Abstract
The risk for venous thromboembolism (VTE) is considered to be low in the general paediatric intensive care unit (PICU) population, and pharmacological thromboprophylaxis is not routinely used. PICU patients considered at high-risk of VTE could possibly benefit from pharmacological thromboprophylaxis, but the incidence of VTE in this group of patients is unclear. This was an observational, prospective study at a tertiary multi-disciplinary paediatric hospital. We used comprehensive ultrasonography screening for VTE in critically ill children with multiple risk factors for VTE. Patients admitted to PICU ≥ 72 h and with ≥ two risk factors for VTE were included. Patients receiving pharmacological thromboprophylaxis during their entire PICU stay were excluded. The primary outcome of the study was VTEs not related to the use of a CVC. Ultrasonography screening of the great veins was performed at PICU discharge. Seventy patients with median (interquartile range) 3 (2–4) risk factors for VTE were evaluated. Median age was 0.3 years (0.03–4.3) and median PICU length of stay 9 days (5–17). Regarding the primary outcome, no symptomatic VTEs occurred and no asymptomatic VTEs were found on ultrasonography screening, resulting in an incidence of VTEs not related to a vascular catheter of 0% (95% CI: 0–5.1%).
Conclusion: Our results indicate that VTEs not related to a vascular catheter are a rare event even in a selected group of severely ill small children considered to be at high risk of VTE.
What is Known: • Children in the PICU often have several risk factors for venous thromboembolism (VTE). • The incidence of VTE in PICU patients is highly uncertain, and there are no evidence-based guidelines regarding VTE prophylaxis. | |
What is New: • This study found an incidence of VTEs not related to a vascular catheter of 0% (95% CI: 0–5.1%). • This indicates that such VTE events are rare even in PICU patients with multiple risk factors for VTE. |
Similar content being viewed by others
Introduction
Paediatric venous thromboembolism (VTE) carries the risk of significant complications, including sepsis and post-thrombotic syndrome [1]. In healthy children, the risk of VTE is low and VTE is mainly considered to be a disease of hospitalized and critically ill children [2]. However, in critically ill children, clinical signs of VTE can easily be missed or ascribed to the underlying condition. This is especially true in small children with limited communication abilities, and where venous collaterals often develop rapidly. Even occlusive VTE can be asymptomatic in children [3]. In order to investigate the true incidence of VTE in paediatric intensive care unit (PICU) patients, screening for VTE is necessary [4].
Even though previous data is limited, VTE seems to be an uncommon event in the general PICU population [5]. This suggests that pharmacological thromboprophylaxis (pTP) might not be indicated in a broad PICU population without specific risk factors for VTE. The evidence-based guidelines on antithrombotic therapy for children by the American College of Chest Physicians do not recommend the universal adoption of pTP in the general PICU population [6]. In line with this, only 4.5% of PICU patients received pTP with low molecular weight heparin (LMWH) in a multinational study of pTP practice in critically ill children published in 2014 [7]. However, most data in the field originate from retrospective studies of symptomatic cases, presumably underestimating the true incidence of VTE. The lack of robust data on which to base guidelines has resulted in a large variation in the routines for prescription of pTP for PICU patients [7; 8]. Moreover, there is a lack of evidence on the efficacy of pTP in children. In adult ICU patients, there is a 50% lower risk of VTE with the use of heparins for VTE prophylaxis [9], but no data from the paediatric setting exist. There are also possible adverse effects of using pTP. Administration of LMWH is painful to children and has potential side-effects such as bleeding [10], heparin-induced thrombocytopenia and neonatal osteopenia [11].
Patients in the PICU are a heterogenous group, and the indication for pTP may be stronger in PICU patients considered to be at high risk of VTE. Recent recommendations suggest that PICU patients with at least two risk factors for VTE may benefit from pTP [11], but there is currently no data describing the incidence of VTE in this population. Such data is necessary to better understand the risk/benefit ratio for pTP in high risk patients, and to adequately power randomized controlled studies investigating the efficacy of pTP in this setting.
In this study, we conducted a comprehensive screening for VTE using doppler ultrasonography (US) in critically ill children admitted to the PICU for ≥ 72 h and with at least two risk factors for VTE. Ultrasound evaluation of the great veins was performed to ensure that also asymptomatic VTEs were included in the study. Our main objective was to provide new data on the incidence of VTE in a group of PICU patients considered to be at high risk of VTE.
