Abstract
Purpose
The objective of this study was to investigate non-cancer causes of death and associated risk factors after bladder cancer (BC) diagnosis.
Methods
Eligible BC patients were obtained from the SEER database. SEER*Stat software 8.3.9.2 was used to calculate the standardized mortality ratios (SMRs). The proportions of different non-cancer cause of death were calculated and analyzed in different follow-up periods. Multivariate competing risk model was used to analyze the risk factors for death of BC and non-cancer diseases.
Results
In total, 240,954 BC patients were included and 106,092 patients experienced death, with 37,205 (35.07%), 13,208 (12.45%) and 55,679 (52.48%) patients experienced BC, other cancer and non-cancer disease-related deaths, respectively. Overall SMR for BC patients who died from non-cancer diseases was 2.42 (95% CI [2.40–2.44]). Cardiovascular diseases were the most common non-cancer cause of death, followed by respiratory diseases, diabetes mellitus, and infectious diseases. Multivariate competing risk analysis identified the following high-risk factors for non-cancer mortality: age > 60 years, male, whites, in situ stage, pathological type of transitional cell carcinoma, not receiving treatment (including surgery, chemotherapy, or radiation), and widowed.
Conclusions
Cardiovascular diseases are the leading non-cancer cause of death in BC patients, followed by respiratory disease, diabetes mellitus and infectious diseases. Physicians should pay attention to the risk of death from these non-cancer diseases. Also, physicians should encourage patients to engage in more proactive self-surveillance and follow up.
Similar content being viewed by others

Avoid common mistakes on your manuscript.
Introduction
Bladder cancer (BC) is estimated to account for more than 500,000 new cases and 200,000 deaths per year worldwide. In the United States alone, there are more than 80,000 new cases and 17,000 deaths each year (Richters et al. 2020; Lenis et al. 2020; Siegel et al. 2019, 2020).
With the updating of various anti-cancer treatment strategies, cancer-related mortality has gradually decreased (Kochanek et al. 2019a; Robertson et al. 2018; Jalanko et al. 2020). As a result, non-cancer causes of death are of increasing concern as survival times increase. Studies have shown that non-cancer diseases such as heart disease are the leading cause of non-cancer mortality in cancer patients. In most cancer patients, the mortality rate from non-cancer diseases even exceeds that from primary cancer (Siegel et al. 2021; Abdel-Rahman 2017; Zhang et al. 2021; Sturgeon et al. 2019).
With the increasing quest for longer survival and higher quality of life, deaths due to non-cancer diseases should be taken into account. Studying non-cancer causes of death allows us to more accurately evaluate the risk of death in patients with cancer. As a result, we can provide early intervention and management. The focus of this study was on standardized mortality ratios (SMRs) for non-cancer causes of death, and on high-risk factors that can contributed to experiencing non-cancer mortality in BC patients.
Materials and methods
Data source
Patients information was obtained from the Surveillance, Epidemiology, and End Results (SEER) database, which includes approximately 28% of the general US population, and used the SEER*Stat software 8.3.9.2 to access the database: Incidence—SEER Research Plus Data, 18 Registries (excl AK), Nov 2020 Sub (2000–2018) for SMRs.
Patients
We included patients who were pathologically diagnosed with BC between 2000 and 2017. Cases diagnosed by death certificate and autopsy were excluded. Cases with incomplete information were also excluded.
Standardized mortality ratio (SMR)
For patients with BC in the SEER database, we calculated the number of deaths under different variables. The focus of this study was on non-cancer mortality in patients with BC. We classify non-cancer diseases into the following six categories: infectious diseases, diabetes mellitus, cardiovascular diseases, respiratory diseases, digestive diseases, and other non-cancer diseases. Supplementary Table 1 shows the definitions of non-cancer causes of death and the ICD-10 codes for the diseases.
