Abstract
The European Paediatric Regulation mandated the European Commission to fund research on off-patent medicines with demonstrated therapeutic interest for children. Responding to this mandate, five FP7 project calls were launched and 20 projects were granted. This paper aims to detail the funded projects and their preliminary results. Publicly available sources have been consulted and a descriptive analysis has been performed. Twenty Research Consortia including 246 partners in 29 European and non-European countries were created (involving 129 universities or public-funded research organisations, 51 private companies with 40 SMEs, 7 patient associations). The funded projects investigate 24 medicines, covering 10 therapeutic areas in all paediatric age groups. In response to the Paediatric Regulation and to apply for a Paediatric Use Marketing Authorisation, 15 Paediatric Investigation Plans have been granted by the EMA-Paediatric Committee, including 71 studies of whom 29 paediatric clinical trials, leading to a total of 7,300 children to be recruited in more than 380 investigational centres.
Conclusion: Notwithstanding the EU contribution for each study is lower than similar publicly funded projects, and also considering the complexity of paediatric research, these projects are performing high-quality research and are progressing towards the increase of new paediatric medicines on the market. Private–public partnerships have been effectively implemented, providing a good example for future collaborative actions. Since these projects cover a limited number of off-patent drugs and many unmet therapeutic needs in paediatrics remain, it is crucial foreseeing new similar initiatives in forthcoming European funding programmes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In Europe, fewer than 30 % of marketed drugs include results from paediatric clinical trials and other information on paediatric use in their documentation (Summary of Product Characteristics, SPC or Product Leaflet, equivalent to US label) [3]. The lack of paediatric medicines is particularly concerning for neonates and in serious and life threatening diseases [4, 6].
The main consequence of this situation is the widespread off-label use in paediatrics, especially in the case of old drugs that have never received a paediatric authorisation. The paediatric ‘off-label use’ specifically refers to ‘all paediatric uses of a marketed drug not detailed in the SPC’ [18]. The off-label paediatric use in Europe accounts for 45–60 % of the total number of prescriptions with rates of up to 90 % in the premature and term neonates, infants and paediatric patients admitted to intensive care units [11].
Moreover, it is well known that studies and trials involving children are affected by many methodological [1] and ethical concerns [17] as well as by economic barriers [13] resulting in difficulties to perform high-quality paediatric studies [5, 22], compliant with the existing guidelines and recommendations for high-quality paediatric studies [10, 14]. One barrier is the lack of incentives for companies to develop medicines that contain an off-patent active pharmaceutical ingredient. The entry into force of the Paediatric Regulation in 2007 (European Commission (EC) 1901/2006 as amended [8]) gave an important contribution to reduce the above-mentioned barriers and to support the development of medicines for children. Among other provisions, the Paediatric Regulation introduced a specific measure to favour work on off-patent medicines, the Paediatric Use Marketing Authorisation (the so called PUMA). This grants a 10-year period of data exclusivity in case of paediatric development of adult medicines that are not protected by a patent or supplementary protection certificate (off-patent drugs). A PUMA application should include the submission of paediatric data in accordance with an agreed Paediatric Investigation Plan (PIP) [9]. According to Article 40 of the Regulation, the European Research Framework Programmes should reserve funds to support PUMAs in case of off-patent drugs recognised as of high therapeutic interest for children and included in a ‘priority list’ (PL) adopted, on annual basis, by the European Medicines Agency (EMA) through its Paediatric Committee. In the last 6 years, such EC funds have been delivered through the Seventh Framework Programme for Research (FP7-FRP). In particular with reference to HEALTH-(2007–2013) Programme area, five calls for proposal have been released with reference to the topic 4.2-1 ‘to develop off-patent medicinal products for the paediatric population’.
These specific calls are characterized by the fact that they should respond both to the criteria for scientific excellence stated in the FP7 EC Programmes and meet standards for high-quality paediatric research as prescribed by the Paediatric Regulation (that is implementing paediatric studies to be conducted according to an agreed PIP). They should also stimulate research capacity and support the broader EU commitment to small-to-medium-sized enterprises (SMEsFootnote 1).
