Abstract
Key message
The high-wood-density species displays greater water limitation tolerance, as it maintains leaf transpiration under drought conditions.
Abstract
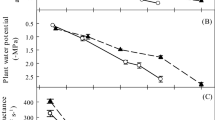
The relationship between environmental conditions and plant hydraulic safety is essential to understand species’ strategies to minimize damage to their hydraulic structure yet maintain function. In the Brazilian semi-arid, the relationships between rainfall seasonality, hydraulic conductivity, wood density, stomatal conductance, and phenology in different species still needs to be clarified. To better understand these relationships, we selected two deciduous trees species with contrasting wood density: (1) Commiphora leptophloeos (Mart.) J.B. Gillett (low wood density) and (2) Cenostigma pyramidale (Tul.) E. Gagnon & G. P. Lewis (high wood density) from the Caatinga dry forest of northeast Brazil. We tracked monthly measurements of whole-tree hydraulic conductivity, leaf stomatal conductance, leaf transpiration rate, xylem water potential, and phenology. We found that the low-wood-density species had a higher whole-tree hydraulic conductivity and an early leaf flush and fall. In addition, lower leaf transpiration rate and higher water storage capacity maintained high xylem water potential and stomatal conductance values, especially in the rainy season. On the other hand, the high-wood-density species had a lower whole-tree hydraulic conductivity and higher leaf transpiration rate, even during the dry season. These results point to the divergent hydraulic strategies employed by each species, further suggesting opposing hydraulic safety pathways during drought.







Similar content being viewed by others
Data availability
All the analyzed data are included in this paper.
References
Aleixo I, Norris D, Hemerik L et al (2019) Amazonian rainforest tree mortality driven by climate and functional traits. Nat Clim Change 9:384–388. https://doi.org/10.1038/s41558-019-0458-0
Allen K, Dupuy JM, Gei MG et al (2017) Will seasonally dry tropical forests be sensitive or resistant to future changes in rainfall regimes? Environ Res Lett. https://doi.org/10.1088/1748-9326/aa5968
Anderegg WRL, Klein T, Bartlett M et al (2016) Meta-analysis reveals that hydraulic traits explain cross-species patterns of drought-induced tree mortality across the globe. Proc Natl Acad Sci U S A 113:5024–5029. https://doi.org/10.1073/pnas.1525678113
Andrade JL, Meinzer FC, Goldstein G, Schnitzer SA (2005) Water uptake and transport in lianas and co-occurring trees of a seasonally dry tropical forest. Trees Struct Funct 19:282–289. https://doi.org/10.1007/s00468-004-0388-x
Barros IO, Soares AA (2013) Adaptações anatômicas em folhas de marmeleiro e velame da caatinga brasileira. Rev Cienc Agron 44:192–198. https://doi.org/10.1590/s1806-66902013000100024
Borchert R (1994) Soil and stem water storage determine phenology and distribution of tropical dry forest trees. Ecology 75:1437–1449. https://doi.org/10.2307/1937467
Borchert R, Pockman WT (2005) Water storage capacitance and xylem tension in isolated branches of temperate and tropical trees. Tree Physiol 25:457–466. https://doi.org/10.1093/treephys/25.4.457
Bosch DD, Marshall LK, Teskey R (2014) Forest transpiration from sap flux density measurements in a Southeastern Coastal Plain riparian buffer system. Agric for Meteorol 187:72–82. https://doi.org/10.1016/j.agrformet.2013.12.002
Bréda N, Huc R, Granier A, Dreyer E (2006) Temperate forest trees and stands under severe drought: a review of ecophysiological responses, adaptation processes and long-term consequences. Ann for Sci 63:625–644
Brito P, Lorenzo JR, González-Rodríguez ÁM et al (2014) Canopy transpiration of a Pinus canariensis forest at the tree line: Implications for its distribution under predicted climate warming. Eur J for Res 133:491–500. https://doi.org/10.1007/s10342-014-0779-5