First, the SMRs were calculated for all causes of deaths (BC, other cancers, non-cancer diseases). We then calculated SMRs for non-cancer causes of death for BC patients at different follow-up period (< 1 year, 1–5 years, and > 5 years) after diagnosis, with the distribution of the population and the age (mean ± SD) by variables. SMR was defined as the observed-to-expected ratio; the observation population was defined as patients diagnosed with BC in the United States from 2000 to 2017, which was collected from the SEER database. The competing risk analysis using Fine-Gray model was performed to adjust for confounding effects of age, sex, race, summary stage, year of diagnosis, histologic type, treatment (surgery, chemotherapy, and radiation therapy), and marital status to evaluate risks for non-cancer diseases mortality and BC mortality, and to plot cumulative mortality curves.
Statistical analysis
All analyses were performed using SEER*Stat software (version 8.3.9.2), R 4.1.1 (R foundation for Statistical Computing, Vienna, Austria), and Microsoft Excel 2019 (Microsoft, Redmond, WA). All statistical analyses were two-sided with a p value < 0.05 being considered statistically significant.
Results
Baseline characteristics
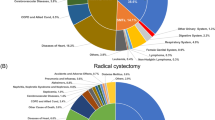
Cumulatively, 240,954 eligible BC patients were obtained from the SEER database, of which 106,092 experienced mortality. Among all the causes of death in BC patients, only the proportion of non-cancer diseases increased with increasing follow-up time (Fig. 1), from 30.06 to 78.97%. Supplementary Table 2 shows the SMR for all causes of death in BC patients after diagnosis, the overall SMR was 3.23 (95% CI [3.21–3.25]), with BC has the highest SMR (SMR [95% CI] 7.59 [7.52–7.67]), followed by other cancers (SMR [95% CI] 2.68 [2.63–2.72]), and non-cancer diseases (SMR [95% CI] 2.42 [2.40–2.44]). Of all death in BC patients, 55,679 patients died from non-cancer diseases with a mean age at death of 75.84 ± 8.71 years. 52,803 (94.83%) patients were older than 60 years at diagnosis, 42,901 (77.05%) were male patients and 51,269 (92.08%) were white. The highest proportion of deaths was observed during the > 5 years follow-up period (41.58%), followed by 1–5 years (40.74%) after diagnosis. Table 1 shows the baseline characteristics of patients with BC who experienced non-cancer mortality: the number of deaths at different follow-up periods (< 1 year, 1–5 years, and > 5 years), and the mean ± SD age at diagnosis. Figure 1 shows the proportion of each cause of death in BC patients at different follow-up period.
SMR after BC diagnosis
Figure 2A shows that SMR decreases with increasing follow-up time. At < 1 year follow-up period, the highest SMR was for BC, followed by other cancers, and then non-cancer diseases, while at 1–5 years follow-up period, the SMR for non-cancer diseases surpassed other cancers in second place. Figure 2B shows that SMR increases with increasing year of diagnosis, while BC consistently has the highest level of SMR.
Figure 3A shows that the SMR for the six non-cancer diseases decreased with increasing follow-up time, with digestive diseases consistently having the highest SMR, followed by infectious diseases and diabetes mellitus, while cardiovascular diseases consistently having the lowest SMR. Figure 3B shows the proportion of the six non-cancer diseases at different follow-up periods, with cardiovascular diseases consistently having the highest proportion, followed by other non-cancer diseases and respiratory diseases.
Table 2 shows the SMR for non–cancer cause of death in BC patients after diagnosis, the overall SMR was 2.42 (95% CI [2.40–2.44]). SMR was always the highest within one year after diagnosis. Table 3 shows the SMR for all non–cancer causes of death in BC patients after diagnosis, the highest SMR was observed in digestive diseases (SMR [95% CI] 4.95 [4.55–5.38]), followed by infectious diseases (SMR [95% CI] 3.33 [3.17–3.49]) and diabetes mellitus (SMR [95% CI] 3.11 [2.98–3.25]).
