Abstract
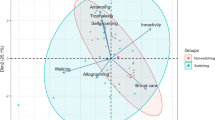
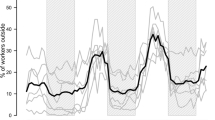
Key structures of insects, such as their mandibles, become worn by the use and this leads to performance constraints, increase of metabolic costs, and decrease of individual’ both lifespan and survival. Studying this phenomenon is interesting in species that build nests as it demands a significant amount of mandible labor; however, the effect of this selective factor is less obvious in species like social vespids that perform different tasks at different ages. By comparing mandible wear between individuals of different ages in 18 colonies of 13 species of neotropical social wasps (Vespidae: Polistinae), we aimed to understand (1) the distribution of mandible wear across ages, and (2) the association between colony size and the coefficient of variation of mandible wear as an indicator of specialization. Despite a general positive trend between mandible wear and age, there are numerous exceptions and no age seems to concentrate this phenomenon. Mandible wear variation was evenly distributed throughout the worker ages, and was not correlated with colony size. Our study suggests that individuals distribute mandible demanding tasks throughout their life to reduce structural attrition which may improve worker’s general output. We proposed that, to extend both life expectancy and general performance of the workers, wasps may spread the more demanding tasks over longer and less intense periods.





Similar content being viewed by others
Availability of data and material
(raw mandible wear data) Dryad https://doi.org/10.5061/dryad.6q573n5x6, and (CV, colony data and mandible wear) Dryad https://doi.org/10.5061/dryad.ffbg79csk.
References
Acosta FJ, Morales MA, Zorrilla JM (1983) Polimorfismo y desgaste en la pieza mandibular de Messor barbarus (Hym., Formicidae). Graellsia 38:167–173
Alboukadel K (2020) ggpubr: 'ggplot2' Based Publication Ready Plots. R package version 0.4.0. https://CRAN.R-project.org/package=ggpubr
Bernays EA (1991) Evolution of insect morphology in relation to plants. Philos Trans R Soc Lond 333:257–264. https://doi.org/10.1098/rstb.1991.0075
Bernays EA, Funk DJ (1999) Specialists make faster decisions than generalists: experiments with aphids. Proc R Soc B Biol Sci 266:151–156. https://doi.org/10.1098/rspb.1999.0615
Chapman RF (1957) The structure and wear of mandibles in some African grasshoppers. Proc Zool Soc Lond 142:107–122
Chittka L, Muller H (2009) Learning, specialisation, efficiency and task allocation in social insects. Commun Integr Biol 2(2):151–154. https://doi.org/10.4161/cib.7600
Clavel J, Escarguel G, Merceron G (2015) mvMORPH: an R package for fitting multivariate evolutionary models to morphometric data. Methods Ecol Evol 6:1311–1319
De Verges J, Nehring V (2016) A critical look at proximate causes of social insect senescence: damage accumulation or hyperfunction? Curr Opin Insect Sci 16:69–75. https://doi.org/10.1016/j.cois.2016.05.003
Dornhaus A, Holley JA, Franks NR (2009) Larger colonies do not have more specialized workers in the ant Temnothorax albipennis. Behav Ecol 20:922–929. https://doi.org/10.1093/beheco/arp070
Dornhaus A, Powell S, Bengston S (2012) Group size and its effects on collective organization. Annu Rev Entomol 57:123–141. https://doi.org/10.1146/annurev-ento-120710-100604
Du H, Chouvenc T, Su NY (2017) Development of age polyethism with colony maturity in Coptotermes formosanus (Isoptera: Rhinotermitidae). Environ Entomol 46:311–318. https://doi.org/10.1093/ee/nvw162
Fjerdingstad EJ, Crozier RH (2006) The evolution of worker caste diversity in social insects. Am Nat 167:390–400. https://doi.org/10.1086/499545
Flores-Prado L, Pinto CF, Rojas A, Fontúrbel FE (2014) Strong selection on mandible and nest features in a carpenter bee that nests in two sympatric host plants. Ecol Evol 4:1820–1827. https://doi.org/10.1002/ece3.995