Methods
The Regional Ethics Review Board in Stockholm (reference No 2015/140–31/1, 15/02/2015) approved the study. The study protocol was registered at the Australian New Zeeland Clinical Trial Registry (ACTRN12615000441516). Consent was obtained from parents and when possible from the child.
Study design
This was a prospective observational study, conducted at the PICU of a multi-disciplinary referral centre for critically ill children in Stockholm, Sweden. Patients were consecutively included from April 2015 to November 2016, although inclusion was paused during 6 weeks in the summers of 2015 and 2016 due to lack of ultrasound resources.
All patients < 18 years of age, weighing ≥ 1250 g, admitted to the PICU for ≥ 72 h, and with at least two risk factors for VTE were eligible for inclusion. Patients were not eligible for inclusion if death was deemed imminent and inevitable.
Study procedure
Data on patient demographics and risk factors were collected using a standardized report form. Risk factors for VTE included the presence of a CVC, congenital heart disease, trauma, cancer, previous VTE, perioperative patient, renal failure, invasive mechanical ventilation, sepsis, age < 1 year or > 12 years, oral contraception, active inflammatory disease or antiphospholipid syndrome. Sepsis was defined according to international consensus guidelines [12]. Acute kidney injury (AKI) was defined as serum creatinine increased > twofold above either baseline or the upper limit age-adjusted reference intervall, or the need for continuous renal replacement therapy (CRRT). The recorded patient data included age, prematurity (for children < 1 year), gender and body weight. PICU admission diagnosis and length of stay were noted. Pediatric Index of Mortality-2 (PIM-2) score was used to determine degree of illness at PICU admission. The PICU requirement for mechanical ventilation, CRRT and extracorporeal membrane oxygenation (ECMO) was documented. Platelet count, activated partial thromboplastin time (aPTT), international normalized ratio (INR), antithrombin III, fibrinogen level and d-dimer level at admission and discharge were registered if available. Children were not submitted to additional needle-pricks in order to obtain missing coagulation parameters. Patients receiving pTP or anticoagulation therapy during their entire PICU stay (defined as < 48 h without pTP/anticoagulation therapy) were excluded from the study.
Outcome measures
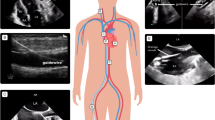
The incidence of VTEs that were not related to a CVC was chosen as the primary outcome of the study. Patients were followed for clinical signs of VTE (swelling of extremity, pain, discoloration or erythema, superior vena cava syndrome, visible collateral veins) during their PICU stay. An extensive screening for VTE using compression US with colour Doppler was performed at the time of discharge from the PICU. The veins evaluated included lower extremity (popliteal, superficial femoral, common femoral), upper extremity (subclavian, internal jugular, brachiocephalic) and intraabdominal (external iliac, common iliac, inferior caval, portal, renal) veins. The veins with a previous or existing CVC were included in the US screening protocol. US was performed by a paediatric radiologist or sonographer. Linear array vascular transducer of 5–12 MHz and a width of 6–8 cm was used. In most patients, Siemens Acuson S 2000 was used, while a small number of patients were examined with a Philips EPIQ7. The diagnosis of venous thrombosis was made on the basis of previously described direct and indirect signs [13]. Symptomatic VTE was defined as the presence of swelling, pain, discoloration or erythema, visible collateral veins or superior vena cava syndrome. Management strategy and follow-up plan for VTEs were decided by the clinical team managing the patient.
Statistical analysis
To assess normality, D’Agostino Pearson omnibus normality test was used. Non-parametric data are presented as median and interquartile range (IQR) and normally distributed data as mean with standard deviation (SD) as distribution measure. Wilcoxon signed-rank test was used to compare groups with paired, non-parametric data. Proportions are presented with their 95% confidence intervals (CI). Analyses were performed using GraphPad Prism 8.0.1(GraphPad Software, 2365 Northside Dr, Suite 560 San Diego, CA 92,108, USA).
