Abstract
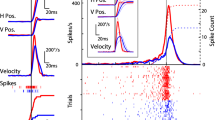
Brain state has profound effects on neural processing and stimulus encoding in sensory cortices. While the synchronized state is dominated by low-frequency local field potential (LFP) activity, low-frequency LFP power is suppressed in the desynchronized state, where a concurrent enhancement in gamma power is observed. Recently, it has been shown that cortical desynchronization co-occurs with enhanced between-trial reliability of spiking activity in sensory neurons, but it is currently unclear whether this effect is also evident in LFP signals. Here, we address this question by recording both spike trains and LFP in primary visual cortex during natural movie stimulation, and using isoflurane anesthesia and basal forebrain (BF) electrical activation as proxies for synchronized and desynchronized brain states. We show that indeed, low-frequency LFP modulations (“LFP events”) also occur more reliably following BF activation. Interestingly, while being more reliable, these LFP events are smaller in amplitude compared to those generated in the synchronized brain state. We further demonstrate that differences in reliability of spiking activity between cortical states can be linked to amplitude and probability of LFP events. The correlated temporal dynamics between low-frequency LFP and spiking response reliability in visual cortex suggests that these effects may both be the result of the same neural circuit activation triggered by BF stimulation, which facilitates switching between processing of incoming sensory information in the desynchronized and reverberation of internal signals in the synchronized state.





Similar content being viewed by others
References
Arieli A, Sterkin A, Grinvald A, Aertsen A (1996) Dynamics of ongoing activity: explanation of the large variability in evoked cortical responses. Science 273:1868–1871. doi:10.1126/science.273.5283.1868
Azouz R, Gray CM (1999) Cellular mechanisms contributing to response variability of cortical neurons in vivo. J Neurosci 19:2209–2223
Bair W, Zohary E, Newsome WT (2001) Correlated firing in macaque visual area MT: time scales and relationship to behavior. J Neurosci 21:1676–1697
Betsch BY, Einhauser W, Kording KP, Konig P (2004) The world from a cat’s perspective—statistics of natural videos. Biol Cybern 90:41–50. doi:10.1007/s00422-003-0434-6
Bhattacharyya A, Veit J, Kretz R, Bondar I, Rainer G (2013) Basal forebrain activation controls contrast sensitivity in primary visual cortex. BMC Neurosci 14:55. doi:10.1186/1471-2202-14-55
Brainard DH (1997) The psychophysics toolbox. Spatial Vis 10:433–436
Buzsaki G, Bickford RG, Ponomareff G, Thal LJ, Mandel R, Gage FH (1988) Nucleus basalis and thalamic control of neocortical activity in the freely moving rat. J Neurosci 8:4007–4026
Campagna JA, Miller KW, Forman SA (2003) Drug therapy: mechanisms of actions of inhaled anesthetics. N Engl J Med 348:2110–2124
Chen NY, Sugihara H, Sur M (2015) An acetylcholine-activated microcircuit drives temporal dynamics of cortical activity. Nat Neurosci 18:892-U340. doi:10.1038/nn.4002
Chisum HJ, Mooser F, Fitzpatrick D (2003) Emergent properties of layer 2/3 neurons reflect the collinear arrangement of horizontal connections in tree shrew visual cortex. J Neurosci 23:2947–2960 (pii: 23/7/2947)
Christophe E, Roebuck A, Staiger JF, Lavery DJ, Charpak S, Audinat E (2002) Two types of nicotinic receptors mediate an excitation of neocortical layer I interneurons. J Neurophysiol 88:1318–1327. doi:10.1152/jn.00199.2002
Churchland MM et al (2010) Stimulus onset quenches neural variability: a widespread cortical phenomenon. Nat Neurosci 13:369-U325. doi:10.1038/nn.2501
