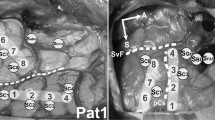
Abstract
Bipolar direct electrical stimulation (DES) of an awake patient is the reference technique for identifying brain structures to achieve maximal safe tumor resection. Unfortunately, DES cannot be performed in all cases. Alternative surgical tools are, therefore, needed to aid identification of subcortical connectivity during brain tumor removal. In this pilot study, we sought to (i) evaluate the combined use of evoked potential (EP) and tractography for identification of white matter (WM) tracts under the functional control of DES, and (ii) provide clues to the electrophysiological effects of bipolar stimulation on neural pathways. We included 12 patients (mean age of 38.4 years) who had had a dMRI-based tractography and a functional brain mapping under awake craniotomy for brain tumor removal. Electrophysiological recordings of subcortical evoked potentials (SCEPs) were acquired during bipolar low frequency (2 Hz) stimulation of the WM functional sites identified during brain mapping. SCEPs were successfully triggered in 11 out of 12 patients. The median length of the stimulated fibers was 43.24 ± 19.55 mm, belonging to tracts of median lengths of 89.84 ± 24.65 mm. The electrophysiological (delay, amplitude, and speed of propagation) and structural (number and lengths of streamlines, and mean fractional anisotropy) measures were correlated. In our experimental conditions, SCEPs were essentially limited to a subpart of the bundles, suggesting a selectivity of action of the DES on the brain networks. Correlations between functional, structural, and electrophysiological measures portend the combined use of EPs and tractography as a potential intraoperative tool to achieve maximum safe resection in brain tumor surgery.







Similar content being viewed by others
Data availability
The anonymized data acquired during this protocol consisting of preoperative MRIs and intraoperative SCEPs are available upon request to any qualified investigator after completion of a data sharing agreement. The source code for all data processing is freely available at https://doi.org/10.5281/zenodo.6784185.
References
Almairac F, Herbet G, Moritz-Gasser S et al (2015) The left inferior fronto-occipital fasciculus subserves language semantics: a multilevel lesion study. Brain Struct Funct 220:1983–1995. https://doi.org/10.1007/s00429-014-0773-1
Andersson JLR, Sotiropoulos SN (2016) An integrated approach to correction for off-resonance effects and subject movement in diffusion MR imaging. Neuroimage 125:1063–1078. https://doi.org/10.1016/j.neuroimage.2015.10.019
Bassett DS, Bullmore ET (2017) Small-world brain networks revisited. Neuroscientist 23:499–516. https://doi.org/10.1177/1073858416667720
Beez T, Boge K, Wager M et al (2013) Tolerance of awake surgery for glioma: a prospective European Low Grade Glioma Network multicenter study. Acta Neurochir (wien) 155:1301–1308. https://doi.org/10.1007/s00701-013-1759-0
Benedictis DE, Marras A, Petit CE, Sarubbo L, S, (2021) The inferior fronto-occipital fascicle: a century of controversies from anatomy theaters to operative neurosurgery. J Neurosurg Sci 65:605–615
Boyer A, Duffau H, Mandonnet E et al (2020) Attenuation and delay of remote potentials evoked by direct electrical stimulation during brain surgery. Brain Topogr 33:143–148. https://doi.org/10.1007/s10548-019-00732-w
Boyer A, Stengel C, Bonnetblanc F et al (2021) Patterns of axono-cortical evoked potentials: an electrophysiological signature unique to each white matter functional site? Acta Neurochir (wien) 163:3121–3130. https://doi.org/10.1007/s00701-020-04656-4