Results
During the study period, 146 ICU admission met the study inclusion criteria. One hundred twenty-one of these were included in the study (Fig. 1). Ultrasound screening was not performed in 42 patients. In 36 cases, this was due to lack of ultrasound resources and in 6 cases due to PICU mortality or limitation of care orders. Causes of death were severe ischemic brain injury (n = 2), necrotizing enterocolitis with bowel gangrene (n = 1), and vein of Galen malformation with severe heart failure (n = 1). In two patients, limitation of care decisions was made during their PICU stay, before transfer to a general ward. Both children suffered from rare congenital syndromes with short predicted life-span. Five patients (aged 8 weeks–17.7 years) were excluded since they received pTP or anticoagulation therapy. Two patients received prophylactic LMWH, one postoperatively and one following severe trauma. One patient received LMWH during the entire PICU stay due to Lemierre’s syndrome with internal jugular vein thrombosis and one patient due to CVC-related VTE. One patient was on CRRT and received a heparin infusion as anticoagulation. In four cases, the ultrasound evaluation could not be completed due to lack of patient cooperation or technical problems. Characteristics of included patients without ultrasound screening are supplied in supplemental Table S1. Seventy admissions were included in the final analysis (Fig. 1).
The median (IQR) patient age and weight were 0.3 (0–4.3) years and 5.2 (3.3–15.1) kg, respectively (Table 1). Forty-six (65.7%) patients were < 1 year of age, and 31 (44.3%) were female (Table 1). The most common admission diagnosis was respiratory failure, followed by surgery for congenital abnormalities, sepsis and seizures (Table 1). The median admission PIM-2 score was 4.6 (1.4–10.8). The median number of risk factors for VTE was 3 (2–4) (Table 2). Most patients (85.7%) needed a CVC during their PICU stay. Mechanical ventilation was required in 51 (72.9%) cases, with a duration of 8 (4–14) days. During the PICU stay, 28 patients fulfilled the criteria for sepsis. Two patients had previously had a VTE, and 1 patient suffered from a disease with increased risk of VTE (carbohydrate-deficient glycoprotein syndrome type Ia) (Table 2). Four (5.7%) patients needed CRRT for a median of 6.5 days (Table 1), three of them received anticoagulation with heparin during CRRT. Six patients (8.6%) required ECMO treatment. Median length of PICU and hospital length of stay were 9 (5–17) days and 22 (11–50.2) days, respectively (Table 1). Coagulation parameters at admission and discharge are shown in Table 3. During PICU stay, platelet count, antithrombin III, fibrinogen and d-dimer levels increased significantly, while INR and aPTT decreased.
Regarding the primary outcome, VTEs not related to a vascular catheter, no patient experienced a symptomatic VTE during their PICU stay and no VTE was found with US screening at PICU discharge. The resulting VTE incidence was 0% (95% CI: 0–5.1%). CVC-related VTE was diagnosed in 8 (11.4%, 95% CI: 4–19%) patients during their PICU stay. No cases of severe bleeding were found. In 46 patients, ultrasound screening could not be performed due to lack of ultrasound resources, incomplete ultrasound or palliation/death. Patient characteristics for these 46 patients are shown in supplemental Table S1. None of these patients developed a symptomatic VTE. This resulted in an incidence of symptomatic VTE not related to a vascular catheter of 0% (95% CI: 0–3.1%), considering all the 116 patients that were followed for signs of symptomatic VTE.
Discussion
The main goal of this study was to obtain new data describing the incidence of VTE in a group of PICU patients considered to be at high risk of VTE. We used an extensive ultrasound screening programme to investigate the risk of VTE in 70 severely ill small children with multiple risk factors for VTE. VTE not related to the use of a CVC was chosen as the primary outcome of the study. The non-CVC associated VTE incidence of 0% (95% CI: 0–5.1%) found in our study indicates that VTEs not related to vascular catheters are an uncommon event even in a selected group of PICU patients with a median of three risk factors for VTE. Ultrasound screening was not performed in 46 patients due to lack of ultrasound resources, incomplete ultrasound or palliative care/death. However, none of these patients had a symptomatic VTE, resulting in an incidence of symptomatic non-CVC associated VTE of 0% (0–3.1%) in 116 patients.
Even though paediatric VTE is an uncommon event [14], severe illness necessitating PICU admission is considered to be a risk factor for VTE in children [15]. pTP is still usually not prescribed to PICU patients, even though there is a large variation in practice [7, 8]. This is most likely due to the low incidence of VTE described in the general PICU population [5]. However, previous studies on the incidence of VTE in the PICU are mainly retrospective or register-based, not including asymptomatic VTE. This carries the risk of underestimating the true incidence of VTE. There is a lack of prospective high quality data on the incidence of VTE in PICU patients, and there are currently no evidence-based guidelines regarding VTE prophylaxis in the PICU population. Considering the low incidence of DVT in children, experts have recommended that specific high risk patient populations should be targeted for VTE research [8, 16].
