Abstract
Background
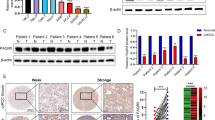
Ephrin-A2, a member of the Eph receptor subgroup, is used in diagnosing and determining the prognosis of prostate cancer. However, the role of ephrin-A2 in prostate cancer is remains elusive.
Methods
We established stable clones overexpressing or silencing ephrin-A2 from prostate cancer cells. Then, CCK-8 was used in analyzing the proliferation ability of cells. CD31 staining was used in evaluating angiogenesis. Migration and invasion assay were conducted in vivo and in vitro. The expression of EMT-related markers was evaluated in prostate cancer cells through Western blotting.
Results
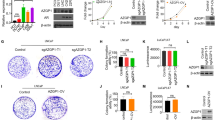
We revealed that the ectopic expression of ephrin-A2 in prostate cancer cells facilitated cell migration and invasion in vitro and promoted tumor metastasis and angiogenesis in vivo and that the silencing of ephrin-A2 completely reversed this effect. Although ephrin-A2 did not affect tumor cell proliferation in vitro, ephrin-A2 significantly promoted primary tumor growth in vivo. Furthermore, to determine the biological function of ephrin-A2, we assayed the expression of EMT-related markers in stable-established cell lines. Results showed that the overexpression of ephrin-A2 in prostate cancer cells down-regulated the expression of epithelial markers (ZO-1, E-cadherin, and claudin-1) and up-regulated the expression of mesenchymal markers (N-cadherin, β-catenin, vimentin, Slug, and Snail), but the knocking out of ephrin-A2 opposed the effects on the expression of EMT markers.
Conclusions
These findings indicate that ephrin-A2 promotes prostate cancer metastasis by enhancing angiogenesis and promoting EMT and may be a potentially therapeutic target in metastatic prostate cancer.






Similar content being viewed by others
Data availability statement
The raw data used and analyzed in the current study are available from the corresponding author upon a reasonable request.
References
Abufaraj M, Shariat SF, Haitel A et al (2017) Prognostic role of N-cadherin expression in patients with non-muscle-invasive bladder cancer. Urol Oncol 35(5):264–271
Cano A, Pérez-Moreno MA, Rodrigo I et al (2000) The transcription factor snail controls epithelial-mesenchymal transitions by repressing E-cadherin expression. Nat Cell Biol 2(2):76–83
Colas E, Pedrola N, Devis L et al (2012) The EMT signaling pathways in endometrial carcinoma. Clin Transl Oncol 14(10):715–720
Dissanayake SK, Wade M, Johnson CE et al (2007) The Wnt5A/protein kinase C pathway mediates motility in melanoma cells via the inhibition of metastasis suppressors and initiation of an epithelial to mesenchymal transition. J Biol Chem 282(23):17259–17271
Fang WB, Ireton RC, Zhuang G, Takahashi T, Reynolds A, Chen J (2008) Overexpression of EPHA2 receptor destabilizes adherens junctions via a RhoA-dependent mechanism. J Cell Sci 121(Pt 3):358–368
Feng YX, Zhao JS, Li JJ et al (2010) Liver cancer: EphrinA2 promotes tumorigenicity through Rac1/Akt/NF-kappaB signaling pathway. Hepatology 51(2):535–544
Foote RL, Weidner N, Harris J et al (2005) Evaluation of tumor angiogenesis measured with microvessel density (MVD) as a prognostic indicator in nasopharyngeal carcinoma: results of RTOG 9505. Int J Radiat Oncol Biol Phys 61(3):745–753
Fox BP, Kandpal RP (2004) Invasiveness of breast carcinoma cells and transcript profile: Eph receptors and ephrin ligands as molecular markers of potential diagnostic and prognostic application. Biochem Biophys Res Commun 318(4):882–892
Gerhardt H, Betsholtz C (2003) Endothelial-pericyte interactions in angiogenesis. Cell Tissue Res 314(1):15–23
Hafner C, Schmitz G, Meyer S et al (2004) Differential gene expression of Eph receptors and ephrins in benign human tissues and cancers. Clin Chem 50(3):490–499
Hazan RB, Phillips GR, Qiao RF, Norton L, Aaronson SA (2000) Exogenous expression of N-cadherin in breast cancer cells induces cell migration, invasion, and metastasis. J Cell Biol 148(4):779–790
Hou F, Yuan W, Huang J et al (2012) Overexpression of EphA2 correlates with epithelial-mesenchymal transition-related proteins in gastric cancer and their prognostic importance for postoperative patients. Med Oncol 29(4):2691–2700
Kandouz M (2012) The Eph/Ephrin family in cancer metastasis: communication at the service of invasion. Cancer Metastasis Rev 31(1–2):353–373
Kuphal S, Bosserhoff AK (2006) Influence of the cytoplasmic domain of E-cadherin on endogenous N-cadherin expression in malignant melanoma. Oncogene 25(2):248–259
Lahat G, Zhu QS, Huang KL et al (2010) Vimentin is a novel anti-cancer therapeutic target; insights from in vitro and in vivo mice xenograft studies. PLoS ONE 5(4):e10105
Li S, Wu Z, Chen Y et al (2016) Diagnostic and prognostic value of tissue and circulating levels of Ephrin-A2 in prostate cancer. Tumour Biol 37(4):5365–5374
Li S, Zhao Y, Chen W et al (2018) Exosomal ephrinA2 derived from serum as a potential biomarker for prostate cancer. J Cancer 9(15):2659–2665
