Abstract
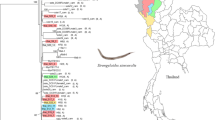
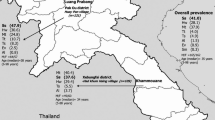
The parasitic nematodes, Strongyloides stercoralis and Strongyloides fuelleborni, can infect humans and non-human primates. We amplified and sequenced a portion of the 18S ribosomal RNA gene (rRNA) and of the mitochondrial cytochrome c oxidase subunit 1 (cox1) gene of Strongyloides from humans in the study area in Thailand, where people have frequent contact with long-tailed macaques. Fresh stool samples were obtained from 213 people and were examined using the agar plate culture method. The overall prevalence of Strongyloides infection was 8.92% (19/213). From a total of 19 worms (one per infected person), 18 adult males had 18S rRNA sequences identical with that of S. stercoralis and one adult female had a sequence almost identical with that of S. fuelleborni. A median-joining network of cox1 sequences revealed nine new haplotypes from S. stercoralis, and an overall haplotype diversity (Hd) of 0.9309. The single haplotype of S. fuelleborni was also new and contributed to an overall haplotype diversity for that species of 0.9842. This is the first molecular identification of S. stercoralis and S. fuelleborni in a human community having contact with long-tailed macaques in Thailand. It is also the first report of S. fuelleborni infecting a human in Thailand.


Similar content being viewed by others
References
Arakaki T, Iwanaga M, Kinjo F, Saito A, Asato R, Ikeshiro T (1990) Efficacy of agar-plate culture in detection of Strongyloides stercoralis infection. J Parasitol 76:425–428
Ashford RW, Barnish G, Viney ME (1992) Strongyloides fuelleborni kellyi: infection and disease in Papua New Guinea. Parasitol Today 8:314–318
Beknazarova M, Whiley H, Ross K (2016) Strongyloidiasis: a disease of socioeconomic disadvantage. Int J Environ Res Public Health 13(5). doi:10.3390/ijerph13050517
Chunlertrith K, Noiprasit A, Kularbkaew C, Sanpool O, Maleewong W, Intapan PM (2015) A complicated case of strongyloidiasis presenting with intestinal lymphadenopathy obstruction: molecular identification. Southeast Asian J Trop Med Public Health 46:1–7
Fisher D, McCarry F, Currie B (1993) Strongyloidiasis in the Northern Territory. Under-recognised and under-treated? Med J Aust 159:88–90
Genta R (1989) Strongyloides stercoralis: loss of ability to disseminate after repeated passage in laboratory beagles. Trans R Soc Trop Med Hyg 83:539–541
Gilles HM, Cook GC (1996) Soil-transmitted helminthes (Geohelminths). In: Cook GC (ed) Manson’s tropical disease. W. B. Saunders, London, pp 1369–1412
Grove DI (1989) Clinical manifestations. In: Grove DI (ed) Strongyloidiasis—a major roundworm infection of man. Taylor & Francis, London, pp 155–173
Grove DI (1996) Human strongyloidiasis. Adv Parasitol 38:251–309
Grove DI, Northern C (1982) Infection and immunity in dogs infected with a human strain of Strongyloides stercoralis. Trans R Soc Trop Med Hyg 76:833–838
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucl Acids Symp 41:95–98
Hasegawa H, Hayashida S, Ikeda Y, Sato H (2009) Hyper-variable regions in 18S rDNA of Strongyloides spp. as markers for species-specific diagnosis. Parasitol Res 104:869–874. doi:10.1007/s00436-008-1269-9
Hasegawa H, Sato H, Fujita S, Nguema PP, Nobusue K, Miyagi K, Kooriyama T, Takenoshita Y, Noda S, Sato A, Morimoto A, Ikeda Y, Nishida T (2010) Molecular identification of the causative agent of human strongyloidiasis acquired in Tanzania: dispersal and diversity of Strongyloides spp. and their hosts. Parasitol Int 59:407–413. doi:10.1016/j.parint.2010.05.007
Hasegawa H, Kalousova B, McLennan MR, Modry D, Profousova-Psenkova I, Shutt-Phillips KA, Todd A, Huffman MA, Petrzelkova KJ (2016) Strongyloides infections of humans and great apes in Dzanga-Sangha Protected Areas, Central African Republic and in degraded forest fragments in Bulindi, Uganda. Parasitol Int 65:367–370. doi:10.1016/j.parint.2016.05.004