This paper aims to describe the paediatric projects funded in the framework of the Paediatric Regulation and evaluate their capacity to improve public health by meeting the policy drivers that justified the funding for supporting research capacity, innovation from SMEs and high-quality paediatric studies that contribute to the development of medicines for children and progress towards PUMAs.
Methodology
Data sources
To collect information on the FP7 Paediatric funded projects (content and status), the following sources have been consulted:
-
(a)
Community Research and Development Information Service (CORDIS) database, available from http://cordis.europa.eu/home_en.html
-
(b)
Project websitesFootnote 2
-
(c)
EC-EMA websites: Priority list of off-patent medicines, different versions developed from 2007 to 2013
-
(d)
EMA: Published Paediatric Committee (PDCO) decisions on Paediatric Investigation Plans (PIPs), available from http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/landing/pip_search.jsp&mid=WC0b01ac058001d129
-
(e)
The European Clinical Trials Register, available from https://www.clinicaltrialsregister.eu/ctr-search/search
-
(f)
The global database of clinical trials ClinicalTrials.gov, available from http://clinicaltrials.gov/
-
(g)
Scientific publications, conference presentations and meeting reports related to the projects, where available
-
(h)
EuOrphan, a database of orphan drugs designed/approved in Europe and in the USA [15]
Information collected
-
Funded projects and economic information (the total costs of the projects and the funds received by the European Commission);
-
General information on the Research Consortia established in the framework of these projects;
-
Information on the investigated active substance(s) or medicinal products, indication(s) and therapeutic area(s);
-
Information on the specific measures and requirements included in the project:
-
Obligation to submit a Paediatric Investigation Plan;
-
Presence of studies to develop age-appropriate formulations (new preparations of the drugs) or forms of medicines (new dosages and strengths);
-
Subgroups of paediatric population;
-
-
Trials and other studies (number, type and study characteristics).
Data were validated, and incomplete or inaccurate data have been sought, by e-mail contacts with the coordinators of the projects.
Results
Description of funded projects
Projects
According to the CORDIS database and other EC sources [7], in the period of 2006–2012, five calls have been launched in the context of the FP7 programme with reference to topic 4.2-1 ‘to develop off-patent medicinal products for the paediatric population’. Under the framework of these calls, a total of 76 proposals have been submitted and 20 have been funded with a success rate of 26.3 %. This analysis describes the 20 projects, listed in Table 1, devoted to paediatric studies to support a PUMA. The total amount awarded to these projects is 98.6 million euros (see Table 1).
Active substances
The 20 approved projects are investigating a total of 24 active substances, 8 of which (methotrexate, 6-mercaptopurine, ciprofloxacin, budesonide, doxorubicin, deferiprone, hydrocortisone and clonidine) have been granted a European Orphan Drug (OD) designation. In four cases (methotrexate, 6-mercaptopurine, hydrocortisone and deferiprone) the orphan conditions matches the Priority List indications (acute lymphoblastic leukaemia for the first two, adrenal insufficiency and sickle cell disease, respectively).
The substances are intended to treat a total of 22 paediatric indications in 10 therapeutic areas (Table 2). The most represented area is ‘infection’ accounting for four projects.
Research capacity
The 20 Research Consortia generated by the projects encompass 246 European and non-European institutions. The smallest Consortium includes four partners; the biggest one includes 18 participant members (Table 1). In addition, an average number of 6–8 investigational centres are included as third parties in each project. A total of 29 countries (22 European Member States and 7 non-European Member States) are involved with the UK, France, Italy and Germany being the most frequently represented, both in terms of number of participants and number of projects (Fig. 1). Four projects (HIP trial, DEEP, NEO-CIRC and GAPP) also include non-EU countries in their partnership. France and Italy together account for 50 % of coordinators (respectively, six and four) followed by UK and Germany (three projects each).
Most of the partners are universities and public-funded national research centres or institutions, 51 are private companies of which almost 40 are SMEs. Seven out of 20 project consortia (03K, DEEP, EPOC, TAIN, CloSed, GAPP and NEO-CIRC) include a patient association, while 9 out of 20 project consortia (NEuroSIS, EPOC, DEEP, NeoMero, PERS, LENA, GAPP, NeoVanc, CloSed) include a not-for-profit research organisation, as member or coordinator.