When prescribing pTP, the potential for patient benefit in terms of preventing a VTE must be weighed against definitive (pain and discomfort) and potential (bleeding, heparin-induced thrombocytopenia and neonatal osteopenia) side-effects. Our study provides new data helping the PICU physician to better assess this risk–benefit ratio for pTP in critically ill prepubertal children. This study also provides data useful for power calculation of RCTs evaluating the use of pTP in PICU patients.
From recent studies on paediatric CVC-related VTE, we have learned that the majority of CVC-related VTEs in children are asymptomatic [17]. Whether or not this is true also for other types of VTEs in the PICU has not previously been studied. Clinical diagnosis of VTE can be difficult in the PICU [16]. Small children are non-verbal, and sedation and mechanical ventilation often prevent communication of symptoms in older children. Also, symptoms can be ascribed to the underlying condition rather than to a VTE. Portal and renal vein thrombosis often present with non-specific symptoms and can be challenging conditions to identify [2]. Our results indicate that asymptomatic non-CVC associated VTE is rare in PICU-patients considered to be at high risk of VTE.
The primary outcome of this study was VTEs not associated with a CVC. There are several reasons for this. The incidence of and risk factors for CVC-related VTE in children have previously been extensively studied [18,19,20,21], whereas the previous data on VTEs not related to a vascular catheter are very sparse. The pathophysiology of CVC-related VTE has several distinct features, including endothelial damage, partial occlusion of the vein, and the presence of foreign material [22]. Also, the effectiveness of LMWH or other pTPs in preventing CVC-related VTE in children or adults is uncertain [22,23,24,25,26].
One of the strengths of our study is that we provide prospective data on the incidence of VTE in PICU patients, including a thorough screening for asymptomatic VTEs. Moreover, we targeted a PICU population considered to be at high risk of VTE. The comprehensive ultrasound screening was performed by experienced paediatric radiologists and sonographers, thereby avoiding underestimation of the true incidence of VTE. By evaluating upper extremity, lower extremity and abdominal veins, we included the majority of veins previously described to be at risk for VTE in children. In the study by Raffini et al. 19.5% of VTEs occurred in the portal, renal or caval veins [14], highlighting the importance of including these veins in US screening. The majority of patients in our study were neonates and infants, making our data representative for a group of patients where the coagulation system is not yet fully matured [27].
There are certain limitations to be considered when interpretating the results. This is a single-centre study from a general, tertiary paediatric ICU and the study population consisted mainly of small, prepubertal children. Our results may not be applicable to other centres or age groups, in particular older, postpubertal children. A larger patient sample would have given a more precise estimation of the true incidence of VTE in this group. The US screening procedure is very resource demanding and not always available, which led to a substantial loss to follow-up. This is a limitation of the study and the possibility of selection bias must be considered when interpreting the results. Moreover, we only included patients considered to be at high risk of VTE, thereby limiting the number of patients possible to include. A limitation of US is the lower sensitivity for VTE in veins where compressibility is difficult to assess [28]. However, US is an accepted screening method for VTE diagnosis [29]. In 6 patients included in the study, ultrasound evaluation could not be performed due to PICU mortality or limitation of care decisions. All these patients suffered from severe medical conditions explaining their deterioration, and none had clinical signs or suspicion of thromboembolic complications. Autopsy was performed in both patients with limitation of care orders and in one patient who died in the PICU due to necrotizing enterocolitis, and no VTEs were found in these patients.
Conclusion
Using a comprehensive prospective screening programme in a group of critically ill children with a median of three risk factors of VTE, we did not find any VTEs that were not related to a CVC. Our results indicate that non-CVC associated VTE is an uncommon event even in a selected group of severely ill small children considered to be at high risk of VTE.