Foster DJ, Cartar RV (2011) What causes wing wear in foraging bumble bees? J Exp Biol 214:1896–1901. https://doi.org/10.1242/jeb.051730
Garrett RW, Carlson KA, Goggans MS et al (2016) Leaf processing behaviour in Atta leafcutter ants: 90% of leaf cutting takes place inside the nest, and ants select pieces that require less cutting. R Soc Open Sci. https://doi.org/10.1098/rsos.150111
Gautrais J, Theraulaz G, Deneubourg JL, Anderson C (2002) Emergent polyethism as a consequence of increased colony size in insect societies. J Theor Biol 215:363–373. https://doi.org/10.1006/jtbi.2001.2506
Giraldo YM, Traniello JFA (2014) Worker senescence and the sociobiology of aging in ants. Behav Ecol Sociobiol 68:1901–1919
Gordon DM (2013) The rewards of restraint in the collective regulation of foraging by harvester ant colonies. Nature 498:91–93. https://doi.org/10.1038/nature12137
Gordon DM (2019) The ecology of collective behavior. Annu Rev Entomol 64:35–50. https://doi.org/10.1371/journal.pbio.1001805
Gordon DM, Chu J, Lillie A et al (2005) Variation in the transition from inside to outside work in the red harvester ant Pogonomyrmex barbatus. Insectes Soc 52:212–217. https://doi.org/10.1007/s00040-004-0796-3
Grafen A (1989) The phylogenetic regression. Philos Trans R Soc Lon B Biol Sci 326:119–157. https://doi.org/10.1098/rstb.1989.0106
Harmon LJ, Weir JT, Brock CD et al (2008) GEIGER: investigating evolutionary radiations. Bioinformatics 24:129–131. https://doi.org/10.1093/bioinformatics/btm538
Hayes EJ, Wall R (1999) Age-grading adult insects: a review of techniques. Physiol Entomol 24:1–10. https://doi.org/10.1046/j.1365-3032.1999.00104.x
Holbrook CT, Barden PM, Fewell JH (2011) Division of labor increases with colony size in the harvester ant Pogonomyrmex californicus. Behav Ecol 22:960–966. https://doi.org/10.1093/beheco/arr075
Hölldobler B, Wilson EO (1990) The ants. Harvard University Press, USA, p 732
Jeanne RL (1986a) The organization of work in Polybia occidentalis: costs and benefits of specialization in a social wasp. Behav Ecol Sociobiol 19:333–341. https://doi.org/10.1007/BF00295706
Jeanne RL (1986b) The evolution of the organization of work in social insects. Monit Zool Ital 20:119–133. https://doi.org/10.1080/00269786.1971.10736166
Jeanne RL (1991a) Polyethism. In: Ross KG, Matthews RW (eds) The social biology of wasps. Cornell University Press, Ithaca, pp 389–425
Jeanne RL (1991b) The Swarm-founding Polistinae. In: Ross KG, Matthews RW (eds) The social biology of wasps. Cornell University Press, Ithaca, pp 191–231
Jeanson R, Fewell JH, Gorelick R, Bertram SM (2007) Emergence of increased division of labor as a function of group size. Behav Ecol Sociobiol 62:289–298. https://doi.org/10.1007/s00265-007-0464-5
JrT G, Dickerman AW, Janis CM, Jones JA (1993) Phylogenetic analysis of covariance by computer simulation. Syst Biol 42:265–292
Kim B, Kim KW, Choe JC (2012) Temporal polyethism in korean yellowjacket foragers, Vespula koreensis (Hymenoptera, Vespidae). Insectes Soc 59:263–268. https://doi.org/10.1007/s00040-011-0212-8
Köhler G, Jentzsch A, Reinhardt K (2000) Age related mandible abrasion in three species of short-horned grasshoppers (Caelifera: Acrididae). J Orthoptera Res 9:81–87. https://doi.org/10.2307/3503638
Kuřavová K, Hajduková L, Kočárek P (2014) Age-related mandible abrasion in the groundhopper Tetrix tenuicornis (Tetrigidae, Orthoptera). Arthropod Struct Dev 43:187–192. https://doi.org/10.1016/j.asd.2014.02.002
Leighton GM, Charbonneau D, Dornhaus A (2017) Task switching is associated with temporal delays in Temnothorax rugatulus ants. Behav Ecol 28:319–327. https://doi.org/10.1093/beheco/arw162
Massey FP, Hartley SE (2009) Physical defences wear you down: progressive and irreversible impacts of silica on insect herbivores. J Anim Ecol 78:281–291. https://doi.org/10.1111/j.1365-2656.2008.01472.x
Menezes RST, Lloyd MW, Brady SG (2020) Phylogenomics indicates Amazonia as the major source of Neotropical swarm-founding social wasp diversity. Proceedings Biol Sci 287:20200480. https://doi.org/10.1098/rspb.2020.0480