Brito NDS, Medeiros MJS, Souza ES, Lima ALA (2022) Drought response strategies for deciduous species in the semiarid Caatinga derived from the interdependence of anatomical, phenological and bio-hydraulic attributes. Flora Morphol Distrib Funct Ecol Plants. https://doi.org/10.1016/j.flora.2022.152009
Brodribb TJ, Holbrook NM, Gutiérrez MV (2002) Hydraulic and photosynthetic co-ordination in seasonally dry tropical forest trees. Plant Cell Environ 25:1435–1444. https://doi.org/10.1046/j.1365-3040.2002.00919.x
Carrasco OL, Bucci SJ, Di Francescantonio D et al (2014) Water storage dynamics in the main stem of subtropical tree species differing in wood density, growth rate and life history traits. Tree Physiol 35:354–365. https://doi.org/10.1093/treephys/tpu087
Chave J, Coomes D, Jansen S et al (2009) Towards a worldwide wood economics spectrum. Ecol Lett 12:351–366. https://doi.org/10.1111/j.1461-0248.2009.01285.x
Chave J (2005) Measuring wood density for tropical forest trees. A field manual for the CTFS sites. Universite Paul Sabatier, Toulouse, France. Available from https://afritron.org/upload/en/manuals/wood_density_english%5B1%5D.pdf. Accessed 21 Mar 2024
Chen L, Zhang Z, Li Z et al (2011) Biophysical control of whole tree transpiration under an urban environment in Northern China. J Hydrol 402:388–400. https://doi.org/10.1016/j.jhydrol.2011.03.034
Chen D, Wang Y, Liu S et al (2014) Response of relative sap flow to meteorological factors under different soil moisture conditions in rainfed jujube (Ziziphus jujuba Mill.) plantations in semiarid Northwest China. Agric Water Manag 136:23–33. https://doi.org/10.1016/j.agwat.2014.01.001
de Lima ALA, de Sá Barretto Sampaio EV, de Castro CC et al (2012) Do the phenology and functional stem attributes of woody species allow for the identification of functional groups in the semiarid region of Brazil? Trees Struct Funct 26:1605–1616. https://doi.org/10.1007/s00468-012-0735-2
de Oliveira CC, Zandavalli RB, de Lima ALA, Rodal MJN (2015) Functional groups of woody species in semi-arid regions at low latitudes. Aust Ecol 40:40–49. https://doi.org/10.1111/aec.12165
Fournier LA (1974) Un método cuantitativo para la medición de características fenológicas en árboles. Turrialba 24:422–423
Gleason SM, Westoby M, Jansen S et al (2016) Weak tradeoff between xylem safety and xylem-specific hydraulic efficiency across the world’s woody plant species. New Phytol 209:123–136. https://doi.org/10.1111/nph.13646
Granier A (1985) Une nouvelle méthode pour la mesure du flux de sève brute dans le tronc des arbres. Ann Des Sci for 42:193–200. https://doi.org/10.1051/forest:19850204
Granier A (1987) Evaluation of transpiration in a Douglas-fir stand by means of sap flow measurements. Tree Physiol 3:309–320. https://doi.org/10.1093/treephys/3.4.309
Greenwood S, Ruiz-Benito P, Martínez-Vilalta J et al (2017) Tree mortality across biomes is promoted by drought intensity, lower wood density and higher specific leaf area. Ecol Lett 20:539–553
Grossiord C, Sevanto S, Borrego I et al (2017) Tree water dynamics in a drying and warming world. Plant Cell Environ 40:1861–1873. https://doi.org/10.1111/pce.12991
Hacke U (2015) Functional and ecological Xylem anatomy. https://doi.org/10.1007/978-3-319-15783-2
Hernandez-Santana V, Fernández JE, Rodriguez-Dominguez CM et al (2016) The dynamics of radial sap flux density reflects changes in stomatal conductance in response to soil and air water deficit. Agric for Meteorol 218–219:92–101. https://doi.org/10.1016/j.agrformet.2015.11.013
Huxman TE, Snyder KA, Tissue D et al (2004) Precipitation pulses and carbon fluxes in semiarid and arid ecosystems. Oecologia 141:254–268. https://doi.org/10.1007/s00442-004-1682-4
IPCC—Intergovernmental Panel on Climate Change (2020) https://www.ipcc.ch/. Accessed 22 July 2020