Non-cancer causes of death at different follow-up period
Throughout the follow-up period, 55,679 (52.48%) of 106,092 patients experiencing non-cancer cause of death at a mean age of 75.84 ± 8.71 years with an overall SMR of (SMR [95% CI]) 2.42 [2.40–2.44].
Within 5 years after BC diagnosis
9843 (17.68%) patients died within one year after diagnosis, the mean age was 77.67 ± 8.62 with the highest overall SMR of 31.07 (95% CI [30.46–31.69]) (Fig. 3B, Tables 1, 2). The most common non-cancer cause of death was cardiovascular diseases (SMR [95% CI] 29.28 [28.47–30.12]), and other non-cancer diseases (SMR [95% CI] 31.22 [30.06–32.41]) (Table 3). While within 1–5 years after diagnosis, 22,686 (40.74%) patients experiencing non-cancer cause of death at a mean age of 77.41 ± 8.33, the overall SMR was 4.12 (95% CI [4.07–4.18]) (Fig. 3B, Tables 1, 2), cardiovascular diseases remain the most common non-cancer cause of death with a SMR of 3.98 (95% CI [3.91–4.06]) (Table 3).
The risk of death from non-cancer cause within 5 years after BC diagnosis was significantly higher than in the general population, especially for digestive diseases, infectious diseases, and diabetes mellitus (Table 3).
Over 5 years after BC diagnosis
25,150 (45.17%) patients died from non-cancer diseases over 5 years follow-up, the mean age was 74.09 ± 8.68, with an overall SMR of 1.35 (95% CI [1.33–1.37]) (Fig. 3B, Tables 1, 2). Cardiovascular diseases remain the most common non-cancer cause of death with a SMR of 1.30 (95% CI [1.27–1.32]) (Table 3). While patients were at significantly higher risk of death from digestive diseases (SMR [95% CI] 2.34 [2.01–2.71]) and diabetes mellitus (SMR [95% CI] 1.74 [1.62–1.86]).
Non-cancer causes of death by different factors
Subgroup analysis of non-cancer causes of death by clinicopathological features at each follow-up period after BC diagnosis (Supplementary Tables 3–34). Some meaningful findings were as indicated below.
Gender
A total of 42,901 (77.05%) male patients and 12,778 (22.95%) female patients experienced non-cancer deaths, and their overall SMR was very similar (SMR [95% CI] 2.41 [2.38–2.43] vs 2.48 [2.44–2.52]). The highest proportion of deaths in male patients was observed between 1 and 5 years after BC diagnosis, and that of female patients occurred over 5 years after diagnosis. Within 1 year after diagnosis, female patients were more likely to develop infectious diseases (SMR [95% CI] 51.47 [43.38–60.63]) and diabetes mellitus (SMR [95% CI] 45.42 [36.02–56.53]) mortality, male patients were more likely to experience death from digestive diseases (SMR [95% CI] 59.19 [47.34–73.10]). And at 1–5 years after diagnosis, the causes of non-cancer mortality were similar for male and female patients, with digestive diseases being the first, followed by infectious diseases and diabetes mellitus. While over 5 years after diagnosis, male patients are at higher risk to develop digestive diseases mortality (SMR [95% CI] 2.40 [2.02–2.82]) (Supplementary Tables 5–6).
Race
A total of 51,269 (92.08%) white patients, 2673 (4.80%) black patients, and 1737 (3.12%) other races patients died from non-cancer diseases. The highest rate of death was observed within 1–5 years after diagnosis for blacks and more than 5 years after diagnosis for whites and other races. Whites had a higher risk of developing death from digestive diseases (SMR [95% CI] 4.91 [4.49–5.35]), while blacks had a lower risk of death from cardiovascular disease as well as other non-cancer diseases; patients of other races were at higher risk of experiencing death from infectious diseases, and diabetes mellitus. Moreover, blacks were at higher risk of experiencing death from infectious diseases within 1 year after diagnosis, and other races had a significantly higher risk of death from diabetes mellitus within 1–5 years after diagnosis. Over 5 years after BC diagnosis, blacks were at higher risk than the general population for experiencing death from infectious disease and diabetes mellitus (Supplementary Tables 7–9).