Molina-Venegas R, Rodríguez MA (2017) Revisiting phylogenetic signal; strong or negligible impacts of polytomies and branch length information? BMC Evol Biol 17:1–10. https://doi.org/10.1186/s12862-017-0898-y
Münkemüller T, Lavergne S, Bzeznik B et al (2012) How to measure and test phylogenetic signal. Methods Ecol Evol 3:743–756. https://doi.org/10.1111/j.2041-210X.2012.00196.x
Nalepa CA (2012) Wing wear is a poor estimate of age in Cerceris fumipennis (Hymenoptera, Crabronidae). J Hymenopt Res 24:43–46. https://doi.org/10.3897/JHR.24.2091
Naug D, Gadagkar R (1998) The role of age in temporal polyethism in a primitively eusocial wasp. Behav Ecol Sociobiol 42:37–47. https://doi.org/10.1007/s002650050409
O’Donnell S, Jeanne RL (1992a) The effects of colony characteristics on life span and foraging behavior of individual wasps (Polybia occidentalis, Hymenoptera: Vespidae). Insectes Soc 39:73–80. https://doi.org/10.1007/BF01240532
O’Donnell S, Jeanne RL (1992b) Lifelong patterns of forager behaviour in a tropical swarm-founding wasp: effects of specialization and activity level on longevity. Anim Behav 44:1021–1027. https://doi.org/10.1016/S0003-3472(05)80314-8
O’Donnell S, Jeanne RL (1995) Implications of senescence patterns for the evolution of age polyethism in eusocial insects. Behav Ecol 6:269–273. https://doi.org/10.1093/beheco/6.3.269
Pacala SW, Gordon DM, Godfray HCJ (1996) Effects of social group size on information transfer and task allocation. Evol Ecol 10:127–165. https://doi.org/10.1007/BF01241782
Pagel M (1999) Inferring the historical patterns of biological evolution. Nature 401:877–884
Paradis E, Schliep K (2019) Ape 5.0: an environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 35:526–528. https://doi.org/10.1093/bioinformatics/bty633
Park SH, Doege SJ, Nakata PA, Korth KL (2009) Medicago truncatula-derived calcium oxalate crystals have a negative impact on chewing insect performance via their physical properties. Entomol Exp Appl 131:208–215. https://doi.org/10.1111/j.1570-7458.2009.00846.x
Paterno GB, Penone C, Werner GDA (2018) sensiPhy: an R-package for sensitivity analysis in phylogenetic comparative methods. Methods Ecol Evol 9:1461–1467. https://doi.org/10.1111/2041-210X.12990
Piekarski PK, Carpenter JM, Lemmon AR et al (2018) Phylogenomic evidence overturns current conceptions of social evolution in wasps (Vespidae). Mol Biol Evol 35:2097–2109. https://doi.org/10.1093/molbev/msy124
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Core Team (2021) nlme: linear and nonlinear mixed effects models. R package version 3.1-152, https://CRAN.R-project.org/package=nlme
Polidori C, Federici M, Papadia C, Andrietti F (2006) Nest sharing and provisioning activity of females of the digger wasp, Cerceris rubida (Hymenoptera, Crabronidae). Ital J Zool 73:55–65. https://doi.org/10.1080/11250000500502079
Purvis A, Gittleman JL, Luh HK (1994) Truth or consequences: effects of phylogenetic accuracy on two comparative methods. J Theor Biol 167:293–300. https://doi.org/10.1006/jtbi.1994.1071
Quicke DLJ, Wyeth P, Fawke JD et al (1998) Manganese and zinc in the ovipositors and mandibles of hymenopterous insects. Zool J Linn Soc 124:387–396. https://doi.org/10.1111/j.1096-3642.1998.tb00583.x
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Rana JS, Dixon AFG, Jarošík V (2002) Costs and benefits of prey specialization in a generalist insect predator. J Anim Ecol 71:15–22. https://doi.org/10.1046/j.0021-8790.2001.00574.x
Revell LJ (2012) phytools: an R package for phylogenetic comparative biology (and other things). Methods Ecol Evol 3:217–223. https://doi.org/10.1111/j.2041-210X.2011.00169.x
Roitberg BD, Gillespie DR, Quiring DMJ et al (2005) The cost of being an omnivore: mandible wear from plant feeding in a true bug. Naturwissenschaften. https://doi.org/10.1007/s00114-005-0013-x