Paediatric studies that contribute to the development of medicines for children
New forms/formulations development
Eighty percent of the projects include studies to develop new age-appropriate formulations or dosage form: ten oral new formulations (six liquid, three tablets soluble or effervescent and one granule), seven new intravenous formulations and three intravenous dosage forms.
Subgroups of paediatric population involved in clinical trials
All paediatric subgroups are represented in the clinical trials (Fig. 2). In particular, 17 out of 29 paediatric clinical trials include preterm and/or term newborns. Projects NeoOpioid, NEuroSIS, NEMO, TINN and TINN2, HIP trial, NEO-CIRC and NeoVanc specifically address neonatal condition and for this reason patients to be enrolled in clinical trials are only preterm/term newborns.
Number and type of studies
A total of 71 studies are to be completed by the end of the 20 projects. They include studies in healthy adult volunteers (3), formulation development (20), clinical trials in children (29), non-interventional studies (6), in silico modelling (10) and non-clinical studies in animals (3).
Two out of three studies planned in healthy adult volunteers were PK, dose ranging and safety and one was a PK study. On a total of 29 paediatric trials, 16 were randomised controlled, eight on clinical pharmacology (PK/pharmacodynamics (PD)/dose finding), 9 non-randomised efficacy and/or safety and 12 were PK/PD/efficacy/safety. The estimated enrolment for all projects is 7,300 children. A pharmacogenetic sub-study is foreseen in 13 clinical trials (Table 3). Almost all trials are multicentre, involving 387 investigational sites, both in European and non-European countries. Thirteen of the paediatric trials are registered on the European Clinical Trials Database (EudraCT).
Start of the studies and patients’ enrolment
Enrolment of participants has been evaluated in 15 of 20 projects, corresponding to 23 clinical trials (5 projects, receiving approval less than a year before the survey, have been excluded from this analysis). At the time of this review (July 2014), a total of 1,400 paediatric patients (equal to 22.4 % of the estimated enrolment for all 15 projects) have been included in trials. This relates to five completed and eight ongoing trials. Ten additional trials have concluded the approval procedures and are in the process of opening.
Support for SMEs
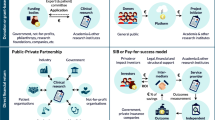
The partnership includes 51 private companies of which 13 are pharmaceutical companies and at least 40 meet the definition of an SME according to the information available to us. The projects are mainly coordinated by universities or other public Institutions while companies and not-for-profit research organisations are involved as coordinators in two and four projects, respectively. There are a significant number of private–public partnerships which would not have happened in the absence of pump-priming funding.
Paediatric Investigation Plans
Fifteen approved PIPs referring to 14 projects are available, while two projects (TINN and LENA) have submitted a PIP application but not yet received an approved plan. As indicated in Fig. 3, no PIP has been completed so far, and the completion date of the paediatric developmental plan ranges from January 2015 to end of 2018. No deferrals are foreseen in the plans. During the PIP approval process, some changes in the projects have been performed on PDCO request. These changes have included new measures and patient populations, additional trials or studies, sample size revision, modified statistical plan, and, more frequently, additional or modified paediatric indications (Table 2). These changes were mainly prompted by valuable scientific or regulatory concerns and were accordingly integrated in the projects. However, the need to come back for approval to the EC also influenced the timing and the complexity of the projects performance. In addition, even if new measures were requested by the PDCO, no additional funds were made available by the funding authority.
Progress towards PUMAs
PUMA applications require a PIP to be agreed, completion of all measures according to the agreed plan and confirmation of compliance with the agreed PIP. According the timelines of the FP7 projects, all of these steps should be completed within 5 years. While some projects may need extension, all of the studies will be conducted in this short period. This is different from the approach taken by many commercially funded PIPs. According to the 5-year report on the impact of the Paediatric Regulation drafted in 2012 by the EMA-PDCO [13], the number of PIPs in which studies are deferred is very high with 44 % of the approved PIPs not progressing as planned [12].
Discussion and conclusions
Data collected and described provide evidence on how these 20 FP7 approved project are contributing to the success of the Paediatric Regulation which entered into force in 2007 and aimed to overcome the existing methodological and ethical issues affecting research in the paediatric population.