Abbreviations
- APTT:
-
Activated partial thromboplastin time
- CRRT:
-
Continuous renal replacement therapy
- CVC:
-
Central venous catheter
- ECMO:
-
Extracorporeal membrane oxygenation
- INR:
-
International normalized ratio
- IQR:
-
Interquartile range
- LMWH:
-
Low molecular weight heparin
- PICU:
-
Paediatric intensive care unit
- PIM-2:
-
Pediatric Index of Mortality-2
- pTP:
-
Pharmacological thromboprophylaxis
- SD:
-
Standard deviation
- US:
-
Ultrasound
- VTE:
-
Venous thromboembolism
References
Biss TT (2016) Venous thromboembolism in children: is it preventable? Semin Thromb Hemost 42:603–611
Jaffray J, Young G (2018) Deep vein thrombosis in pediatric patients. Pediatr Blood Cancer 65
Ostlund A, Flaring U, Norberg A, Dahlberg A, Berner J, Kaiser S, Vermin L, Svenningsson A, Frisk T, Larsson P, Andersson A (2019) Incidence of and risk factors for venous thrombosis in children with percutaneous non-tunnelled central venous catheters. Br J Anaesth 123:316–324
Boddi M, Peris A (2017) Deep vein thrombosis in intensive care. Adv Exp Med Biol 906:167–181
Higgerson RA, Lawson KA, Christie LM, Brown AM, McArthur JA, Totapally BR, Hanson SJ (2011) Incidence and risk factors associated with venous thrombotic events in pediatric intensive care unit patients. Pediatr Crit Care Med : J Soc Crit Care Med World Federation Pediatr Intensive Crit Care Soc 12:628–634
Monagle P, Chan AKC, Goldenberg NA, Ichord RN, Journeycake JM, Nowak-Gottl U, Vesely SK (2012) Antithrombotic therapy in neonates and children: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 141:e737S-e801S
Faustino EV, Hanson S, Spinella PC, Tucci M, O’Brien SH, Nunez AR, Yung M, Truemper E, Qin L, Li S, Marohn K, Randolph AG (2014) A multinational study of thromboprophylaxis practice in critically ill children. Crit Care Med 42:1232–1240
Faustino EV, Patel S, Thiagarajan RR, Cook DJ, Northrup V, Randolph AG (2011) Survey of pharmacologic thromboprophylaxis in critically ill children. Crit Care Med 39:1773–1778
Alhazzani W, Lim W, Jaeschke RZ, Murad MH, Cade J, Cook DJ (2013) Heparin thromboprophylaxis in medical-surgical critically ill patients: a systematic review and meta-analysis of randomized trials. Crit Care Med 41:2088–2098
Petty JK (2017) Venous thromboembolism prophylaxis in the pediatric trauma patient. Semin Pediatr Surg 26:14–20
Newall F, Branchford B, Male C (2018) Anticoagulant prophylaxis and therapy in children: current challenges and emerging issues. Journal of thrombosis and haemostasis : JTH 16:196–208
Goldstein B, Giroir B, Randolph A (2005) International pediatric sepsis consensus conference: definitions for sepsis and organ dysfunction in pediatrics. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 6:2–8
Tanaka S, Nishigami K, Taniguchi N, Matsuo H, Hirai T, Kaneda S, Ogasawara M, Satoh H, Tobe H (2008) Criteria for ultrasound diagnosis of deep venous thrombosis of lower extremities. J Med Ultrason (2001) 35:33–36
Raffini L, Huang YS, Witmer C, Feudtner C (2009) Dramatic increase in venous thromboembolism in children’s hospitals in the United States from 2001 to 2007. Pediatrics 124:1001–1008
Mahajerin A, Branchford BR, Amankwah EK, Raffini L, Chalmers E, van Ommen CH, Goldenberg NA (2015) Hospital-associated venous thromboembolism in pediatrics: a systematic review and meta-analysis of risk factors and risk-assessment models. Haematologica 100:1045–1050
Chima RS, Hanson SJ (2017) Venous Thromboembolism in Critical Illness and Trauma: Pediatric Perspectives. Front Pediatr 5:47
Citla Sridhar D, Abou-Ismail MY, Ahuja SP (2020) Central venous catheter-related thrombosis in children and adults. Thromb Res 187:103–112
Beck C, Dubois J, Grignon A, Lacroix J, David M (1998) Incidence and risk factors of catheter-related deep vein thrombosis in a pediatric intensive care unit: a prospective study. J Pediatr 133:237–241
Faustino EV, Spinella PC, Li S, Pinto MG, Stoltz P, Tala J, Card ME, Northrup V, Baker KE, Goodman TR, Chen L, Silva CT (2013) Incidence and acute complications of asymptomatic central venous catheter-related deep venous thrombosis in critically ill children. J Pediatr 162:387–391
Gray BW, Gonzalez R, Warrier KS, Stephens LA, Drongowski RA, Pipe SW, Mychaliska GB (2012) Characterization of central venous catheter-associated deep venous thrombosis in infants. J Pediatr Surg 47:1159–1166
Szeps I, Östlund Å, Norberg Å, Fläring U, Andersson A (2021) Thromboembolic complications of vascular catheters used for pediatric continuous renal replacement therapy: prevalence in a single-center, retrospective cohort. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 22:743–752