Treatment
Treatment is divided into surgery (TURBT: transurethral resection of bladder tumor; PC: partial cystectomy; RC: radical cystectomy), chemotherapy, and radiation therapy.
There were 2710 (4.87%) deaths in BC patients who did not receive surgical treatment, 49,655 (89.18%) deaths in patients treated with TURBT, 599 (1.08%) deaths in patients treated with PC, and 2715 (4.87%) deaths in patients treated with RC. The highest proportion of non-cancer deaths in BC patients without surgical treatment was observed 1–5 years after diagnosis (38.49%), and the highest proportion of deaths in patients who received surgical treatment all occurred 5 years after diagnosis. The risk of non-cancer causes of death was significantly higher in all BC patients compared to the general population, with the top three causes of non-cancer death being digestive disease, infectious disease and diabetes mellitus for patients who did not undergo surgery, patients who received TURBT or RC; while for patients treated with PC, the top three causes of non-cancer death were infectious disease, diabetes mellitus and cardiovascular disease. Within 1 year after diagnosis, patients treated with RC were more likely to experience non-cancer death (SMR [95% CI] 62.80 [58.39–67.44]). Although patients treated with TURBT had the lowest risk of non-cancer cause of death (SMR [95% CI] 2.36 [2.34–2.38]), it was still significantly higher than the general population (Supplementary Tables 13–16).
Patients receiving either chemotherapy (SMR [95% CI] 3.30 [3.22–3.38]) or radiation therapy (SMR [95% CI] 4.10 [3.93–4.27]) had a significantly higher risk of non-cancer deaths. Patients receiving chemotherapy appeared to be more likely to experience death from digestive (SMR [95% CI] 9.41 [7.46–11.71]) or infectious diseases (SMR [95% CI] 5.74 [5.08–6.45]), while those receiving chemotherapy had a higher risk of respiratory disease death (SMR [95% CI] 5.61 [5.03–6.24]) (Supplementary Tables 21–24).
Marital status
The lowest risk of non-cancer mortality was observed in widowed patients, whereas separated, divorced, and unmarried patients all had a higher risk of non-cancer mortality. Within 1 year after diagnosis, separated patients had the highest risk of non-cancer mortality (SMR [95% CI] 57.94 [42.57–77.05]), mainly from respiratory disease (SMR [95% CI] 165.13 [44.99–422.79]). Between 1 and 5 years after diagnosis, divorced patients had the highest risk of non-cancer mortality (SMR [95% CI] 6.40 [6.10–6.72]), reflected mainly in deaths from digestive and infectious diseases. Similarly, at more than 5 years after diagnosis, it was still separated patients who had the highest risk of non-cancer mortality (SMR [95% CI] 1.96 [1.86–2.05]), which was mainly represented by deaths from diabetes mellitus and digestive diseases (Supplementary Tables 25–29).
In addition, patients younger than 60 years of age had a significantly higher risk of non-cancer death than those older than 60 years of age (SMR [95% CI] 13.71 [13.22–14.22]) (Supplementary Tables 3–4). Patients diagnosed with BC in 2012–2017 had a higher risk of all non-cancer deaths than those diagnosed earlier (SMR [95% CI] 5.96 [5.84–6.07]) (Supplementary Tables 10–12). Patients with a pathological type of neuroendocrine carcinoma had the highest risk of non-cancer deaths (SMR [95% CI] 4.68 [4.06–5.37]), mainly reflected in deaths from infectious diseases (Supplementary Tables 30–34).
