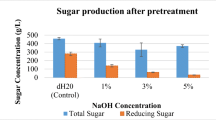
Abstract
The competitiveness of the second-generation bioethanol by biotechnological process requires an effective and quantitative control of biochemical reactions. In this study, the potential of isothermal calorimetry technique to measure heat and kinetics of a non-homogeneous substrate enzymatic hydrolysis is intended. Using this technique, optimum temperature of the enzymes used for lignocellulosic molecules hydrolysis was determined. Thus, the amount of substrate-to-enzyme ratio was highlighted as an important parameter of the hydrolysis yield. Furthermore, a new enzymes’ cocktail efficiency consisting of a mix of cellulases and cellobiose dehydrogenase (CDH) was qualified by this technique. The results showed that this cocktail allowed the production of a high amount of gluconic acid that could improve the attractiveness of these second-generation biofuels. From the set of experiments, the hydrolysis heat of wheat straw was derived and a meaningful value of −32.2 ± 3.2 J g−1 (gram reducing sugars product) is calculated. Then, isothermal measurements were used to determine kinetic constants of the cellulases and CDH mix on wheat straw. Results showed that this enzyme cocktail has an optimal rate at 45 °C in the range of temperatures tested (40–55 °C).







Similar content being viewed by others
Notes
Genencor-Danisco, Rochester, NY, USA.
Novozyme, Franklinton, NC, USA.
Abbreviations
- CDH:
-
Cellobiose dehydrogenase
- K eq :
-
Equilibrium constant
- ∆H :
-
Enthalpy
- w :
-
Weight
- Km :
-
Michaelis–Menten constant
- V M :
-
Maximum reaction rate
- Q :
-
Exchanged heat
- V :
-
Volume
- r :
-
Reaction rate
- [S]:
-
Substrate concentration at time t
- [S]0 :
-
Substrate concentration at time 0
- t :
-
Time
References
Han M, Moon S-K, Choi G-W (2014) Pretreatment solution recycling and high-concentration output for economical production of bioethanol. Bioprocess Biosyst Eng 37(11):2205–2213
Sun Y, Cheng J (2001) Hydrolysis of lignocellulosic materials for ethanol production: a review. Bioresour Technol 83(1):1–11
Alvira P, Tomás-Pejó E, Ballesteros M, Negro MJ (2010) Pretreatment technologies for an efficient bioethanol production process based on enzymatic hydrolysis: a review. Bioresour Technol 101(13):4851–4861
Ahn DJ, Kim SK, Yun HS (2011) Optimization of pretreatment and saccharification for the production of bioethanol from water hyacinth by Saccharomyces cerevisiae. Bioprocess Biosyst Eng 35(1–2):35–41
Cook C, Devoto A (2011) Fuel from plant cell walls: recent developments in second generation bioethanol research. J Sci Food Agric 91(10):1729–1732
Limayem A, Ricke SC (2012) Lignocellulosic biomass for bioethanol production: current perspectives, potential issues and future prospects. Prog Energy Combust Sci 38(4):449–467
Tengerdy RP, Szakacs G (2003) Bioconversion of lignocellulose in solid substrate fermentation. Biochem Eng J 13(2–3):169–179
Wang L, Sharifzadeh M, Templer R, Murphy RJ (2013) Bioethanol production from various waste papers: economic feasibility and sensitivity analysis. Appl Energy 111:1172–1182
Ergün BG, Çalık P (2015) Lignocellulose degrading extremozymes produced by Pichia pastoris: current status and future prospects. Bioprocess Biosyst Eng 39(1):1–36
Wiman M, Dienes D, Hansen MAT, van der Meulen T, Zacchi G, Lidén G (2012) Cellulose accessibility determines the rate of enzymatic hydrolysis of steam-pretreated spruce. Bioresour Technol 126:208–215
García JC, Díaz MJ, Garcia MT, Feria MJ, Gómez DM, López F (2013) Search for optimum conditions of wheat straw hemicelluloses cold alkaline extraction process. Biochem Eng J 71:127–133
Zhu Z, Zhu M, Wu Z (2012) Pretreatment of sugarcane bagasse with NH4OH–H2O2 and ionic liquid for efficient hydrolysis and bioethanol production. Bioresour Technol 119:199–207
Bianconi ML (2007) Calorimetry of enzyme-catalyzed reactions. Biophys Chem 126(1–3):59–64
Baranauskienė L, Petrikaitė V, Matulienė J, Matulis D (2009) Titration calorimetry standards and the precision of isothermal titration calorimetry data. Int J Mol Sci 10(6):2752–2762
Soric A, Ferrasse J-H, Roche N (2011) Microcalorimetric qualitative analysis of biofilm development in porous media used in wastewater treatment by constructed wetland. J Therm Anal Calorim 104(1):113–118