Labes EM, Nurcahyo W, Deplazes P, Mathis A (2011) Genetic characterization of Strongyloides spp. from captive, semi-captive and wild Bornean orangutans (Pongo pygmaeus) in Central and East Kalimantan, Borneo, Indonesia. Parasitology 138:1417–1422. doi:10.1017/S0031182011001284
Laymanivong S, Hangvanthong B, Insisiengmay B, Vanisaveth V, Laxachack P, Jongthawin J, Sanpool O, Thanchomnang T, Sadaow L, Phosuk I, Rodpai R, Maleewong W, Intapan PM (2016) First molecular identification and report of genetic diversity of Strongyloides stercoralis, a current major soil-transmitted helminth in humans from Lao People’s Democratic Republic. Parasitol Res 115:2973–2980. doi:10.1007/s00436-016-5052-z
Librado P, Rozas J (2009) DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25:1451–1452. doi:10.1093/bioinformatics/btp187
Little MD (1966) Comparative morphology of six species of Strongyloides (Nematoda) and redefinition of the genus. J Parasitol 52:69–84
Nolan TJ, Genta R, Schad GA (1998) Strongyloidosis. In: Palmer SR, Soulsby L, Simpson DIH (eds) Zoonoses. Oxford University Press, Oxford, pp 747–758
Olsen A, van Lieshout L, Marti H, Polderman T, Polman K, Steinmann P, Stothard R, Thybo S, Verweij JJ, Magnussen P (2009) Strongyloidiasis—the most neglected of the neglected tropical diseases? Trans R Soc Trop Med Hyg 103:967–972. doi:10.1016/j.trstmh.2009.02.013
Pampiglione S, Ricciardi ML (1971) The presence of Strongyloides fülleborni von Linstow, 1905, in man in Central and East Africa. Parassitologia 13:257–269
Puthiyakunnon S, Boddu S, Li Y, Zhou X, Wang C, Li J, Chen X (2014) Strongyloidiasis—an insight into its global prevalence and management. PLoS Negl Trop Dis 8:e3018. doi:10.1371/journal.pntd.0003018.eCollection
Siddiqui AA, Berk SL (2001) Diagnosis of Strongyloides stercoralis infection. Clin Infect Dis 33:1040–1047
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729. doi:10.1093/molbev/mst197
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTALW: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Viney ME (2006) The biology and genomics of Strongyloides. Med Microbiol Immunol 195:49–54
Viney ME, Lok JB (2007) Strongyloides spp. WormBook 23:1–15. doi:10.1895/wormbook.1.141.1
Wenz-Mücke A, Sithithaworn P, Petney TN, Taraschewski H (2013) Human contact influences the foraging behaviour and parasite community in long-tailed macaques. Parasitology 140:709–718. doi:10.1017/S003118201200203X
Acknowledgements
We would like to thank David Blair for valuable suggestions and assistance with the presentation of this paper through Khon Kaen University Publication Clinic.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was supported by a TRF Senior Research Scholar Grant, Thailand Research Fund (TT, PMI, and WM, grant no. RTA5880001); the Higher Education Research Promotion and National Research University Project of Thailand, Office of the Higher Education Commission, Thailand, through the Health Cluster (SHeP-GMS) (WM); the Faculty of Medicine, Khon Kaen University (WM and PMI, grant no. TR57201); Scholarship under Doctoral Training Program from Graduate School Research Affairs and Khon Kaen University (OS, grant no. 58101) and the Faculty of Medicine, Mahasarakham University (TT).
Ethical approval
The study protocol was approved by the Khon Kaen University Ethics Committee for Human Research (HE571286). Each participant was informed of study methods, risks, and benefits of the process. Before enrolment, written consent was obtained from all adult participants and from parents or legal guardians of minors.
Conflict of interests
The authors declare that they have no conflicts of interest.
Electronic supplementary material
ESM 1
(DOC 329 kb)
Rights and permissions
About this article
Cite this article
Thanchomnang, T., Intapan, P.M., Sanpool, O. et al. First molecular identification and genetic diversity of Strongyloides stercoralis and Strongyloides fuelleborni in human communities having contact with long-tailed macaques in Thailand. Parasitol Res 116, 1917–1923 (2017). https://doi.org/10.1007/s00436-017-5469-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-017-5469-z