Crochet S, Petersen CCH (2006) Correlating whisker behavior with membrane potential in barrel cortex of awake mice. Nat Neurosci 9:608–610. doi:10.1038/nn1690
Curto C, Sakata S, Marguet S, Itskov V, Harris KD (2009) A simple model of cortical dynamics explains variability and state dependence of sensory responses in urethane-anesthetized auditory cortex. J Neurosci 29:10600–10612. doi:10.1523/Jneurosci.2053-09.2009
Disney AA, Aoki C (2008) Muscarinic acetylcholine receptors in macaque V1 are most frequently expressed by parvalbumin-immunoreactive neurons. J Comp Neurol 507:1748–1762. doi:10.1002/cne.21616
Disney AA, Domakonda KV, Aoki C (2006) Differential expression of muscarinic acetylcholine receptors across excitatory and inhibitory cells in visual cortical areas V1 and V2 of the macaque monkey. J Comp Neurol 499:49–63. doi:10.1002/cne.21096
Disney AA, Aoki C, Hawken MJ (2007) Gain modulation by nicotine in macaque v1. Neuron 56:701–713. doi:10.1016/j.neuron.2007.09.034
Ecker AS, Berens P, Keliris GA, Bethge M, Logothetis NK, Tolias AS (2010) Decorrelated neuronal firing in cortical microcircuits. Science 327:584–587. doi:10.1126/science.1179867
Ecker AS et al (2014) State dependence of noise correlations in macaque primary visual cortex. Neuron 82:235–248. doi:10.1016/j.neuron.2014.02.006
Engel AK, Fries P, Singer W (2001) Dynamic predictions: oscillations and synchrony in top-down processing. Nat Rev Neurosci 2:704–716. doi:10.1038/35094565
Erisir A, Levey AI, Aoki C (2001) Muscarinic receptor M-2 in cat visual cortex: laminar distribution, relationship to gamma-aminobutyric acidergic neurons, and effect of cingulate lesions. J Comp Neurol 441:168–185. doi:10.1002/Cne.1405
Fitzpatrick D, Usrey WM, Schofield BR, Einstein G (1994) The sublaminar organization of corticogeniculate neurons in layer 6 of macaque striate cortex. Vis Neurosci 11:307–315
Fuller P, Sherman D, Pedersen NP, Saper CB, Lu J (2011) Reassessment of the structural basis of the ascending arousal system. J Comp Neurol 519:933–956. doi:10.1002/cne.22559
Gil Z, Connors BW, Amitai Y (1997) Differential regulation of neocortical synapses by neuromodulators and activity. Neuron 19:679–686 (pii: S0896-6273(00)80380-3)
Goard M, Dan Y (2009) Basal forebrain activation enhances cortical coding of natural scenes. Nat Neurosci 12:1444–1449. doi:10.1038/nn.2402
Haensel JX, Spain A, Martin C (2015) A systematic review of physiological methods in rodent pharmacological MRI studies. Psychopharmacology 232:489–499. doi:10.1007/s00213-014-3855-0
Han F, Caporale N, Dan Y (2008) Reverberation of recent visual experience in spontaneous cortical waves. Neuron 60:321–327. doi:10.1016/j.neuron.2008.08.026
Hansen BJ, Chelaru MI, Dragoi V (2012) Correlated variability in laminar cortical circuits. Neuron 76:590–602. doi:10.1016/j.neuron.2012.08.029
Hara K, Harris RA (2002) The anesthetic mechanism of urethane: the effects on rotransmitter-gated ion channels. Anesth Analg 94:313–318. doi:10.1097/00000539-200202000-00015
Harris KD, Thiele A (2011) Cortical state and attention. Nat Rev Neurosci 12:509–523. doi:10.1038/nrn3084
Hasenstaub A, Sachdev RNS, McCormick DA (2007) State changes rapidly modulate cortical neuronal responsiveness. J Neurosci 27:9607–9622. doi:10.1523/Jneurosci.2184-07.2007
Hsieh CY, Cruikshank SJ, Metherate R (2000) Differential modulation of auditory thalamocortical and intracortical synaptic transmission by cholinergic agonist. Brain Res 880:51–64 (pii: S0006-8993(00)02766-9)
Kalmbach A, Waters J (2014) Modulation of high- and low-frequency components of the cortical local field potential via nicotinic and muscarinic acetylcholine receptors in anesthetized mice. J Neurophysiol 111:258–272. doi:10.1152/jn.00244.2013