Although many of the projects are still ongoing, these results allow us to discuss the many positive achievements obtained until now. Thanks to Article 40 of the Paediatric Regulation mandate, significant funds to support drug development in children have been provided. The FP7 paediatric projects have received a total of 98.6 million euros (representing 15 % of all the EC investments for research projects related to child health in the considered time period) [7] to conduct a total of 71 paediatric studies including 32 clinical trials, corresponding to an average of only 1.4 million euros for each study or trial. This average value does not differ from similar projects funded in the FP7 [7] or other projects, such as Innovative Medicines Initiative (IMI) [16]).
However, the funding scheme of the described projects, devoted to cover the development of drugs in order to put them on the market, is a novelty for Europe considering that before FP7, regulatory clinical trials were not included in the projects funded by the commission. However, it seems that the paediatric consortia generated by the FP7 paediatric projects are conducting these studies and trials using a limited amount of money in comparison with the recognised cost of paediatric trials in an approved PIP which is estimated to be three to four times higher [2, 21]. In addition, in a very similar programme set up in the USA, the Paediatric Trials Network has received US$95 million to support 16 paediatric clinical trials, corresponding to 4.3 million euros for each trial [19]. Although it is difficult to compare the costs of conducting clinical studies in the USA and in Europe, since the payment system for investigators and researchers is different, it seems that the research consortia involved in these projects are able to manage good clinical trials with a reduced amount of resources. If this is the case, it suggests that this funding stream has unlocked a great deal of enthusiasm among child health professionals, who are also contributing to the projects with their time and other resources, while providing enough money to encourage institutions and SMEs to participate.
A very large scientific community (246 partners organisations and hundreds recruiting centres including academic, research organisations and public hospitals) covering EU and non-EU countries has been mobilised. Recent literature data demonstrated a very low involvement of researchers from academic or public research institutions in paediatric research in Europe compared with the USA [2]. The paediatric consortia born within these projects represent a critical mass of competencies that is also attracting public and private companies, scientific societies and patients’ organisations. The effect of this networking action is crucial and destined to last well beyond the end of the single research project. At present, each consortium has a very high risk to disintegrate at the end of the funding period. Thus, there is a need to sustain this capacity. The role of SMEs is striking and it is important that these and other SMEs are not discouraged from contributing to this type of work in future.
Eighty percent of the projects are developing new formulations and dosage forms of medicines specifically for the paediatric population. Age-appropriate formulations are an urgent need [20]. The 5-year report drafted in 2012 by the EMA-PDCO states that, although for authorised medicines, 26 new pharmaceutical forms were authorised for paediatric use since the entry of the Paediatric Regulation, a lack of age-appropriate formulations, in terms of safety of excipients, palatability, acceptability, dosing flexibility, accuracy and practical handling still exist [13].
The rich variety of study designs which have been adopted following peer review and detailed scrutiny by EMA suggests that high-quality studies have been tailored to the specific situations. In addition pharmacogenetic substudies intended to correlate the different pharmacological response to genetic variability are strongly represented as a fundamental step in the way to identify rational drug dosing.
The 29 ongoing paediatric trials represent a valuable percentage (14 %) of all the paediatric trials included in the EudraCT database with reference to an approved PIP, and the number of patients recruited or to be recruited (around 7,300 representing 23 % of all the paediatric patients included in clinical trials in Europe from 2007 to 2011 [13]) is highly remarkable. Furthermore, to date, 22 % of the projected enrolment for these trials is complete even though the majority of trials are yet to open. This is in contrast with the reported low recruitment capacity and difficulties with the conduct of paediatric trials [21, 22] and again demonstrates the relevance of the FP7 projects in the contest of paediatric research. The inclusion of neonates and younger paediatric subgroups is particularly important, and in contrast with the historic deficiency of medicine development in these populations. Finally, the strong engagement of commercial partners and sponsors should stimulate a critical appraisal of the PUMA concept. Commercial entities are clearly enthusiastic about developing medicines for children, but so far, only 2 PUMAs have been approved out of more than 1,000 PIPs. The PUMA is not an attractive incentive for companies [13], and in particular, SMEs, to facilitate the development and repurposing of marketed drugs for children. Developing a more appropriate incentive is of fundamental relevance for the future, since companies should be encouraged to invest in this sector. The fee reduction policy for SMEs that has been established and periodically renewed by the EMA, a larger use of free charge regulatory consultation for SME, the availability of large paediatric research infrastructures like what provided by the EnpREMA can be considered as promising instruments to increase the commitment of companies in this field.
