolution CT findings of pulmonary Mycobacterium tuberculosis infection in renal transplant recipients. Br J Radiol. 2016;89(1058):20150686.
He W, Chen BD, Lv Y, Zhou Z, Xu JP, Lv PX, et al. Use of low-dose computed tomography to assess pulmonary tuberculosis among healthcare workers in a tuberculosis hospital. Infect Dis Poverty. 2017;6(1):68.
Kienzl-Palma D, Prosch H. Thoracic manifestation of tuberculosis. Radiologe. 2016;56(10):866–73.
McGuinness G, Naidich DP, Jagirdar J, Leitman B, McCauley DI. High resolution CT findings in miliary lung disease. J Comput Assist Tomogr. 1992;16:384–90.
Chu H, Li B, Zhao L, Huang D, Xu J, Zhang J, et al. Tree-in bud pattern of chest CT images for diagnosis of Mycobacterium abscesses. Int J Clin Exp Med. 2015;8(10):18705–12.
Schiff HF, Jones S, Achaiah A, Pereira A, Stait G, Green B. Clinical relevance of non-tuberculous mycobacteria isolated from respiratory specimens: seven year experience in a UK hospital. Sci Rep. 2019;9(1):1730.
Yasin H, Mangano WE, Malhotra P, Farooq A, Mohamed H. Hot tub lung: a diagnostic challenge. Cureus. 2017;9(8):e1617.
Kim C, Park SH, Oh SY, Kim SS, Jo KW, Shim TS, et al. Comparison of chest CT findings in nontuberculous mycobacterial diseases vs. Mycobacterium tuberculosis lung disease in HIV-negative patients with cavities. PLoS One. 2017;12(3):e174240.
Yuan MK, Chang CY, Tsai PH, Lee YM, Huang JW, Chang SC. Comparative chest computed tomography findings of non-tuberculous mycobacterial lung diseases and pulmonary tuberculosis in patients with acid fast bacilli smear-positive sputum. BMC Pulm Med. 2014;14:65.
Kuhlman JE, Deutsch JH, Fishman EK, Siegelman SS. CT features of thoracic mycobacterial disease. Radiographics. 1990;10:413–31.
Uitenbroek DG. SISA Exact test. 1997. https://www.quantitativeskills.com/sisa/statistics/fiveby2.htm. Accessed 18 Feb 2020.
Ko JM, Park HJ, Kim CH. Clinicoradiologic evidence of pulmonary lymphatic spread in adult patients with tuberculosis. AJR Am J Roentgenol. 2015;204(1):38–43.
Mehta AR, Mehta PR, Mehta RL. A cough conundrum in a patient with a previous history of BCG immunotherapy for bladder cancer. BMJ Case Rep. 2012;24:2012.
Marusić A, Katalinić-Janković V, Popović-Grle S, Janković M, Mazuranić I, Puljić I, et al. Mycobacterium xenopi pulmonary disease—epidemiology and clinical features in non-immunocompromised patients. J Infect. 2009;58(2):108–12.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
A. Marušić, I. Kuhtić, I. Mažuranić, M. Janković, G. Glodić, I. Sabol, and L. Stanić declare that they have no competing interests.
Ethical standards
All procedures performed in studies involving human participants or on human tissue were in accordance with the ethical standards of the institutional and/or national research committee and with the 1975 Helsinki declaration and its later amendments or comparable ethical standards. This is a retrospective study. For this type of study, formal consent is not required, because no personal data were contained and there was no concern about identifying information.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Author Contribution
All authors contributed to the study conception and design. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Rights and permissions
About this article
Cite this article
Marušić, A., Kuhtić, I., Mažuranić, I. et al. Nodular distribution pattern on chest computed tomography (CT) in patients diagnosed with nontuberculous mycobacteria (NTM) infections. Wien Klin Wochenschr 133, 470–477 (2021). https://doi.org/10.1007/s00508-020-01701-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00508-020-01701-1