Kang JI, Vaucher E (2009) Cholinergic pairing with visual activation results in long-term enhancement of visual evoked potentials. PLoS One 4:e5995. doi:10.1371/journal.pone.0005995
Kim T et al (2015) Cortically projecting basal forebrain parvalbumin neurons regulate cortical gamma band oscillations (vol 112, pg 3535. P Natl Acad Sci USA 112:E2848–E2848. doi:10.1073/pnas.1507465112
Kleiner M, Brainard D, Pelli D (2007) What's new in psychtoolbox-3? In: Abstract supplement of the 30th European conference on visual perception (ECVP), vol 36, p 14
Kohn A, Zandvakili A, Smith MA (2009) Correlations and brain states: from electrophysiology to functional imaging. Curr Opin Neurobiol 19:434–438. doi:10.1016/j.conb.2009.06.007
Lee SH, Dan Y (2012) Neuromodulation of brain states. Neuron 76:209–222. doi:10.1016/j.neuron.2012.09.012
Marguet SL, Harris KD (2011) State-dependent representation of amplitude-modulated noise stimuli in rat auditory cortex. J Neurosci 31:6414–6420. doi:10.1523/Jneurosci.5773-10.2011
McLin DE 3rd, Miasnikov AA, Weinberger NM (2002) Induction of behavioral associative memory by stimulation of the nucleus basalis. Proc Natl Acad Sci USA 99:4002–4007. doi:10.1073/pnas.062057099
Metherate R, Cox CL, Ashe JH (1992) Cellular bases of neocortical activation: modulation of neural oscillations by the nucleus basalis and endogenous acetylcholine. J Neurosci 12:4701–4711
Mitchell JF, Sundberg KA, Reynolds JH (2009) Spatial attention decorrelates intrinsic activity fluctuations in macaque area V4. Neuron 63:879–888. doi:10.1016/j.neuron.2009.09.013
Munk AA, Adjemian RA, Zhao J, Ogbaghebriel A, Shrier A (1996) Electrophysiological properties of morphologically distinct cells isolated from the rabbit atrioventricular node. J Physiol-Lond 493:801–818
Niell CM, Stryker MP (2008) Highly selective receptive fields in mouse visual cortex. J Neurosci 28:7520–7536. doi:10.1523/JNEUROSCI.0623-08.2008
O’Leary MA et al (2013) The placental mammal ancestor and the post-K-Pg radiation of placentals. Science 339:662–667. doi:10.1126/science.1229237
Parent A, Pare D, Smith Y, Steriade M (1988) Basal forebrain cholinergic and noncholinergic projections to the thalamus and brain-stem in cats and monkeys. J Comp Neurol 277:281–301. doi:10.1002/cne.902770209
Pinto L et al (2013) Fast modulation of visual perception by basal forebrain cholinergic neurons. Nat Neurosci 16:1857–1863. doi:10.1038/nn.3552
Poirot J, De Luna P, Rainer G (2016) Neural coding of image structure and contrast polarity of Cartesian, hyperbolic, and polar gratings in the primary and secondary visual cortex of the tree shrew. J Neurophysiol 115:2000–2013. doi:10.1152/jn.01000.2015
Poulet JFA, Petersen CCH (2008) Internal brain state regulates membrane potential synchrony in barrel cortex of behaving mice. Nature 454:881-U836. doi:10.1038/nature07150
Ray S, Hsiao SS, Crone NE, Franaszczuk PJ, Niebur E (2008) Effect of stimulus intensity on the spike-local field potential relationship in the secondary somatosensory cortex. J Neurosci 28:7334–7343. doi:10.1523/JNEUROSCI.1588-08.2008
Renart A, de la Rocha J, Bartho P, Hollender L, Parga N, Reyes A, Harris KD (2010) The asynchronous state in cortical circuits. Science 327:587–590. doi:10.1126/science.1179850
Rieke F, Warland D, Steveninck RDRV, Bialek W (1999) Spikes: exploring the neural code. MIT Press, Cambridge. https://mitpress.mit.edu/books/spikes
Ringach DL, Sapiro G, Shapley R (1997) A subspace reverse-correlation technique for the study of visual neurons. Vision Res 37:2455–2464 (pii: S0042-6989(96)00247-7)