To complete the picture provided by this analysis, some critical aspects should be discussed. As underlined in the introduction, the challenging issue for FP7 paediatric projects is to respond to different requirements imposed by the Research Programmes framework (deadline, limited resources, scientific publications, etc.) and by the Paediatric Regulation (PIP should be agreed, all the paediatric population should be covered, unmet paediatric needs prevail over scientific interest). The recommendations of the PDCO mean that relevant differences can be created from the original project mainly in term of (a) number of studies, (b) patients populations, (c) paediatric indications and (d) studies design. For these reasons, the implementation of PIPs has represented a critical point in the framework of these projects causing prolongation of the contractual procedures with the EC and often, delays in the start of the studies.
A further potential weakness is represented by time and trials management. As a result of the complexity of the administrative and ethic procedures for trial approval, each consortium has experienced the need to address specific regulatory and organisation activities that are usually outside the fields of competence of the academic and not-for-profit research groups. These activities include structured interoperability among participants, standard operating procedures to be adopted, to adhere to GCP and the requirements of trials included in the applications for MA, ethical, administrative and contractual requirements, etc.
Our results demonstrate that the majority of these challenges can be positively addressed and mainly resolved. A valid regulatory expertise seems to have been incorporated in many FP7 Paediatric Consortia that demonstrate the ability to mobilise a large scientific and clinical community. These private–public partnerships have devised clinical development plans and have conducted paediatric clinical studies that are acceptable to regulators and Ethics Committees. However, further efforts are needed, such as high level educational activities addressed to researchers and health professionals involved in paediatric trials and an increased active collaboration and resources exchange among academics, health care professionals, regulatory bodies and industry. Our expectation is that the Global Research in Paediatric (GRiPFootnote 3), implementing an infrastructure matrix to stimulate and facilitate the development and safe use of medicines in children will provide a fundamental contribution in both these fields.
In conclusion, despite the reported difficulties and limitations, these projects are successful in many ways, are meeting the expectations of the European Commission and of the evolving Paediatric Regulation aimed to improve the health of children and led to functional paediatric drug development pathways [23]. However, much work remains to be done to make the most of the opportunities provided by these common regulatory perspectives. Therefore, new initiatives are required that will consolidate the experience and communities which have been developed by the FP7 projects. On one hand, expanded funding programmes for paediatric medicines should be made available in “Horizon 2020” or other EC Research Funds, including the Innovative Medicine Initiative (IMI) project, to meet the mandate from the European Parliament provided by Paediatric Regulation Article 40. On the other hand, it could be important that government and regulatory institutions provide the right framework allowing companies to be rewarded for their investments in paediatric clinical research.
Progress to date suggests that further work to develop medicines for children through EC-funded private–public partnerships will be productive, will provide value for money and will continue to improve public health.