Bullmore E, Sporns O (2009) Complex brain networks: graph theoretical analysis of structural and functional systems. Nat Rev Neurosci 10:186–198. https://doi.org/10.1038/nrn2575
Calabrese E, Badea A, Cofer G et al (2015) A diffusion MRI tractography connectome of the mouse brain and comparison with neuronal tracer data. Cereb Cortex 25:4628–4637. https://doi.org/10.1093/cercor/bhv121
Calamuneri A, Arrigo A, Mormina E et al (2018) White matter tissue quantification at low b-values within constrained spherical deconvolution framework. Front Neurol 9:716. https://doi.org/10.3389/fneur.2018.00716
Catani M, Thiebaut de Schotten M (2008) A diffusion tensor imaging tractography atlas for virtual in vivo dissections. Cortex 44:1105–1132. https://doi.org/10.1016/j.cortex.2008.05.004
Conner CR, Ellmore TM, DiSano MA et al (2011) Anatomic and electro-physiologic connectivity of the language system: a combined DTI-CCEP study. Comput Biol Med 41:1100–1109. https://doi.org/10.1016/j.compbiomed.2011.07.008
Crowther LJ, Brunner P, Kapeller C et al (2019) A quantitative method for evaluating cortical responses to electrical stimulation. J Neurosci Methods 311:67–75. https://doi.org/10.1016/j.jneumeth.2018.09.034
Dalboni da Rocha JL, Bramati I, Coutinho G et al (2020) Fractional anisotropy changes in Parahippocampal cingulum due to alzheimer’s disease. Sci Rep 10:2660. https://doi.org/10.1038/s41598-020-59327-2
De Witt Hamer PC, Robles SG, Zwinderman AH et al (2012) Impact of intraoperative stimulation brain mapping on glioma surgery outcome: a meta-analysis. J Clin Oncol 30:2559–2565. https://doi.org/10.1200/JCO.2011.38.4818
Duffau H (2005) Lessons from brain mapping in surgery for low-grade glioma: insights into associations between tumour and brain plasticity. Lancet Neurol 4:476–486. https://doi.org/10.1016/S1474-4422(05)70140-X
Duffau H (2008) The anatomo-functional connectivity of language revisited. New insights provided by electrostimulation and tractography. Neuropsychologia 46:927–934. https://doi.org/10.1016/j.neuropsychologia.2007.10.025
Duffau H (2015) Stimulation mapping of white matter tracts to study brain functional connectivity. Nat Rev Neurol 11:255–265. https://doi.org/10.1038/nrneurol.2015.51
Enatsu R, Kanno A, Ohtaki S et al (2016) Intraoperative subcortical fiber mapping with subcortico-cortical evoked potentials. World Neurosurg 86:478–483. https://doi.org/10.1016/j.wneu.2015.10.043
Filipiak P, Almairac F, Papadopoulo T et al (2020) Towards linking diffusion MRI based macro- and microstructure measures with cortico-cortical transmission in brain tumor patients. Neuroimage 226:117567. https://doi.org/10.1016/j.neuroimage.2020.117567
Friederici AD (2015) White-matter pathways for speech and language processing. Handb Clin Neurol 129:177–186. https://doi.org/10.1016/B978-0-444-62630-1.00010-X
Gil-Robles S, Duffau H (2010) Surgical management of World Health Organization Grade II gliomas in eloquent areas: the necessity of preserving a margin around functional structures. Neurosurg Focus 28:E8. https://doi.org/10.3171/2009.12.FOCUS09236
Gramfort A, Luessi M, Larson E et al (2013) MEG and EEG data analysis with MNE-Python. Front Neurosci 7:267. https://doi.org/10.3389/fnins.2013.00267
Gras-Combe G, Moritz-Gasser S, Herbet G, Duffau H (2012) Intraoperative subcortical electrical mapping of optic radiations in awake surgery for glioma involving visual pathways. J Neurosurg 117:466–473. https://doi.org/10.3171/2012.6.JNS111981
Henderson F, Abdullah KG, Verma R, Brem S (2020) Tractography and the connectome in neurosurgical treatment of gliomas: the premise, the progress, and the potential. Neurosurg Focus 48:E6. https://doi.org/10.3171/2019.11.FOCUS19785
