Abstract
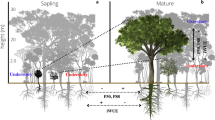
Plant ecophysiological trade-offs between different strategies for tolerating stresses are widely theorized to shape forest functional diversity and vulnerability to climate change. However, trade-offs between hydraulic and stomatal regulation during natural droughts remain under-studied, especially in tropical forests. We investigated eleven mature forest canopy trees in central Amazonia during the strong 2015 El Niño. We found greater xylem embolism resistance (\({P}_{50}\) = − 3.3 ± 0.8 MPa) and hydraulic safety margin (HSM = 2.12 ± 0.57 MPa) than previously observed in more precipitation-seasonal rainforests of eastern Amazonia and central America. We also discovered that taller trees exhibited lower embolism resistance and greater stomatal sensitivity, a height-structured trade-off between hydraulic resistance and active stomatal regulation. Such active regulation of tree water status, triggered by the onset of stem embolism, acted as a feedback to avoid further increases in embolism, and also explained declines in photosynthesis and transpiration. These results suggest that canopy trees exhibit a conservative hydraulic strategy to endure drought, with trade-offs between investment in xylem to reduce vulnerability to hydraulic failure, and active stomatal regulation to protect against low water potentials. These findings improve our understanding of strategies in tropical forest canopies and contribute to more accurate prediction of drought responses.




Similar content being viewed by others
References
Aleixo I, Norris D, Hemerik L et al (2019) Amazonian rainforest tree mortality driven by climate and functional traits. Nat Clim Change 9:384–388. https://doi.org/10.1038/s41558-019-0458-0
Allen CD, Breshears DD, McDowell NG (2015) On underestimation of global vulnerability to tree mortality and forest die-off from hotter drought in the Anthropocene. Ecosphere 6:1–55. https://doi.org/10.1890/ES15-00203.1
Anderegg WRL, Klein T, Bartlett M et al (2016) Meta-analysis reveals that hydraulic traits explain cross-species patterns of drought-induced tree mortality across the globe. Proc Natl Acad Sci U S A 113:5024–5029. https://doi.org/10.1073/pnas.1525678113
Anderegg WRL, Wolf A, Arango-Velez A et al (2018) Woody plants optimise stomatal behaviour relative to hydraulic risk. Ecol Lett 21:968–977
Aragão LEOC, Malhi Y, Roman-Cuesta RM et al (2007) Spatial patterns and fire response of recent Amazonian droughts. Geophys Res Lett 34:1–5. https://doi.org/10.1029/2006GL028946
Araújo AC, Nobre AD, Kruijt B et al (2002) Comparative measurements of carbon dioxide fluxes from two nearby towers in a central Amazonian rainforest: the Manaus LBA site. J Geophys Res 107:8090. https://doi.org/10.1029/2001JD000676
Bates D, Maechler M (2007) Linear mixed-effects models using S4 classes. R Package Version 2:74
Bennett AC, Mcdowell NG, Allen CD et al (2015) Drought-induced mortality patterns and rapid biomass recovery in a terra firme forest in the Colombian Amazon. Nat Plants 1:1–5. https://doi.org/10.1002/ecy.1950
Binks O, Meir P, Rowland L et al (2016) Plasticity in leaf-level water relations of tropical rainforest trees in response to experimental drought. New Phytol 211:477–488. https://doi.org/10.1111/nph.13927
Bonal D, Barigah TS, Granier A, Guehl JM (2000) Late-stage canopy tree species with extremely low δ13C and high stomatal sensitivity to seasonal soil drought in the tropical rainforest of French Guiana. Plant Cell Environ 23:445–459. https://doi.org/10.1046/j.1365-3040.2000.00556.x
Brienen RJW, Phillips OL, Feldpausch TR et al (2015) Long-term decline of the Amazon carbon sink. Nature 519:344–348. https://doi.org/10.1038/nature14283
Brodribb TJ, Holbrook NM (2004) Diurnal depression of leaf hydraulic conductance in a tropical tree species. Plant Cell Environ 27:820–827. https://doi.org/10.1111/j.1365-3040.2004.01188.x
Brum M, Vadeboncoeur MA, Ivanov V et al (2019) Hydrological niche segregation defines forest structure and drought tolerance strategies in a seasonal Amazon forest. J Ecol 107:318–333. https://doi.org/10.1111/1365-2745.13022