Karthikeyan R, Surianarayanan M, Sudharshan S, Gunasekaran P, Asit Baran M (2011) Biocalorimetric and respirometric studies on production of Penicillin G acylase from Bacillus badius pac in E. coli DH5α. Biochem Eng J 55(3):223–229
James AM (2013) Thermal and energetic studies of cellular biological systems. Butterworth-Heinemann
Sivaprakasam S, Mahadevan S, Sekar S (2008) Calorimetric on-line monitoring of proteolytic activity of P. aeruginosa cultivated in a bench-scale biocalorimeter. Biochem Eng J 39(1):149–156
Braissant O, Wirz D, Göpfert B, Daniels AU (2010) Biomedical use of isothermal microcalorimeters. Sensors 10(10):9369–9383
Braissant O, Bonkat G, Wirz D, Bachmann A (2013) Microbial growth and isothermal microcalorimetry: growth models and their application to microcalorimetric data. Thermochim Acta 555:64–71
Hansen LD, Christensen JJ, Izatt RM (1965) Entropy titration. A calorimetric method for the determination of ΔG°(K), ΔH° and ΔS°. Chem Commun Lond 3:36–38
Underdown DR, Yun Sock Sung, Bear JL (1974) The thermodynamics of nickel malonate formation in water–dioxane mixtures. J Inorg Nucl Chem 36(9):2043–2046
Baumann MJ, Murphy L, Lei N, Krogh KBRM, Borch K, Westh P (2011) Advantages of isothermal titration calorimetry for xylanase kinetics in comparison to chemical-reducing-end assays. Anal Biochem 410(1):19–26
Lonhienne T, Gerday C, Feller G (2000) Psychrophilic enzymes: revisiting the thermodynamic parameters of activation may explain local flexibility. Biochim Biophys Acta 1543(1):1–10
Siddiqui KS, Ertan H, Charlton T, Poljak A, Daud Khaled AK, Yang X, Marshall G, Cavicchioli R (2014) Versatile peroxidase degradation of humic substances: use of isothermal titration calorimetry to assess kinetics, and applications to industrial wastes. J Biotechnol 178:1–11
Murphy L, Borch K, McFarland KC, Bohlin C, Westh P (2010) A calorimetric assay for enzymatic saccharification of biomass. Enzyme Microb Technol 46(2):141–146
Bey M, Berrin J-G, Poidevin L, Sigoillot J-C (2011) Heterologous expression of Pycnoporus cinnabarinus cellobiose dehydrogenase in Pichia pastoris and involvement in saccharification processes. Microb Cell Factor 10(1):113
Navarro D, Couturier M, da Silva GGD, Berrin J-G, Rouau X, Asther M, Bignon C (2010) Automated assay for screening the enzymatic release of reducing sugars from micronized biomass. Microb Cell Factor 9:58
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31(3):426–428
Forsberg Z, Vaaje-Kolstad G, Westereng G, Bunæs AC, Stenstrøm Y, MacKenzie A, Sørlie M, Horn SJ, Eijsink VGH (2011) Cleavage of cellulose by a CBM33 protein. Protein Sci 20:1479–1483
Larson D (2015) Clinical chemistry: fundamentals and laboratory techniques. Elsevier, Amsterdam
Todd MJ, Gomez J (2001) Enzyme kinetics determined using calorimetry: a general assay for enzyme activity? Anal Biochem 296(2):179–187
Tewari YB, Goldberg RN (1989) Thermodynamics of hydrolysis of disaccharides. Cellobiose, gentiobiose, isomaltose, and maltose. J Biol Chem 264(7):3966–3971
Jeoh T, Baker JO, Ali MK, Himmel ME, Adney WS (2005) β-d-Glucosidase reaction kinetics from isothermal titration microcalorimetry. Anal Biochem 347(2):244–253
Illanes A (2008) Enzyme biocatalysis: principles and applications. Springer, Berlin
Gautam SP, Bundela PS, Pandey AK, Khan J, Awasthi MK, Sarsaiya S (2011) Optimization for the production of cellulase enzyme from municipal solid waste residue by two novel cellulolytic fungi. Biotechnol Res Int 2011:810425
Robert D, Vian B (2004) Éléments de biologie cellulaire. Doin
Acknowledgements
This research was funded by the Council of the PACA Region in the frame of MOTBIO project. The authors would like to thank the Research Federation ECCOREV for partially financing this work. The authors are grateful to Simeng ZHOU from Biotechnology of Filamentous Fungi laboratory—Aix Marseille Université, INRA, UMR 1163, for biochemical analyses.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tafoukt, D., Soric, A., Sigoillot, JC. et al. Determination of kinetics and heat of hydrolysis for non-homogenous substrate by isothermal calorimetry. Bioprocess Biosyst Eng 40, 643–650 (2017). https://doi.org/10.1007/s00449-016-1728-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-016-1728-0