Competing risk analysis
Multivariate competing risk models were used to assess prognostic factors for the development of BC-related death and non-cancer cause of death in BC patients (Table 4) and cumulative mortality (Supplementary Figs. 1 and 2). The risk of both mortalities increased significantly with increasing age, but patients of advanced age appeared to be more likely to experience non-cancer cause of death (HR [95% CI] 4.626 [4.481–4.775]) (Supplementary Figs. 1A and 2A). Female patients had a significantly lower risk of non-cancer cause of death than male patients, but their risk of BC-related death was significantly higher than that of male patients (Supplementary Figs. 1B and 2B). Black patients have the highest risk of BC-related death (HR [95% CI] 1.226 [1.180–1.274]) (Supplementary Fig. 1C). Patients who developed distant metastases had a very high risk of BC-related death (HR [95% CI] 30.345 [29.082–31.664]), in contrast to their very low risk of non-cancer cause of death (HR [95% CI] 0.429 [0.401–0.458]) (Supplementary Figs. 1D and 2D). Patients diagnosed between 2012 and 2017 had the lowest risk of non-cancer cause of death (HR [95% CI] 0.733 [0.715–0.751]) (Table 4). Patients with bladder adenocarcinoma had the lowest risk of both mortalities (Supplementary Figs. 1E and 2E). Patients who underwent RC surgery had the lowest risk of non-cancer cause of death (HR [95% CI] 0.685 [0.655–0.717]) (Supplementary Figs. 1F and 2F). The risk of non-cancer cause of death was higher in patients who did not receive either chemotherapy or radiation therapy (Supplementary Figs. 1G and 2H). Widowed patients have the highest risk of both BC-related and non-cancer cause of death (Supplementary Figs. 1I and 2I).
Discussion
Bladder cancer as one of the most common malignancies in urology, patients are often more concerned about BC-related mortality. However, due to continuous medical advances and developments, the overall survival of BC patients has been effectively improved(Richters et al. 2020; Lenis et al. 2020; Marrie et al. 2021), leading to the prominence of deaths from non-cancer diseases (Wang et al. 2022a).
Our study found that non-cancer causes of death from BC accounted for 52.48% of all deaths, with cardiovascular disease being the most common non-cancer cause of death, followed by respiratory disease, diabetes mellitus, and infectious diseases. Published studies showed that cardiovascular disease is one of the leading causes of non-cancer mortality (Du et al. 2021; Oh et al. 2020). Studies show that cancer patients are 2–6 times more likely to die from cardiovascular disease than the general population (Sturgeon et al. 2019). Studies have shown that chemotherapy and radiation therapy for cancer increase heart disease-specific mortality in patients (Yang et al. 2021; Dai et al. 2022; Guan et al. 2021). Meanwhile, other non-cancer diseases such as diabetes mellitus and nephrotic syndrome increase the cardiac burden by altering the body's metabolic status and hemodynamics. The risk of cardiovascular disease mortality in BC patients continues throughout the treatment and follow-up period. Therefore, we recommend more proactive self-observation and follow-up rather than reactive management only at the onset of clinical symptoms or complications, and that such self-observation and follow-up should continue from the start of oncologic treatment and for the rest of the life.
The incidence of respiratory disease is higher among cancer patients (Li et al. 2020; Deckx et al. 2012). Among them, pulmonary infections caused by neutropenia are the most common. Published meta-analysis showed that the use of immune checkpoint inhibitors, compared to chemotherapy, significantly increased the incidence of pneumonia (Nishijima et al. 2017). Also, the incidence of chronic obstructive pulmonary disease was significantly higher in cancer patients (Song et al. 2022; Zhang et al. 2021). Also, the increase in smoking and air pollution has led to an increase in the incidence of pneumonia or chronic obstructive pulmonary disease in cancer patients.
In cancer patients, it is very common for antitumor therapy (e.g., chemotherapy) to cause myelosuppression (Simonaggio et al. 2019). Neutropenia is an independent risk factor for sepsis (Kochanek et al. 2019b). Some studies have shown that cancer patients who have developed sepsis or bacteremia have an approximately 2.3 times higher risk of death than non-cancer patients (Abou Dagher et al. 2017). In addition, Liyanage et al. (2002) and Schreiber et al. (2011) showed that tumors can also cause an increased risk of infection in cancer patients through metastatic invasion and mechanical damage to the immune system.