Chmura DJ, Guzicka M, McCulloh KA, Zytkowiak R (2015) Limited variation found among Norway spruce half-sib families in physiological response to drought and resistance to embolism. Tree Physiol 36:252–266. https://doi.org/10.1093/treephys/tpv141
Choat B, Jansen S, Brodribb TJ et al (2012) Global convergence in the vulnerability of forests to drought. Nature 491:752–755. https://doi.org/10.1038/nature11688
Crowther TW, Glick HB, Covey KR et al (2015) Mapping tree density at a global scale. Nature 525:201–205. https://doi.org/10.1038/nature14967
Cuartas LA, Tomasella J, Nobre AD et al (2012) Distributed hydrological modeling of a micro-scale rainforest watershed in Amazonia: model evaluation and advances in calibration using the new HAND terrain model. J Hydrol 462–463:15–27. https://doi.org/10.1016/j.jhydrol.2011.12.047
de Barros FV, Bittencourt PRL, Brum M et al (2019) Hydraulic traits explain differential responses of Amazonian forests to the 2015 El Niño-induced drought. New Phytol 223:1253–1266. https://doi.org/10.1111/nph.15909
Domingues TF, Meir P, Feldpausch TR et al (2010) Co-limitation of photosynthetic capacity by nitrogen and phosphorus in West Africa woodlands. Plant Cell Environ 33:959–980. https://doi.org/10.1111/j.1365-3040.2010.02119.x
dos Santos VAHF, Ferreira MJ, Rodrigues JVFC et al (2018) Causes of reduced leaf-level photosynthesis during strong El Niño drought in a Central Amazon forest. Glob Change Biol 24:4266–4279. https://doi.org/10.1111/gcb.14293
Doughty CE, Metcalfe DB, Girardin CAJJ et al (2015) Drought impact on forest carbon dynamics and fluxes in Amazonia. Nature 519:78–82. https://doi.org/10.1038/nature14213
Fauset S, Johnson MO, Gloor M et al (2015) Hyperdominance in Amazonian forest carbon cycling. Nat Commun 6:1–9. https://doi.org/10.1038/ncomms7857
Fauset S, Gloor M, Fyllas NM et al (2019) Individual-based modeling of Amazon forests suggests that climate controls productivity while traits control demography. Front Earth Sci 7:1–19. https://doi.org/10.3389/feart.2019.00083
Fettig CJ, Mortenson LA, Bulaon BM, Foulk PB (2019) Tree mortality following drought in the central and southern Sierra Nevada, California, U.S. For Ecol Manage 432:164–178. https://doi.org/10.1016/j.foreco.2018.09.006
Field A (2018) Discovering Statistics Using IBM SPSS Statistics
Fontes CG, Dawson TE, Jardine K et al (2018) Dry and hot: the hydraulic consequences of a climate change–type drought for Amazonian trees. Philos Trans R Soc B Biol Sci. https://doi.org/10.1098/rstb.2018.0209
Hernandez-Santana V, Rodriguez-Dominguez CM, Fernández JE, Diaz-Espejo A (2016) Role of leaf hydraulic conductance in the regulation of stomatal conductance in almond and olive in response to water stress. Tree Physiol 36:725–735. https://doi.org/10.1093/treephys/tpv146
Hole W, Nepstad DC, Tohver IM et al (2007) Mortality of large trees and lianas following experimental drought in an Amazon forest. Ecol Soc Am 88:2259–2269
Ivanov VY, Hutyra LR, Wofsy SC et al (2012) Root niche separation can explain avoidance of seasonal drought stress and vulnerability of overstory trees to extended drought in a mature Amazonian forest. Water Resour Res 48:1–21. https://doi.org/10.1029/2012WR011972
Jiménez-Muñoz JC, Mattar C, Barichivich J et al (2016) Record-breaking warming and extreme drought in the Amazon rainforest during the course of El Niño 2015–2016. Sci Rep 6:1–7. https://doi.org/10.1038/srep33130
Li L, McCormack ML, Ma C et al (2015) Leaf economics and hydraulic traits are decoupled in five species-rich tropical-subtropical forests. Ecol Lett 18:899–906. https://doi.org/10.1111/ele.12466
Lloyd J, Farquhar GD (2008) Effects of rising temperatures and [CO2] on the physiology of tropical forest trees effects of rising temperatures and [CO2] on the physiology of tropical forest trees. Philos Trans R Soc 363:1811–1817. https://doi.org/10.1098/rstb.2007.0032