Herbet G, Duffau H (2020) Revisiting the functional anatomy of the human brain: toward a meta-networking theory of cerebral functions. Physiol Rev 100:1181–1228. https://doi.org/10.1152/physrev.00033.2019
Herbet G, Moritz-Gasser S, Lemaitre A-L et al (2019) Functional compensation of the left inferior longitudinal fasciculus for picture naming. Cogn Neuropsychol 36:140–157. https://doi.org/10.1080/02643294.2018.1477749
Herrick IA, Craen RA, Gelb AW et al (1997) Propofol sedation during awake craniotomy for seizures: electrocorticographic and epileptogenic effects. Anesth Analg 84:1280–1284
Hervey-Jumper SL, Berger MS (2016) Maximizing safe resection of low- and high-grade glioma. J Neurooncol 130:269–282. https://doi.org/10.1007/s11060-016-2110-4
Jellison BJ, Field AS, Medow J et al (2004) Diffusion tensor imaging of cerebral white matter: a pictorial review of physics, fiber tract anatomy, and tumor imaging patterns. AJNR Am J Neuroradiol 25:356–369
Jenkinson M, Smith S (2001) A global optimisation method for robust affine registration of brain images. Med Image Anal 5:143–156. https://doi.org/10.1016/s1361-8415(01)00036-6
Jenkinson M, Bannister P, Brady M, Smith S (2002) Improved optimization for the robust and accurate linear registration and motion correction of brain images. Neuroimage 17:825–841. https://doi.org/10.1016/s1053-8119(02)91132-8
Jeurissen B, Tournier J-D, Dhollander T et al (2014) Multi-tissue constrained spherical deconvolution for improved analysis of multi-shell diffusion MRI data. Neuroimage 103:411–426. https://doi.org/10.1016/j.neuroimage.2014.07.061
Jones SE, Zhang M, Avitsian R et al (2014) Functional magnetic resonance imaging networks induced by intracranial stimulation may help defining the epileptogenic zone. Brain Connect 4:286–298. https://doi.org/10.1089/brain.2014.0225
Kamali A, Flanders AE, Brody J et al (2014) Tracing superior longitudinal fasciculus connectivity in the human brain using high resolution diffusion tensor tractography. Brain Struct Funct 219:269–281. https://doi.org/10.1007/s00429-012-0498-y
La Corte E, Eldahaby D, Greco E et al (2021) The Frontal aslant tract: a systematic review for neurosurgical applications. Front Neurol 12:641586. https://doi.org/10.3389/fneur.2021.641586
Lewis JD, Theilmann RJ, Sereno MI, Townsend J (2009) The relation between connection length and degree of connectivity in young adults: a DTI analysis. Cereb Cortex 19:554–562. https://doi.org/10.1093/cercor/bhn105
Louis DN, Perry A, Wesseling P et al (2021) The 2021 WHO classification of tumors of the central nervous system: a summary. Neuro Oncol 23:1231–1251. https://doi.org/10.1093/neuonc/noab106
Lucena O, Vos SB, Vakharia V et al (2021) Enhancing the estimation of fiber orientation distributions using convolutional neural networks. Comput Biol Med 135:104643. https://doi.org/10.1016/j.compbiomed.2021.104643
Maier-Hein KH, Neher PF, Houde J-C et al (2017) The challenge of mapping the human connectome based on diffusion tractography. Nat Commun 8:1349. https://doi.org/10.1038/s41467-017-01285-x
Mandonnet E, Dadoun Y, Poisson I et al (2016) Axono-cortical evoked potentials: a proof-of-concept study. Neurochirurgie 62:67–71. https://doi.org/10.1016/j.neuchi.2015.09.003
Martino J, da Silva-Freitas R, Caballero H et al (2013) Fiber dissection and diffusion tensor imaging tractography study of the temporoparietal fiber intersection area. Neurosurgery 72:87–97
Matsumoto R, Nair DR, LaPresto E et al (2004) Functional connectivity in the human language system: a cortico-cortical evoked potential study. Brain 127:2316–2330. https://doi.org/10.1093/brain/awh246