Notes
The category of micro, small and medium-sized enterprises (SMEs) is made up of enterprises which employ fewer than 250 persons (50 and 10 in case of small and micro, respectively) and which have an annual turnover not exceeding 50 million euros, and/or an annual balance sheet total not exceeding 43 million euros (Commission Recommendation of 6 May 2003 concerning the definition of micro, small and medium-sized enterprises, notified under document number C(2003) 1422) (2003/361/EC)
Not all the project websites provide the same amount of information and their update is not homogeneous
Abbreviations
- CORDIS:
-
Community Research and Development Information Service database
- EC:
-
European Commission
- EMA:
-
European Medicines Agency
- EU:
-
European
- FP7-FRP:
-
Seventh Framework Programme for Research
- IMI:
-
Innovative Medicines Initiative
- Non-EU:
-
Non-European
- OD:
-
Orphan drug
- PD:
-
Pharmacodynamics
- PDCO:
-
Paediatric Committee
- PIP:
-
Paediatric Investigation Plan
- PK:
-
Pharmacokinetics
- PUMA:
-
Paediatric Use Marketing Authorisation
- SME:
-
Small-to-medium-sized enterprise
- SPC:
-
Summary of Product Characteristics
References
Baiardi P, Giaquinto C, Girotto S, Manfredi C, Ceci A (2001) Innovative study design for paediatric clinical trials. Eur J Clin Pharmacol 1:109–115. doi:10.1007/s00228-011-0990-y
Baker-Smith CM, Benjamin DK Jr, Grabowski HG, Reid ED, Mangum B, Goldsmith JV, Murphy MD, Edwards R, Eisenstein EL, Sun J, Califf RM, Li JS (2008) The economic returns of pediatric clinical trials of anti-hypertensive drugs. Am Heart J 156:682–688. doi:10.1016/j.ahj.2008.05.001
Ceci A, Felisi M, Baiardi P, Bonifazi F, Catapano M, Giaquinto C, Nicolosi A, Sturkenboom M, Neubert A, Wong I (2006) Medicines for children licensed by the European Medicines Agency (EMEA): the balance after 10 years. Eur J Clin Pharmacol 62:947–952. doi:10.1007/s00228-006-0193-0
Ceci A, Felisi M, Catapano M, Baiardi P, Cipollina L, Ravera S, Bagnulo S, Reggio S, Rondini G (2002) Medicines for children licensed by the European Agency for the Evaluation of Medicinal Products. Eur J Clin Pharmacol 58:495–500. doi:10.1007/s00228-002-0511-0
Cohen E, Uleryk E, Jasuja M, Parkin PC (2007) An absence of pediatric randomized controlled trials in general medical journals, 1985–2004. J Clin Epidemiol 60:118–123. doi:10.1016/j.jclinepi.2006.03.015
Conroy S, McIntyre J (2005) The use of unlicensed and off-label medicines in the neonate. Semin Fetal Neonatal Med 10:115–122. doi:10.1016/j.siny.2004.11.003
Costescu A (2013) Off-patent Paediatric Medicines-7th call Overview of FP7-Funding HORIZON 2020 state of play. Proceedings of the Sixth Foresight Training Course “Biotech and Innovative Science to meet patient needs”, 30th September-2nd October 2013
European Commission (2006) Regulation No 1901/2006 of the European Parliament and of the Council of 12 December 2006 on medicinal products for paediatric use and amending Regulation (EEC) No 1768/92, Directive 2001/20/EC, Directive 2001/83/EC and Regulation (EC) No 726/2004. Official Journal L 378, 1–19
European Commission (2008) Communication from the Commission Guideline on the format and content of applications for agreement or modification of a paediatric investigation plan and requests for waivers or deferrals and concerning the operation of the compliance check and on criteria for assessing significant studies (2008/C 243/01)
European Commission (2008) Ethical considerations for clinical trials on medicinal products conducted with the paediatric population—recommendations of the ad hoc group for the development of implementing guidelines for Directive 2001/20/EC relating to good clinical practice in the conduct of clinical trials on medicinal products for human use.