Malhi Y, Nobre AD, Grace J et al (1998) Carbon dioxide transfer over a Central Amazonian rain forest. J Geophys Res Atmos 103:31593–31612. https://doi.org/10.1029/98JD02647
Malhi Y, Aragão LEOC, Galbraith D et al (2009) Exploring the likelihood and mechanism of a climate-change-induced dieback of the Amazon rainforest. PNAS 106:20611–20615. https://doi.org/10.1073/pnas.0804619106
McDowell NG, Allen CD (2015) Darcy’s law predicts widespread forest mortality under climate warming. Nat Clim Change 5:669–672. https://doi.org/10.1038/nclimate2641
McDowell NG, Williams AP, Xu C et al (2015) Multi-scale predictions of massive conifer mortality due to chronic temperature rise. Nat Clim Change 6:295–300. https://doi.org/10.1038/nclimate2873
McDowell N, Allen CD, Anderson-Teixeira K et al (2018) Drivers and mechanisms of tree mortality in moist tropical forests. New Phytol 219:851–869. https://doi.org/10.1111/nph.15027
Miranda EJ, Vourlitis GL, Filho NP et al (2005) Seasonal variation in the leaf gas exchange of tropical forest trees in the rain forest—savanna transition of the southern Amazon Basin How to cite this article: seasonal variation in the leaf gas exchange of tropical forest trees. J Trop Ecol 21:451–460. https://doi.org/10.1017/S0266467405002427
Mitchell PJ, Grady APO, Tissue DT et al (2013) Drought response strategies define the relative contributions of hydraulic dysfunction and carbohydrate depletion during tree mortality. New Phytol 197:862–872. https://doi.org/10.1111/nph.12064
Nepstad DC, de Carvalho CR, Davidson EA et al (1994) The role of deep roots in the hydrological and carbon cycles of Amazonian forests and pastures. Nature 372:666–669
Oliveira RS, Dawson TE, Burgess SSO, Nepstad DC (2005) Hydraulic redistribution in three Amazonian trees. Oecologia 145:354–363. https://doi.org/10.1007/s00442-005-0108-2
Pereira L, Bittencourt PRL, Oliveira RS et al (2016) Plant pneumatics: stem air flow is related to embolism - new perspectives on methods in plant hydraulics. New Phytol 211:357–370. https://doi.org/10.1111/nph.13905
Phillips OL, Aragao LEOC, Lewis SL et al (2009) Drought sensitivity of the Amazon rainforest. Science 323:1344–1347. https://doi.org/10.1126/science.1164033
Phillips OL, van der Heijden G, Lewis SL et al (2010) Drought-mortality relationships for tropical forests. New Phytol 187:631–646. https://doi.org/10.1111/j.1469-8137.2010.03359.x
Powers JS, Vargas GG, Brodribb TJ et al (2020) A catastrophic tropical drought kills hydraulically vulnerable tree species. Glob Change Biol 26:3122–3133. https://doi.org/10.1111/gcb.15037
Pratt RB, Castro V, Fickle JC, Jacobsen AL (2020) Embolism resistance of different aged stems of a California Oak species (Quercus Douglasii): optical and microCT methods differ from the benchtop-dehydration standard. Tree Physiol 40(1):5–18
R Development Core Team (2011) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/ (ISBN 3-900051-07-0)
Resco V, Ewers BE, Sun W et al (2009) Drought-induced hydraulic limitations constrain leaf gas exchange recovery after precipitation pulses in the C3 woody legume, Prosopis velutina. New Phytol 181:672–682. https://doi.org/10.1111/j.1469-8137.2008.02687.x
Rowland L, Da Costa ACLL, Galbraith DR et al (2015a) Death from drought in tropical forests is triggered by hydraulics not carbon starvation. Nature 528:1–13. https://doi.org/10.1038/nature15539
Rowland L, Lobo-do-Vale RL, Christoffersen BO et al (2015b) After more than a decade of soil moisture deficit, tropical rainforest trees maintain photosynthetic capacity, despite increased leaf respiration. Glob Chang Biol 21:4662–4672. https://doi.org/10.1111/gcb.13035
Saleska SR, Didan K, Huete AR, Rocha HR (2007) Amazon forests green-up during 2005 drought. Science 318:2007–2007. https://doi.org/10.1126/science.1146663
Scholander PF, Hammel HT, Bradstreet D, Hemmingsen EA (1964) Sap pressure in vascular plants negative hydrostatic pressure can be measured in plants. Science 148:339–346
Skelton RP, West AG, Dawson TE (2015) Predicting plant vulnerability to drought in biodiverse regions using functional traits. Proc Natl Acad Sci U S A 112:5744–5749. https://doi.org/10.1073/pnas.1503376112