Janssen TAJ, Hölttä T, Fleischer K et al (2020) Wood allocation trade-offs between fiber wall, fiber lumen, and axial parenchyma drive drought resistance in neotropical trees. Plant Cell Environ 43:965–980. https://doi.org/10.1111/pce.13687
Jiao L, Lu N, Fu B et al (2018) Evapotranspiration partitioning and its implications for plant water use strategy: evidence from a black locust plantation in the semi-arid Loess Plateau, China. For Ecol Manag 424:428–438. https://doi.org/10.1016/j.foreco.2018.05.011
Kassambara A, Mundt F (2019) factoextra: Extract and visualize the results of multivariate data analyses. R package version 1.0.6. https://CRAN.R-project.org/package=factoextra
Kottek M, Grieser J, Beck C et al (2006) World map of the Köppen-Geiger climate classification updated. Meteorol Zeitschrift 15:259–263. https://doi.org/10.1127/0941-2948/2006/0130
Le S, Josse J, Husson F (2008) FactoMineR: an R package for multivariate analysis. J Stat Softw 25:1–18. https://doi.org/10.18637/jss.v025.i01
Lima ALA, Rodal MJN (2010) Phenology and wood density of plants growing in the semi-arid region of northeastern Brazil. J Arid Environ 74:1363–1373. https://doi.org/10.1016/j.jaridenv.2010.05.009
Lima ALA, Rodal MJN, Castro CC et al (2021) Phenology of high- and low-density wood deciduous species responds differently to water supply in tropical semiarid regions. J Arid Environ. https://doi.org/10.1016/j.jaridenv.2021.104594
Markesteijn L, Poorter L, Paz H et al (2011) Ecological differentiation in xylem cavitation resistance is associated with stem and leaf structural traits. Plant Cell Environ 34:137–148. https://doi.org/10.1111/j.1365-3040.2010.02231.x
Oliveira RS, Costa FRC, van Baalen E et al (2019) Embolism resistance drives the distribution of Amazonian rainforest tree species along hydro-topographic gradients. New Phytol 221:1457–1465. https://doi.org/10.1111/nph.15463
Paloschi RA, Ramos DM, Ventura DJ et al (2021) Environmental drivers of water use for caatinga woody plant species: combining remote sensing phenology and sap flow measurements. Remote Sens 13:1–18. https://doi.org/10.3390/rs13010075
Pereira PDC, Da Silva TGF, Zolnier S et al (2015) Morfogênese da palma forrageira irrigada por gotejamento. Rev Caatinga 28:184–195
Pérez-Harguindeguy N, Díaz S, Garnier E et al (2013) New handbook for standardised measurement of plant functional traits worldwide. Aust J Bot 61:167–234. https://doi.org/10.1071/BT12225
Pineda-García F, Paz H, Meinzer FC (2012) Drought resistance in early and late secondary successional species from a tropical dry forest: the interplay between xylem resistance to embolism, sapwood water storage and leaf shedding. Plant Cell Environ 36:405–418. https://doi.org/10.1111/j.1365-3040.2012.02582.x
Pineda-García F, Paz H, Meinzer FC, Angeles G (2015) Exploiting water versus tolerating drought: water-use strategies of trees in a secondary successional tropical dry forest. Tree Physiol 36:208–217. https://doi.org/10.1093/treephys/tpv124
Pinho BX, Tabarelli M, Engelbrecht BMJ et al (2019) Plant functional assembly is mediated by rainfall and soil conditions in a seasonally dry tropical forest. Basic Appl Ecol 40:1–11. https://doi.org/10.1016/j.baae.2019.08.002
Pinto AM, Morellato LPC, Barbosa AP (2008) Fenologia reprodutiva de Dipteryx odorata (Aubl.) Willd (Fabaceae) em duas áreas de floresta na Amazônia Central. Acta Amaz 38:643–649. https://doi.org/10.1590/S0044-59672008000400006
Poorter L, Wright SJ, Paz H et al (2008) Are functional traits good predictors of demographic rates? Evidence from five neotropical forests. Ecology 89:1908–1920. https://doi.org/10.1890/07-0207.1
Poorter L, Rozendaal DMA, Bongers F et al (2019) Wet and dry tropical forests show opposite successional pathways in wood density but converge over time. Nat Ecol Evol 3:928–934. https://doi.org/10.1038/s41559-019-0882-6