European Medicine Agency (2010) Report on the survey of all paediatric uses of medicinal products in Europe established according to article 42–43 of Regulation (EC) No 1901/2006 of the European Parliament and of the Council on medicinal products for Paediatric use EMA/794083/2009
European Medicine Agency (2013) Report to the European Commission on companies and products that have benefited from any of the rewards and incentives in the Paediatric Regulation and on the companies that have failed to comply with any of the obligations in this Regulation EMA/328413/2013 30 April 2013
European Medicine Agency /Paediatric Committee (2012) 5-year Report to the European Commission General report on the experience acquired as a result of the application of the Paediatric Regulation EMA/428172/2012
European Medicine Agency Topic E 11 Clinical investigation of medicinal products in the paediatric population note for guidance on clinical investigation of medicinal products in the paediatric population (CHMP/ICH/2711/99)
Giannuzzi V, Bartoloni F, Bonifazi F, Conte R, Felisi M, Ruggieri L, Baiardi P, Ceci “Why developing an orphan drug registry?” Official documents of the European Platform for Rare Disease second workshop “Rare disease and orphan drug registries”, Istituto Superiore di Sanità, Rome (2013) 21–22 October 2013 http://www.iss.it/binary/pres/cont/2ndWorkshop_EPIRARE_AbstractBook.pdf
Innovative Medicines Initiative, IMI Ongoing Projects, Available from: http://www.imi.europa.eu/content/ongoing-projects
Knellwolf AL, Bauzon S, Della Casa Alberighi O, Lutsar I, Bácsy E, Alfarez D, Panei P (2011) Framework conditions facilitating paediatric clinical research. Ital J Pediatr 37:12
Neubert A, Wong IC, Bonifazi A, Catapano M, Felisi M, Baiardi P, Giaquinto C, Knibbe CA, Sturkenboom MC, Ghaleb MA, Ceci A (2008) Defining off-label and unlicensed use of medicines for children: results of a Delphi survey. Pharmacol Res 58:316–322. doi:10.1016/j.phrs.2008.09.007
Pediatric trials network new national initiative to rigorously study pediatric dosing safety of prescription medicines. Accessed 15 Feb 2014 Available from: https://pediatrictrials.org/
Richey RH, Shah UU, Peak M, Craig JV, Ford JL, Barker CE, Nunn AJ, Turner MA (2013) Manipulation of drugs to achieve the required dose is intrinsic to paediatric practice but is not supported by guidelines or evidence. BMC Pediatr 13:81. doi:10.1186/1471-2431-13-81
Smit-Marshall P (2010) Pediatric trials: a worldview. Appl Clin Trials 19:1
Thomson D, Hartling L, Cohen E, Vandermeer B, Tjosvold L, Klassen TP (2010) Controlled trials in children: quantity, methodological quality and descriptive characteristics of pediatric controlled trials published 1948–2006. PLoS One 5(9):e13106. doi:10.1371/journal.pone.0013106
Turner MA, Catapano M, Hirschfeld S, Giaquinto C (2014) Paediatric drug development: the impact of evolving regulations. Adv Drug Deliv Rev 73:2–13. doi:10.1016/j.addr.2014.02.003
Acknowledgments
The research leading to these results has received funding from the European Union Seventh Framework Programme FP7/2007–2013 under grant agreement no. 261060. The authors acknowledge Angelique Aschrafi, Hugo Lagercrantz, Gene Dempsey, Christian F. Poets and all the others coordinators/reference contacts of the projects for participating to the survey for data collection.
Authors’ contribution
L. Ruggieri collected data, figures, and drafted the manuscript.
V. Giannuzzi revised the manuscript.
P. Baiardi revised the manuscript.
F. Bonifazi revised the manuscript.
E.H. Davies collaborated with the drafting of the manuscript.
C. Giaquinto revised the manuscript.
D. Bonifazi commented on and revised the final manuscript and provided information on the DEEP project.
M. Felisi commented on and revised the final manuscript and provided information on the DEEP project.
C. Chiron commented on and revised the final manuscript and provided information on the Kiekids project.
R. Pressler commented on and revised the final manuscript and provided information on the NEMO project.
H. Rabe commented on and revised the final manuscript and provided information on the NEO-CIRC project.
M. J. Whitaker commented on and revised the final manuscript and provided information on the TAIN project.
A. Neubert commented on and revised the final manuscript and provided information on the CloSed project.
E. Jacqz-Aigrain commented on and revised the final manuscript and provided information on the TINN and TINN-2 projects.
I. Eichler commented on and revised the final manuscript.
M.A. Turner collaborated with the drafting of the manuscript and did major manuscript revisions.
A. Ceci did the data analysis, study design and manuscript drafting.
Conflict of interest
The authors declare that they have no conflicts of interest in the research.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
Communicated by Beat Steinmann
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Ruggieri, L., Giannuzzi, V., Baiardi, P. et al. Successful private–public funding of paediatric medicines research: lessons from the EU programme to fund research into off-patent medicines. Eur J Pediatr 174, 481–491 (2015). https://doi.org/10.1007/s00431-014-2398-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-014-2398-z