Morshed RA, Young JS, Lee AT, Hervey-Jumper SL (2021) Functional mapping for glioma surgery, part 2: intraoperative mapping tools. Neurosurg Clin N Am 32:75–81. https://doi.org/10.1016/j.nec.2020.09.001
Parker D, Ould Ismail AA, Wolf R et al (2020) Freewater estimatoR using iNtErpolated iniTialization (FERNET): Characterizing peritumoral edema using clinically feasible diffusion MRI data. PLoS ONE 15:e0233645. https://doi.org/10.1371/journal.pone.0233645
Piccioni F, Fanzio M (2008) Management of anesthesia in awake craniotomy. Minerva Anestesiol 74:393–408
Premoli I, Costantini A, Rivolta D et al (2017) The Effect of Lamotrigine and Levetiracetam on TMS-Evoked EEG responses depends on stimulation intensity. Front Neurosci 11:585. https://doi.org/10.3389/fnins.2017.00585
Rheault F, Poulin P, Valcourt Caron A et al (2020) Common misconceptions, hidden biases and modern challenges of dMRI tractography. J Neural Eng 17:011001. https://doi.org/10.1088/1741-2552/ab6aad
Salat DH, Tuch DS, van der Kouwe AJW et al (2010) White matter pathology isolates the hippocampal formation in Alzheimer’s disease. Neurobiol Aging 31:244–256. https://doi.org/10.1016/j.neurobiolaging.2008.03.013
Sarubbo S, De Benedictis A, Maldonado IL et al (2013) Frontal terminations for the inferior fronto-occipital fascicle: anatomical dissection, DTI study and functional considerations on a multi-component bundle. Brain Struct Funct 218:21–37. https://doi.org/10.1007/s00429-011-0372-3
Schilling KG, Rheault F, Petit L et al (2021) Tractography dissection variability: what happens when 42 groups dissect 14 white matter bundles on the same dataset? Neuroimage 243:118502. https://doi.org/10.1016/j.neuroimage.2021.118502
Schober P, Boer C, Schwarte LA (2018) Correlation coefficients: appropriate use and interpretation. Anesth Analg 126:1763–1768. https://doi.org/10.1213/ANE.0000000000002864
Scholz J, Tomassini V, Johansen-Berg H (2013) Individual Differences in White Matter Microstructure in the Healthy Brain. Diffusion MRI: From Quantitative Measurement to In vivo Neuroanatomy, 2nd edn. Elsevier, Amsterdam, pp 301–316
Seidel K, Szelényi A, Bello L (2022) Intraoperative mapping and monitoring during brain tumor surgeries. Handb Clin Neurol 186:133–149. https://doi.org/10.1016/B978-0-12-819826-1.00013-2
Silverstein BH, Asano E, Sugiura A et al (2020) Dynamic tractography: Integrating cortico-cortical evoked potentials and diffusion imaging. Neuroimage 215:116763. https://doi.org/10.1016/j.neuroimage.2020.116763
Suzuki Y, Enatsu R, Kanno A et al (2019) The Influence of Anesthesia on Corticocortical Evoked potential monitoring network between frontal and Temporoparietal cortices. World Neurosurg 123:e685–e692. https://doi.org/10.1016/j.wneu.2018.11.253
Szelényi A, Bello L, Duffau H et al (2010) Intraoperative electrical stimulation in awake craniotomy: methodological aspects of current practice. Neurosurg Focus 28:E7. https://doi.org/10.3171/2009.12.FOCUS09237
Tallus J, Mohammadian M, Kurki T et al (2023) A comparison of diffusion tensor imaging tractography and constrained spherical deconvolution with automatic segmentation in traumatic brain injury. Neurimage Clin 37:103284. https://doi.org/10.1016/j.nicl.2022.103284
Tournier J-D, Smith R, Raffelt D et al (2019) MRtrix3: a fast, flexible and open software framework for medical image processing and visualisation. Neuroimage 202:116137. https://doi.org/10.1016/j.neuroimage.2019.116137
Trebaul L, Deman P, Tuyisenge V et al (2018) Probabilistic functional tractography of the human cortex revisited. Neuroimage 181:414–429. https://doi.org/10.1016/j.neuroimage.2018.07.039