The relationship between BC and diabetes mellitus should not be underestimated. Patients with a long history of impaired fasting glucose or diabetes mellitus are at significantly higher risk of BC due to diet, obesity and other factors (Choi et al. 2022; Lam et al. 2021; Gill et al. 2021). A meta-analysis suggests that diabetes mellitus has an increased risk of disease progression, recurrence and death in BC patients (Lu and Tao 2021). Meanwhile, our study showed a significantly higher SMR for diabetes mellitus -related deaths among BC patients, consistent with published studies. In addition, some studies have suggested that metformin use is associated with a better prognosis in non-muscle invasive BC (Wang et al. 2022b; Liu et al. 2022; Klose et al. 2021), but larger population studies are needed to confirm this.
Multivariate competing risk analysis showed that patients with BC are at significantly higher risk of experiencing death from non-cancer causes compared to the general population, with potential risk factors including: age > 60 years, male, whites, in situ stage, pathological type of transitional cell carcinoma, not receiving treatment (including surgery, chemotherapy, or radiation), and widowed. Therefore, for high-risk patients, we recommend aggressive treatment and follow-up. It is estimated that primary prevention of 30–40% of cancers can be achieved by modifying lifestyle and environmental risk factors known to be associated with cancer incidence. A healthy lifestyle, including reducing smoking, low BMI, being physically active, avoiding excessive alcohol consumption and maintaining a healthy nutrition are the best strategies to prevent and treat non-cancer diseases (Zhang et al. 2020).
Our study is limited by the inherent bias of the SEER database and the fact that this is a retrospective study without external validation. We reduce bias due to factors such as age, sex, and race by strictly controlling inclusion criteria and using SMR, while quantitatively assessing the impact of different factors on non-cancer mortality in BC patients through multivariate competing risk analysis. However, our inability to know whether patients had relevant non-cancerous diseases before diagnosis and the lack of data on some high-risk factors, such as smoking, alcohol consumption, and lifestyle, led to certain shortcomings in our study. Therefore, future high-quality studies are needed to explore the major causes of non-cancer mortality in BC patients and the associated risk factors.
Conclusion
Non-cancer diseases accounted for the highest proportion of deaths during each follow-up period after BC diagnosis, with cardiovascular diseases being the most common causes. SMR for non-cancer causes of death decreased progressively with increasing follow-up time and was approximately 7.5 times higher within 1 year than 1–5 years after diagnosis. Additionally, the risk of non-cancer causes of death in BC patients is impacted by a number of factors that are inherently heterogeneous, such as age, gender, treatment modality (surgery, chemotherapy, and radiation therapy), and marital status. Therefore, physicians should pay attention to the risk of death from non-cancer diseases. Also, physicians should encourage patients to engage in more proactive self-surveillance and follow up.
Availability of data and materials
The data in this study were obtained from the publicly available SEER database (https://seer.cancer.gov/: Incidence—SEER Research Plus Data, 18 Registries (excl AK), Nov 2020 Sub (2000–2018) for SMRs).