Poorter L, McDonald I, Alarcon A et al (2010) The importance of wood traits and hydraulic conductance for the performance and life history strategies of 42 rainforest tree species. New Phytol 185:481–492. https://doi.org/10.1111/j.1469-8137.2009.03092.x
R Core Team (2022) A language and environment for statistical computing, R foundation for statistical computing, Vienna, Austria., https://www.R-project.org/
Resco de Dios V, Gessler A (2021) Sink and source co-limitation in the response of stored non-structural carbohydrates to an intense but short drought. Trees Struct Funct. https://doi.org/10.1007/s00468-021-02116-9
Rodriguez-Dominguez CM, Carins Murphy MR, Lucani C, Brodribb TJ (2018) Mapping xylem failure in disparate organs of whole plants reveals extreme resistance in olive roots. New Phytol 218:1025–1035. https://doi.org/10.1111/nph.15079
Rosner S, Heinze B, Savi T, Dalla-Salda G (2019) Prediction of hydraulic conductivity loss from relative water loss: new insights into water storage of tree stems and branches. Physiol Plant 165:843–854. https://doi.org/10.1111/ppl.12790
Santos M, Barros V, Lima L et al (2021) Whole plant water status and non-structural carbohydrates under progressive drought in a Caatinga deciduous woody species. Trees. https://doi.org/10.1007/s00468-021-02113-y
Sevanto S, Mcdowell NG, Dickman LT et al (2014) How do trees die? A test of the hydraulic failure and carbon starvation hypotheses. Plant Cell Environ 37:153–161. https://doi.org/10.1111/pce.12141
Silva PF, Lima JRS, Antonino ACD et al (2017) Seasonal patterns of carbon dioxide, water and energy fluxes over the Caatinga and grassland in the semi-arid region of Brazil. J Arid Environ 147:71–82. https://doi.org/10.1016/j.jaridenv.2017.09.003
Silva RD, Santos AMM, Tabarelli M (2003) Riqueza e diversidade de plantas lenhosas em cinco unidades de paisagem da Caatinga. In: Ecologia e conservação da caatinga. Ed. Universitária da UFRPE, Recife, pp 337–365. Available from https://repositorio.ufpe.br/handle/123456789/742. Accessed 21 Mar 2024
Song L, Zhu J, Li M et al (2018) Canopy transpiration of Pinus sylvestris var. mongolica in a sparse wood grassland in the semiarid sandy region of Northeast China. Agric for Meteorol 250–251:192–201. https://doi.org/10.1016/j.agrformet.2017.12.260
Sperry JS (2000) Hydraulic constraints on plant gas exchange. Agric For Meteorol 104:13–23. https://doi.org/10.1016/S0168-1923(00)00144-1
Sperry JS, Donnelly JR, Tyree MT (1988) A method for measuring hydraulic conductivity and embolism in xylem. Plant Cell Environ 11:35–40. https://doi.org/10.1111/j.1365-3040.1988.tb01774.x
Sperry JS, Stiller V, Hacke UG (2003) Xylem hydraulics and the soil–plant–atmosphere continuum. Agron J 95:1362. https://doi.org/10.2134/agronj2003.1362
Steppe K, Vandegehuchte MW, Tognetti R, Mencuccini M (2015) Sap flow as a key trait in the understanding of plant hydraulic functioning. Tree Physiol 35:341–345. https://doi.org/10.1093/treephys/tpv033
Tie Q, Hu H, Tian F et al (2017) Environmental and physiological controls on sap flow in a subhumid mountainous catchment in North China. Agric for Meteorol. https://doi.org/10.1016/j.agrformet.2017.03.018
Tognetti R, Giovannelli A, Lavini A et al (2009) Assessing environmental controls over conductances through the soil-plant-atmosphere continuum in an experimental olive tree plantation of southern Italy. Agric for Meteorol 149:1229–1243. https://doi.org/10.1016/j.agrformet.2009.02.008
Tyree MT, Ewers FW (1991) The hydraulic architecture of trees and other woody plants. New Phytol 119:345–360. https://doi.org/10.1111/j.1469-8137.1991.tb00035.x
Van Den Honert TH (1948) Water transport in plants as a catenary process. Discuss Faraday Soc 3:146–153