Santoro D, Hartley S, Lester PJ (2019) Behaviourally specialized foragers are less efficient and live shorter lives than generalists in wasp colonies. Sci Rep 9:1–10. https://doi.org/10.1038/s41598-019-41791-0
Sarmiento CE (2004) A test of adaptive hypotheses: mandibular traits, nest construction materials, and feeding habits in neotropical social wasps (Vespidae, Polistinae). Insectes Soc 51:387–391. https://doi.org/10.1007/s00040-004-0756-y
Schaber BD, Kokko EG, Entz T, Richards KW (1993) The effect of spring burning of seed alfalfa fields on leaf characteristics and on mandibular wear of female leafcutter bees [Megachile rotundata (F.)] (Hymenoptera: Megachilidae). Can Entomol 125:881–886. https://doi.org/10.4039/Ent125881-5
Schofield RMS, Emmett KD, Niedbala JC, Nesson MH (2011) Leaf-cutter ants with worn mandibles cut half as fast, spend twice the energy, and tend to carry instead of cut. Behav Ecol Sociobiol 65:969–982. https://doi.org/10.1007/s00265-010-1098-6
Schofield RMS, Choi S, Coon JJ et al (2016) Is fracture a bigger problem for smaller animals? Force and fracture scaling for a simple model of cutting, puncture and crushing. Interface Focus. https://doi.org/10.1098/rsfs.2016.0002
Seeley TD (1982) Adaptive significance of the age polyethism schedule in honeybee colonies. Behav Ecol Sociobiol 11:287–293. https://doi.org/10.1007/BF00299306
Silveira OT, dos Santos JNA (2011) Comparative morphology of the mandibles of female polistine social wasps (Hymenoptera, Vespidae, Polistinae). Rev Bras Entomol 55:479–500. https://doi.org/10.1590/S0085-56262011000400004
Tofilski A (2002) Influence of age polyethism on longevity of workers in social insects. Behav Ecol Sociobiol 51:234–237. https://doi.org/10.1007/s00265-001-0429-z
Toth AL, Sumner S, Jeanne RL (2016) Patterns of longevity across a sociality gradient in vespid wasps. Curr Opin Insect Sci 16:28–35. https://doi.org/10.1016/j.cois.2016.05.006
Wenzel JW, Carpenter JM (1994) Comparing methods: adaptive traits and tests of adaptation. In: Eggleton P, Vane-Wright RI (eds) Phylogenetics and ecology. Academic Press, New York, pp 79–101
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer, New York
Acknowledgements
We thank Diana Maritza Monroy for her help during the first phase of the research as well as Joan Sebastian Joya for his help in nests collection, R.B. Jeanne for insightful comments on the early versions of the manuscript, P.K. Piekarski and R. Menezes for sharing their molecular phylogenies, Cristian Román-Palacios for his suggestions, members of the Laboratorio de Sistemática y Biología Comparada de Insectos for their comments on the research, volunteers who graded tooth wear, and the Laboratorio de Microscopía Electrónica de la Universidad Nacional for technical support. Many thanks to the Universidad Nacional de Colombia that provided continuous support for this research through Hermes project code 16632. Dr. Th. Defler reviewed the writing. We thank the referees and editors for their insightful comments.
Funding
This research was partially supported by the Universidad Nacional de Colombia, Hermes project code 16632.
Author information
Authors and Affiliations
Contributions
JJL-O: methodology (equal); data curation (leading); formal analysis (equal); writing-original draft (equal); writing-review and editing (equal).CES: conceptualization (leading); methodology (equal); data curation (equal); formal analysis (equal); writing-original draft (equal); writing-review and editing (equal).
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
Collection of specimens follow national guidelines and permits.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lagos-Oviedo, J.J., Sarmiento, C.E. Task specialization and structure attrition: neotropical social wasps may disperse the cost of mandible demanding labors throughout their lives. Zoomorphology 140, 269–278 (2021). https://doi.org/10.1007/s00435-021-00527-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00435-021-00527-3