Shadlen MN, Newsome WT (1998) The variable discharge of cortical neurons: implications for connectivity, computation, and information coding. J Neurosci 18:3870–3896
Siegel M, Konig P (2003) A functional gamma-band defined by stimulus-dependent synchronization in area 18 of awake behaving cats. J Neurosci 23:4251–4260
Sirota A, Montgomery S, Fujisawa S, Isomura Y, Zugaro M, Buzsaki G (2008) Entrainment of neocortical neurons and gamma oscillations by the hippocampal theta rhythm. Neuron 60:683–697. doi:10.1016/j.neuron.2008.09.014
Softky WR, Koch C (1993) The highly irregular firing of cortical-cells is inconsistent with temporal integration of random EPSPS. J Neurosci 13:334–350
Sompolinsky H, Yoon H, Kang KJ, Shamir M (2001) Population coding in neuronal systems with correlated noise. Phys Rev E. doi:10.1103/Physreve.64.051904
Steriade M, Parent A, Pare D, Smith Y (1987) Cholinergic and noncholinergic neurons of cat basal forebrain project to reticular and mediodorsal thalamic nuclei. Brain Res 408:372–376. doi:10.1016/0006-8993(87)90408-2
Steriade M, Mccormick DA, Sejnowski TJ (1993) Thalamocortical oscillations in the sleeping and aroused brain. Science 262:679–685. doi:10.1126/science.8235588
Steriade M, Timofeev I, Grenier F (2001) Natural waking and sleep states: a view from inside neocortical neurons. J Neurophysiol 85:1969–1985
Thiele A, Herrero JL, Distler C, Hoffmann KP (2012) Contribution of cholinergic and GABAergic mechanisms to direction tuning, discriminability, response reliability, and neuronal rate correlations in macaque middle temporal area. J Neurosci 32:16602–16615. doi:10.1523/Jneurosci.0554-12.2012
Van Hooser SD, Roy A, Rhodes HJ, Culp JH, Fitzpatrick D (2013) Transformation of receptive field properties from lateral geniculate nucleus to superficial V1 in the tree shrew. J Neurosci 33:11494–11505. doi:10.1523/Jneurosci.1464-13.2013
Veit J, Bhattacharyya A, Kretz R, Rainer G (2011) Neural response dynamics of spiking and local field potential activity depend on CRT monitor refresh rate in the tree shrew primary visual cortex. J Neurophysiol 106:2303–2313. doi:10.1152/jn.00388.2011
Violet JM, Downie DL, Nakisa RC, Lieb WR, Franks NP (1997) Differential sensitivities of mammalian neuronal and muscle nicotinic acetylcholine receptors to general anesthetics. Anesthesiology 86:866–874. doi:10.1097/00000542-199704000-00017
Worgotter F, Suder K, Zhao YQ, Kerscher N, Eysel UT, Funke K (1998) State-dependent receptive-field restructuring in the visual cortex. Nature 396:165–168. doi:10.1038/24157
Xing D, Yeh CI, Burns S, Shapley RM (2012) Laminar analysis of visually evoked activity in the primary visual cortex. Proc Natl Acad Sci USA 109:13871–13876. doi:10.1073/pnas.1201478109
Zhu YJ, Qiao WH, Liu KF, Zhong HY, Yao HS (2015) Control of response reliability by parvalbumin-expressing interneurons in visual cortex. Nat Commun. doi:10.1038/Ncomms7802
Zohary E, Shadlen MN, Newsome WT (1994) Correlated neuronal discharge rate and its implications for psychophysical performance. Nature 370:140–143. doi:10.1038/370140a0
Acknowledgements
This work was supported by the Schweizerische Nationalfonds (Grant Number 143390, to GR), the EURYI program (to GR), and the University of Fribourg. We thank M. Harvey and S. Sachidhanandam for helpful comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
De Luna, P., Veit, J. & Rainer, G. Basal forebrain activation enhances between-trial reliability of low-frequency local field potentials (LFP) and spiking activity in tree shrew primary visual cortex (V1). Brain Struct Funct 222, 4239–4252 (2017). https://doi.org/10.1007/s00429-017-1468-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-017-1468-1