Baumann Kreuziger L, Jaffray J, Carrier M (2017) Epidemiology, diagnosis, prevention and treatment of catheter-related thrombosis in children and adults. Thromb Res 157:64–71
Massicotte P, Julian JA, Gent M, Shields K, Marzinotto V, Szechtman B, Chan AK, Andrew M (2003) An open-label randomized controlled trial of low molecular weight heparin for the prevention of central venous line-related thrombotic complications in children: the PROTEKT trial. Thromb Res 109:101–108
Schroeder AR, Axelrod DM, Silverman NH, Rubesova E, Merkel E, Roth SJ (2010) A continuous heparin infusion does not prevent catheter-related thrombosis in infants after cardiac surgery. Pediatr Crit Care Med : J Soc Crit Care Med World Federation Pediatr Intensive Crit Care Soc 11:489–495
Vidal E, Sharathkumar A, Glover J, Faustino EV (2014) Central venous catheter-related thrombosis and thromboprophylaxis in children: a systematic review and meta-analysis. J Thromb Haemost : JTH 12:1096–1109
Faustino EVS, Shabanova V, Raffini LJ, Kandil SB, Li S, Pinto MG, Cholette JM, Hanson SJ, Nellis ME, Silva CT, Chima R, Sharathkumar A, Thomas KA, McPartland T, Tala JA, Spinella PC (2021) Efficacy of early prophylaxis against catheter-associated thrombosis in critically ill children: a Bayesian Phase 2b Randomized Clinical Trial. Crit Care Med 49:e235–e246
Ignjatovic V, Mertyn E, Monagle P (2011) The coagulation system in children: developmental and pathophysiological considerations. Semin Thromb Hemost 37:723–729
Male C, Chait P, Ginsberg JS, Hanna K, Andrew M, Halton J, Anderson R, McCusker P, Wu J, Abshire T, Cherrick I, Mahoney D, Mitchell L (2002) Comparison of venography and ultrasound for the diagnosis of asymptomatic deep vein thrombosis in the upper body in children: results of the PARKAA study. Prophylactic Antithrombin Replacement in Kids with ALL treated with Asparaginase. Thromb Haemost 87:593–598
Mitchell LG, Male C (2011) Outcome measures in interventional trials for prevention or treatment of venous thrombosis in the pediatric population. Semin Thromb Hemost 37:840–847
Acknowledgements
The authors would like to thank E. Mellgren and A. Abdella-Mussa, MC. Johansson and J Meijers at the Department of Paediatric Perioperative Medicine and Intensive Care, Stockholm for collecting data and providing care for the study patients.
Funding
Open access funding provided by Karolinska Institute. This work was supported by grants from the Samariten foundation for paediatric research, Fredrik and Ingrid Thuring’s foundation, Her Royal Highness Crown Princess Lovisa’s foundation, the Swedish Society of Medicine, and the Swedish Association for Paediatric Anaesthesiology and Intensive Care. The study was also funded by the Department of Paediatric Perioperative Medicine and Intensive Care, Karolinska University Hospital.
Author information
Authors and Affiliations
Contributions
Å.Ö.: patient recruitment, data collection, data analysis, drafting the manuscript, editing and approval of the manuscript. U.F.: study design, patient recruitment, data collection, data analysis, drafting the manuscript, editing and approval of the manuscript. Å.N.: data analysis, drafting the manuscript, editing and approval of the manuscript. A.D.: patient recruitment, data collection, editing and approval of the manuscript. J.B., S.K, L.V., T.F.: study design, data collection, editing and approval of the manuscript. P.L.: study design, patient recruitment, data collection, drafting the manuscript, editing and approval of the manuscript. A.A.: study design, patient recruitment, data collection, data analysis, drafting the manuscript, editing and approval of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by The Regional Ethics Review Board in Stockholm (reference No 2015/140–31/1, 15/02/2015).
Consent to participate
All children aged 12 years and above and all parents gave a written informed consent prior to inclusion in the study.
Consent for publication
Not applicable.
Conflict of interest
The authors declare competing interests.
Additional information
Communicated by Piet Leroy
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Östlund, Å.K.M., Fläring, U., Larsson, P. et al. Incidence of venous thromboembolic events not related to vascular catheters in a prospective cohort of critically ill children. Eur J Pediatr 181, 3031–3038 (2022). https://doi.org/10.1007/s00431-022-04487-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-022-04487-8