Abbreviations
- BC:
-
Bladder cancer
- SEER:
-
The Surveillance, Epidemiology, and End Results
- SMR:
-
Standardized mortality ratio
- CI:
-
Confidence interval
- Tcc:
-
Transitional cell carcinoma
- Scc:
-
Squamous cell carcinoma
- Nec:
-
Neuroendocrine carcinoma
- Ac:
-
Adenocarcinoma
- Oet:
-
Other epithelial tumors
- TURBT:
-
Transurethral resection of bladder tumor
- PC:
-
Partial cystectomy
- RC:
-
Radical cystectomy
References
Abdel-Rahman O (2017) Risk of cardiac death among cancer survivors in the United States: a SEER database analysis. Expert Rev Anticancer Ther 17(9):873–878
Abou Dagher G, El Khuri C, Chehadeh AA et al (2017) Are patients with cancer with sepsis and bacteraemia at a higher risk of mortality? A retrospective chart review of patients presenting to a tertiary care centre in Lebanon. BMJ Open 7(3):e13502
Choi YH, Park Y, Hur KJ et al (2022) Duration of diabetes mellitus and risk of kidney and bladder cancer: a longitudinal nationwide cohort study. Am J Cancer Res 12(8):4050–4061
Dai ZH, Tang M, Chen YL et al (2022) Incidence and risk factors for cerebrovascular-specific mortality in patients with colorectal cancer: a registry-based cohort study involving 563,298 patients. Cancers (basel). 14(9):2053
Deckx L, van den Akker M, Metsemakers J et al (2012) Chronic diseases among older cancer survivors. J Cancer Epidemiol 2012:206414
Du B, Wang F, Wu L et al (2021) Cause-specific mortality after diagnosis of thyroid cancer: a large population-based study. Endocrine 72(1):179–189
Gill E, Sandhu G, Ward DG et al (2021) The sirenic links between diabetes, obesity, and bladder cancer. Int J Mol Sci 22(20):11150
Guan T, Qiu Z, Su M et al (2021) Cardiovascular death risk in primary central nervous system lymphoma patients treated with chemotherapy: a registry-based cohort study. Front Oncol 11:641955
Jalanko T, de Jong JJ, Gibb EA et al (2020) Genomic subtyping in bladder cancer. Curr Urol Rep 21(2):9
Klose K, Packeiser E, Müller P et al (2021) Metformin and sodium dichloroacetate effects on proliferation, apoptosis, and metabolic activity tested alone and in combination in a canine prostate and a bladder cancer cell line. PLoS ONE 16(9):e257403
Kochanek KD, Murphy SL, Xu J et al (2019a) Deaths: final data for 2017. Natl Vital Stat Rep 68(9):1–77
Kochanek M, Schalk E, von Bergwelt-Baildon M et al (2019b) Management of sepsis in neutropenic cancer patients: 2018 guidelines from the infectious diseases working party (AGIHO) and intensive care working party (iCHOP) of the german society of hematology and medical oncology (DGHO). Ann Hematol 98(5):1051–1069
Lam BQ, Srivastava R, Morvant J et al (2021) Association of diabetes mellitus and alcohol abuse with cancer: molecular mechanisms and clinical significance. Cells-Basel 10(11):3077
Lenis AT, Lec PM, Chamie K et al (2020) Bladder cancer: a review. JAMA 324(19):1980–1991
Li J, Pang H, Sun Z et al (2020) Health status of middle-aged and older cancer survivors: a nationwide cross-sectional study from the China Health and Retirement Longitudinal Study (CHARLS). Ann Transl Med 8(5):183
Liu C, Sun J, Xu J et al (2022) Metformin use on incidence and oncologic outcomes of bladder cancer patients with T2DM: an updated meta-analysis. Front Pharmacol 13:865988
Liyanage UK, Moore TT, Joo HG et al (2002) Prevalence of regulatory T cells is increased in peripheral blood and tumor microenvironment of patients with pancreas or breast adenocarcinoma. J Immunol 169(5):2756–2761
Lu Y, Tao J (2021) Diabetes mellitus and obesity as risk factors for bladder cancer prognosis: a systematic review and meta-analysis. Front Endocrinol 12:699732
Marrie RA, Maxwell C, Mahar A et al (2021) Cancer incidence and mortality rates in multiple sclerosis: a matched cohort study. Neurology 96(4):e501–e512
Nishijima TF, Shachar SS, Nyrop KA et al (2017) Safety and tolerability of PD-1/PD-L1 inhibitors compared with chemotherapy in patients with advanced cancer: a meta-analysis. Oncologist 22(4):470–479