Liu W, Guan M, Hu T, Gu X, Lu Y (2011) Re-expression of AKAP12 inhibits progression and metastasis potential of colorectal carcinoma in vivo and in vitro. PLoS ONE 6:e24015
Macrae M, Neve RM, Rodriguez-Viciana P et al (2005) A conditional feedback loop regulates Ras activity through EphA2. Cancer Cell 8(2):111–118
Manning G, Whyte DB, Martinez R, Hunter T, Sudarsanam S (2002) The protein kinase complement of the human genome. Science 298(5600):1912–1934
Mariotti A, Perotti A, Sessa C, Rüegg C (2007) N-cadherin as a therapeutic target in cancer. Expert Opin Investig Drugs 16(4):451–465
Miura K, Nam JM, Kojima C, Mochizuki N, Sabe H (2009) EphA2 engages Git1 to suppress Arf6 activity modulating epithelial cell-cell contacts. Mol Biol Cell 20(7):1949–1959
Nakajima S, Doi R, Toyoda E et al (2004) N-cadherin expression and epithelial-mesenchymal transition in pancreatic carcinoma. Clin Cancer Res 10(12):4125–4133
Nassar ZD, Moon H, Duong T et al (2013) PTRF/Cavin-1 decreases prostate cancer angiogenesis and lymphangiogenesis. Oncotarget 4(10):1844–1855
Pasquale EB (2010) Eph receptors and ephrins in cancer: bidirectional signalling and beyond. Nat Rev Cancer 10(3):165–180
Saito T, Masuda N, Miyazaki T et al (2004) Expression of EphA2 and E-cadherin in colorectal cancer: correlation with cancer metastasis. Oncol Rep 11(3):605–611
Satelli A, Li S (2011) Vimentin in cancer and its potential as a molecular target for cancer therapy. Cell Mol Life Sci 68(18):3033–3046
Shi Z, Xiaofu Y, Xiaojia W, et al (2015) A primary study on ephrin-A2 promoted migration of MDA-MB-231 breast cancer cells. J Med Res **
Smith RA, Manassaram-Baptiste D, Brooks D et al (2014) Cancer screening in the United States, 2014: a review of current American Cancer Society guidelines and current issues in cancer screening. CA Cancer J Clin 64(1):30–51
Su B, Bu Y, Engelberg D, Gelman IH (2010) SSeCKS/Gravin/AKAP12 inhibits cancer cell invasiveness and chemotaxis by suppressing a protein kinase C- Raf/MEK/ERK pathway. J Biol Chem 285:4578–4586
Yuan W, Chen Z, Wu S et al (2009) Expression of EphA2 and E-cadherin in gastric cancer: correlated with tumor progression and lymphogenous metastasis. Pathol Oncol Res 15(3):473–478
Zheng J, Zhao S, He X et al (2016) The up-regulation of long non-coding RNA CCAT2 indicates a poor prognosis for prostate cancer and promotes metastasis by affecting epithelial-mesenchymal transition. Biochem Biophys Res Commun 480(4):508–514
Funding
This study was supported by the grant from the Postdoctoral Science Foundation of China (No.2020M670076ZX), the National Natural Science Foundation of China (No. 81702061), the Natural Science Foundation of Jiangsu Province (No. BK20160233) and the Jiangsu Provincial Medical Youth Talent (No. QNRC2016781).
Author information
Authors and Affiliations
Contributions
SBL, YZ and CCC contributed to the idea, drafting and editing of the manuscript. LUS and JWW completed statistical analysis. MMZ and HLZ contributed to literature search and feeding animals and data analysis. PM and SBL revised the manuscript with critical reviews and comments and all the authors approved the final version.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical statement
The animals in this study were obtained in accordance with the ethical standards of the ethics committee of the affiliated Hospital of Xuzhou Medical University.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
432_2021_3618_MOESM1_ESM.tif
Supplementary file1 Supporting Fig. 1 Ephrin-A2 promotes tumor angiogenesis. VEGF was evaluated in tumor tissues of tumor-bearing mice (DU145 group) through immunohistochemical staining. The criterion for a positive immune reaction for VEGF is a dark brown cytoplasmic precipitate. Scale bar, 100 µm. (TIF 1420 KB)
432_2021_3618_MOESM2_ESM.tif
Supplementary file2 Supporting Fig. 2 Effects of EphrinA2 on migration of PC-3 cells in vitro. Wound areas of overexpression ephrin-A2 PC-3 cells. The cells were cultured in regular medium and wound healing abilities were quantified by measuring the average gap area between 0 and 24 h with ImageJ. (TIF 179 KB)
432_2021_3618_MOESM3_ESM.tif
Supplementary file3 Supporting Fig. 3 Effects of EphrinA2 on migration of LNCaP cells in vitro. Wound areas of knockdown ephrin-A2 LNCaP cells. The cells were cultured in regular medium and wound healing abilities were quantified by measuring the average gap area between 0 and 24 h with ImageJ. (TIF 187 KB)
432_2021_3618_MOESM4_ESM.tif
Supplementary file4 Supporting Fig. 4 Effects of EphrinA2 on migration of DU145 cells in vitro. Wound areas of knockdown ephrin-A2 DU145 cells. The cells were cultured in regular medium and wound healing abilities were quantified by measuring the average gap area between 0 and 24 h with ImageJ. (TIF 159 KB)
Rights and permissions
About this article
Cite this article
Zhao, Y., Cai, C., Zhang, M. et al. Ephrin-A2 promotes prostate cancer metastasis by enhancing angiogenesis and promoting EMT. J Cancer Res Clin Oncol 147, 2013–2023 (2021). https://doi.org/10.1007/s00432-021-03618-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-021-03618-2