Sombroek W (2001) Spatial and temporal patterns of Amazon rainfall. BioOne 30:388–396. https://doi.org/10.1579/0044-7447-30.7.388
Sperry JS (2000) Hydraulic constraints on plant gas exchange. Agric For Meteorol 104:13–23. https://doi.org/10.1016/S0168-1923(00)00144-1
Sperry JS, Love DM (2015) What plant hydraulics can tell us about responses to climate–change droughts. New Phytol 207:14–27. https://doi.org/10.1111/nph.13354
Stpaul NKM, Limousin JM, Rodrígesuez-Calcerrada J et al (2012) Photosynthetic sensitivity to drought varies among populations of Quercus ilex along a rainfall gradient. Funct Plant Biol 39:25–37. https://doi.org/10.1071/FP11090
ter Steege H, Pitman NCA, Sabatier D et al (2013) Hyperdominance in the Amazonian Tree Flora. Science 342:1243092–1243092. https://doi.org/10.1126/science.1243092
Turner NC (1981) Techniques and experimental approaches for the measurement of plant water status. Plan Soil 58(1):339–366. https://doi.org/10.1007/BF02180062
Venturas MD, MacKinnon ED, Dario HL, Jacobsen AL, Brandon Pratt R, Davis SD (2016) Chaparral shrub hydraulic traits, size, and life history types relate to species mortality during California’s Historic Drought of 2014. PLoS ONE 11(7):e0159145
Wang Y, Sperry JS, Anderegg WRL et al (2020) A theoretical and empirical assessment of stomatal optimization modeling. New Phytol 227:311–325. https://doi.org/10.1111/nph.16572
Wu J, Serbin SP, Ely KS et al (2019) The response of stomatal conductance to seasonal drought in tropical forests. Glob Change Biol 81:gcb14820. https://doi.org/10.1111/gcb.14820
Zhang Y, Lamarque LJ, Torres-Ruiz JM et al (2018) Testing the plant pneumatic method to estimate xylem embolism resistance in stems of temperate trees. Tree Physiol 38:1016–1025. https://doi.org/10.1093/treephys/tpy015
Ziegler C, Coste S, Stahl C et al (2019) Large hydraulic safety margins protect Neotropical canopy rainforest tree species against hydraulic failure during drought. Ann For Sci. https://doi.org/10.1007/s13595-019-0905-0
Zuleta D, Duque A, Cardenas D et al (2017) Drought-induced mortality patterns and rapid biomass recovery in a terra firme forest in the Colombian Amazon. Ecology 98:2538–2546. https://doi.org/10.1002/ecy.1950
Acknowledgements
We thank the National Institute of Amazon Research (INPA) and the Large-Scale Biosphere-Atmosphere experiment (LBA) for logistical support. MJF and RSO thanks CNPq for a productivity scholarship.
Funding
This work was supported by the GOAmazon project, jointly funded by the Brazilian Research Foundations of São Paulo State (FAPESP #13/50533-5), and Amazonas State (FAPEAM #062.00570/2014), and the U.S. Department of Energy (awards SC0008383 and SC0011078). Also, we acknowledge additional support by the U.S. National Science Foundation (award #1754803) and Brazil’s Coordination of Improvement of Higher Level Personnel (CAPES).
Author information
Authors and Affiliations
Contributions
MNG, RSO, and MJF designed the research. MNG, VAHFS, AVG, and JVBC collected the data. MNG analyzed the data. MNG, SVCM, SRS, and VI interpreted the analyses. MNG wrote the first draft and RSO, MJF, SVCM, SRS, VAHFS, and VI authors contributed to editing the manuscript.
Corresponding author
Additional information
Communicated by Kate McCulloh.
We found tradeoffs between stomatal regulation and hydraulic safety in individuals Amazon trees facing extreme drought. This is novel for tropical forests, and provides new insight into potential climate change responses in a seasonally wet tropical ecosystem that is particularly at risk during El Nino climate events.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Garcia, M.N., Ferreira, M.J., Ivanov, V. et al. Importance of hydraulic strategy trade-offs in structuring response of canopy trees to extreme drought in central Amazon. Oecologia 197, 13–24 (2021). https://doi.org/10.1007/s00442-021-04924-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-021-04924-9