van den Heuvel MP, de Reus MA, Feldman Barrett L et al (2015) Comparison of diffusion tractography and tract-tracing measures of connectivity strength in rhesus macaque connectome. Hum Brain Mapp 36:3064–3075. https://doi.org/10.1002/hbm.22828
Veraart J, Novikov DS, Christiaens D et al (2016) Denoising of diffusion MRI using random matrix theory. Neuroimage 142:394–406. https://doi.org/10.1016/j.neuroimage.2016.08.016
Vincent M, Guiraud D, Duffau H et al (2017) Electrophysiological brain mapping: basics of recording evoked potentials induced by electrical stimulation and its physiological spreading in the human brain. Clin Neurophysiol 128:1886–1890. https://doi.org/10.1016/j.clinph.2017.07.402
Vincent MA, Bonnetblanc F, Mandonnet E et al (2020) Measuring the electrophysiological effects of direct electrical stimulation after awake brain surgery. J Neural Eng 17:016047. https://doi.org/10.1088/1741-2552/ab5cdd
Wycoco V, Shroff M, Sudhakar S, Lee W (2013) White matter anatomy: what the radiologist needs to know. Neuroimaging Clin N Am 23:197–216. https://doi.org/10.1016/j.nic.2012.12.002
Yamao Y, Matsumoto R, Kunieda T et al (2014) Intraoperative dorsal language network mapping by using single-pulse electrical stimulation. Hum Brain Mapp 35:4345–4361. https://doi.org/10.1002/hbm.22479
Yamao Y, Suzuki K, Kunieda T et al (2017) Clinical impact of intraoperative CCEP monitoring in evaluating the dorsal language white matter pathway. Hum Brain Mapp 38:1977–1991. https://doi.org/10.1002/hbm.23498
Yang JY-M, Yeh C-H, Poupon C, Calamante F (2021) Diffusion MRI tractography for neurosurgery: the basics, current state, technical reliability and challenges. Phys Med Biol. https://doi.org/10.1088/1361-6560/ac0d90
Funding
This study was supported and granted by the University Hospital of Nice (grant number: 19-AOIP-01).
Author information
Authors and Affiliations
Contributions
FA: Conceptualization, Methodology, Validation, Investigation, Resources, Writing—Original Draft, Writing—Review & Editing, Visualization, Supervision, Project administration, Funding acquisition. PI: Methodology, Software, Validation, Investigation, Writing—Original Draft, Writing—Review & Editing, Visualization. MO: Methodology, Investigation, Writing—Original Draft, Visualization. TPo: Methodology, Software, Validation, Investigation, Resources, Data Curation, Writing—Review & Editing. LM: Methodology, Validation, Investigation, Writing—Original Draft, Visualization. SCt: Validation, Investigation, Writing—Original Draft, Visualization. CFz: Investigation, Writing—Original Draft. MCc: Methodology, Software, Validation, Investigation, Resources, Data Curation. RD: Validation, Investigation, Resources, Data Curation, Writing—Review & Editing. DF: Conceptualization, Methodology, Investigation, Resources, Writing—Original Draft, Supervision. PF: Conceptualization, Methodology, Software, Validation, Investigation, Writing—Original Draft, Writing—Review & Editing, Visualization.
Corresponding author
Ethics declarations
Conflict of interests
The authors have no relevant financial or non-financial interests to disclose.
Ethics Approval
This study was approved by a French ethical committee and was registered in the Clinical Trials database (NCT04163315).
Consent to participate
All participating patients have freely given their written informed consent according to the Declaration of Helsinki.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Almairac, F., Isan, P., Onno, M. et al. Identifying subcortical connectivity during brain tumor surgery: a multimodal study. Brain Struct Funct 228, 815–830 (2023). https://doi.org/10.1007/s00429-023-02623-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-023-02623-0