Venturas MD, Sperry JS, Hacke UG (2017) Plant xylem hydraulics: what we understand, current research, and future challenges. J Integr Plant Biol 59:356–389. https://doi.org/10.1111/jipb.12534
Vico G, Thompson SE, Manzoni S et al (2014) Climatic, ecophysiological, and phenological controls on plant ecohydrological strategies in seasonally dry ecosystems. Ecohydrology 8:660–681. https://doi.org/10.1002/eco.1533
Weiner J (2019) pca3d: Three Dimensional PCA Plots. R package version 0.10.1. https://CRAN.R-project.org/package=pca3d
Wright CL, de Lima ALA, de Souza ES et al (2021) Plant functional types broadly describe water use strategies in the Caatinga, a seasonally dry tropical forest in northeast Brazil. Ecol Evol 11:11808–11825. https://doi.org/10.1002/ece3.7949
Wright CL, West JB, de Lima ALA et al (2023) Contrasting water-use strategies revealed by species-specific transpiration dynamics in the Caatinga dry forest. Tree Physiol. https://doi.org/10.1093/treephys/tpad137
Wright IJ, Reich PB, Westoby M et al (2004) The worldwide leaf economics spectrum. Nat 428:821–827. https://doi.org/10.1038/nature02403
Xu X, Medvigy D, Powers JS et al (2016) Diversity in plant hydraulic traits explains seasonal and inter-annual variations of vegetation dynamics in seasonally dry tropical forests. New Phytol 212:80–95. https://doi.org/10.1111/nph.14009
Zeppel MJB, Macinnis-Ng CMO, Yunusa IAM et al (2008) Long term trends of stand transpiration in a remnant forest during wet and dry years. J Hydrol 349:200–213. https://doi.org/10.1016/j.jhydrol.2007.11.001
Zhang B, Xu D, Liu Y et al (2016) Multi-scale evapotranspiration of summer maize and the controlling meteorological factors in north China. Agric for Meteorol 216:1–12. https://doi.org/10.1016/j.agrformet.2015.09.015
Zhang H, Wei W, Chen L, Wang L (2017) Effects of terracing on soil water and canopy transpiration of Pinus tabulaeformis in the Loess Plateau of China. Ecol Eng 102:557–564. https://doi.org/10.1016/j.ecoleng.2017.02.044
Acknowledgements
We thank the members of the Semiarid Ecohydrology Study Group of the Federal Rural University of Pernambuco, Academic Unit of Serra Talhada (UFRPE/UAST) for assistance in the fieldwork, especially Mirna Clarissa Almeida, Erison Martins, Romário Horas, Lypson Simões, and Fernando Barros for technical support in the field. We also thank the Graduate Program in Plant Production of UFRPE/UAST for the availability of equipment and laboratories and Senhor Homem Bom Magalhães for accessing the field where we carried out the study.
Funding
This work was funded by the Fundação do Amparo a Ciência e Tecnologia de Pernambuco-FACEPE (APQ-1196-5.03/15). MM is funded by (FACEPE Fellow-IBPG-1009-5.01/18); Observatório Nacional da Água e da Dinâmica do Carbono no Bioma Caatinga-ONDACBC: FACEPE (APQ-0532-5. 01/14); additional funding includes: Conselho Nacional de Desenvolvimento Científico e Tecnológico-CNPq (465764/2014-2); Coordenação de Aperfeiçoamento de Pessoal de Nível Superior-CAPES (88887.136369/2017-00); AIC-GRHASSA: FACEPE (APQ-0296-5.01/17), and PEGASUS: CNPq (441305/2017-2); CAPES/TAMU (006/2014); NSF GRFP (DGE-1252521), 2017.
Author information
Authors and Affiliations
Contributions
MM, CW, ALAL, and ES conceived the study. MM, CW, NDSB, RS, and JRIS collected the data. MM, NDSB, RS, and JRIS analyzed the data. MM wrote the first draft and all authors contributed substantially to the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that they have no competing interests.
Additional information
Communicated by H. Ishii.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Medeiros, M., Wright, C.L., de Lima, A.L.A. et al. Divergent hydraulic strategies of two deciduous tree species to deal with drought in the Brazilian semi-arid region. Trees (2024). https://doi.org/10.1007/s00468-024-02506-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00468-024-02506-9