Oh CM, Lee D, Kong HJ et al (2020) Causes of death among cancer patients in the era of cancer survivorship in Korea: attention to the suicide and cardiovascular mortality. Cancer Med 9(5):1741–1752
Richters A, Aben K, Kiemeney L (2020) The global burden of urinary bladder cancer: an update. World J Urol 38(8):1895–1904
Robertson AG, Kim J, Al-Ahmadie H et al (2018) Comprehensive molecular characterization of muscle-invasive bladder cancer. Cell 174(4):1033
Schreiber RD, Old LJ, Smyth MJ (2011) Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science 331(6024):1565–1570
Siegel RL, Miller KD, Jemal A (2019) Cancer statistics, 2019. CA Cancer J Clin 69(1):7–34
Siegel RL, Miller KD, Jemal A (2020) Cancer statistics, 2020. CA Cancer J Clin 70(1):7–30
Siegel RL, Miller KD, Fuchs HE et al (2021) Cancer statistics, 2021. CA Cancer J Clin 71(1):7–33
Simonaggio A, Michot JM, Voisin AL et al (2019) Evaluation of readministration of immune checkpoint inhibitors after immune-related adverse events in patients with cancer. JAMA Oncol 5(9):1310–1317
Song P, Lu N, Zhang J et al (2022) Cause of death of patients with non-muscular invasive, non-metastatic muscular invasive and metastatic bladder cancer after diagnosis. Am J Transl Res 14(5):3494–3515
Sturgeon KM, Deng L, Bluethmann SM et al (2019) A population-based study of cardiovascular disease mortality risk in US cancer patients. Eur Heart J 40(48):3889–3897
Wang S, Ge C, Zhang J (2022a) Cardiovascular mortality risk in patients with bladder cancer: a population-based study. J Cardiovasc Dev Dis 9(8):255
Wang Z, Ong WYF, Shen T et al (2022b) Beyond diabetes mellitus: role of metformin in non-muscle-invasive bladder cancer. Singap Med J 63(4):209–213
Yang F, Li C, Guo Y et al (2021) Effects of radical cystectomy, radiotherapy, and chemotherapy on the risk of long-term heart-specific death in bladder cancer patients. Transl Androl Urol 10(10):3826–3836
Zhang Y, Pan X, Chen J et al (2020) Combined lifestyle factors, incident cancer, and cancer mortality: a systematic review and meta-analysis of prospective cohort studies. Br J Cancer 122(7):1085–1093
Zhang H, Lin W, Chen D et al (2021) Cardiovascular and other competing causes of death in male breast cancer patients: a population-based epidemiologic study. Clin Interv Aging 16:1393–1401
Zhang J, Chang S, Chiang M et al (2021) Survival impact of current-smoking-related COPD or COPD with acute exacerbation. J Pers Med 11(10):958
Acknowledgements
The authors would like to thank all working group members for their contribution to this study.
Funding
None.
Author information
Authors and Affiliations
Contributions
SW had full access to all of the data in the study, and took responsibility for the integrity of the data and the accuracy of the data analysis. SW and CG contributed to the writing of the manuscript. CG conceptualized the research, supervised the work, and contributed to the editing and writing of the final manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
432_2023_4867_MOESM1_ESM.tif
Supplementary file1 Supplementary figure 1 Cumulative mortality curves for bladder cancer-related deaths in patients with bladder cancer stratified by age (A), sex (B), race (C), summary stage (D), histologic type (E), surgery (F), chemotherapy (G), radiation therapy (H), and marital status (I) (TIF 7240 KB)
432_2023_4867_MOESM2_ESM.tif
Supplementary file2 Supplementary figure 2 Cumulative mortality curves for non-cancer deaths in patients with bladder cancer stratified by age (A), sex (B), race (C), summary stage (D), histologic type (E), surgery (F), chemotherapy (G), radiation therapy (H), and marital status (I) (TIF 6596 KB)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, S., Ge, C. High risk of non-cancer mortality in bladder cancer patients: evidence from SEER-Medicaid. J Cancer Res Clin Oncol 149, 10203–10215 (2023). https://doi.org/10.1007/s00432-023-04867-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-04867-z