Abstract
Main conclusion
The introgression of the A ft allele into domesticated tomato induced a shift from flavonol to anthocyanin production in response to UV-B radiation, while the hp - 1 allele negatively influenced the response of flavonoid biosynthesis to UV-B.
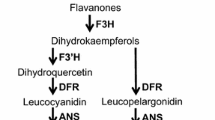
Introgression of the dominant allele Anthocyanin fruit (Aft) from Solanum chilense induces anthocyanin accumulation in the peel of tomato (Solanum lycopersicum L.) fruit. UV-B radiation can influence plant secondary metabolism regulating the expression of several genes, among which those involved in flavonoid biosynthesis. Here, we investigated whether post-harvest UV-B treatment could up-regulate flavonoid production in tomato fruits and whether the Aft allele could affect flavonoid biosynthesis under UV-B radiation. Mature green fruits of an anthocyanin-rich tomato mutant line (SA206) and of its wild-type reference, cv. Roma, were daily subjected to post-harvest UV-B treatment until full ripening. Up-regulation of CHS and CHI transcription by UV-B treatment induced flavonoid accumulation in the peel of cv. Roma. Conversely, UV-B decreased the total flavonoid content and CHS transcript levels in the SA206 peel. SA206 being a double mutant containing also hp-1 allele, we investigated also the behavior of hp-1 fruit. The decreased peel flavonoid accumulation and gene transcription in response to UV-B suggest that hp-1 allele is involved in the marked down-regulation of the flavonoid biosynthesis observed in SA206 fruit. Interestingly, in SA206, UV-B radiation promoted the synthesis of delphinidin, petunidin, and malvidin by increasing F3′5′H and DFR transcription, but it decreased rutin production, suggesting a switch from flavonols to anthocyanins. Finally, although UV-B radiation does not reach the inner fruit tissues, it down-regulated flavonoid biosynthesis in the flesh of both genotypes. This study provides, for the first time, evidence that the presence of the functional Aft allele, under UV-B radiation, redirects flavonoid synthesis towards anthocyanin production and suggests that the hp-1 allele negatively influences the response of flavonoid biosynthesis to UV-B.






Similar content being viewed by others
Abbreviations
- Aft :
-
Anthocyanin fruit
- ANS :
-
Anthocyanidin synthase
- CHI :
-
Chalcone isomerase
- CHS :
-
Chalcone synthase
- COP1 :
-
CONSTITUTIVE PHOTOMORPHOGENIC 1
- DFR :
-
Dihydroflavonol 4-reductase
- F3H :
-
Flavanone 3-hydroxylase
- F3′H :
-
Flavonoid 3′-hydroxylase
- F3′5′H :
-
Flavonoid 3′5′-hydroxylase
- hp-1 :
-
High-pigment-1
- UVR8 :
-
UV RESISTANCE LOCUS8
References
Adato A, Mandel T, Mintz-Oron S, Venger I, Levy D, Yativ M, Domínguez E, Wang Z, De Vos RCH, Jetter R, Schreiber L, Heredia A, Rogachev I, Aharoni A (2009) Fruit-surface flavonoid accumulation in tomato is controlled by a SLMYB12-regulated transcriptional network. PLoS Genet 5(12):e1000777
Alonso Borbalán ÁM, Zorro L, Guillén DA, García Barroso C (2003) Study of the polyphenol content of red and white grape varieties by liquid chromatography-mass spectrometry and its relationship to antioxidant power. J Chromatogr A 1012:31–38
Ballester AR, Molthoff J, de Vos R, Hekkert BTL, Orzaez D, Fernández Moreno JP, Tripodi P, Grandillo S, Martin C, Heldens J, Ykema M, Granell A, Bovy A (2010) Biochemical and molecular analysis of pink tomatoes: deregulated expression of the gene encoding transcription factor SlMYB12 leads to pink tomato fruit color. Plant Physiol 152(1):71–84
Ban Y, Honda C, Hatsuyama Y, Igarashi M, Bessho H, Moriguchi T (2007) Isolation and functional analysis of a MYB Transcription factor gene that is a key regulator for the development of red coloration in apple skin. Plant Cell Physiol 48(7):958–970
Bartley GE, Ishida BK (2003) Developmental gene regulation during tomato fruit ripening and in vitro sepal morphogenesis. BMC Plant Biol 3:4
Becatti E, Chkaiban L, Tonutti P, Forcato C, Bonghi C, Ranieri A (2010) Short-term postharvest carbon dioxide treatments induce selective molecular and metabolic changes in grape berries. J Agric Food Chem 58(13):8012–8020
Boches P, Myers J (2007) The anthocyanin fruit tomato gene (Aft) is associated with a DNA polymorphism in a MYB transcription factor. Hortic Sci 42:856
Bovy A, De Vos R, Kemper M, Schijlen E, Almenar Pertejo M, Muir S, Collins G, Robinson S, Verhoeyen M, Hughes S, Santos-Buelga C, van Tunen A (2002) High-flavonol tomatoes resulting from the heterologous expression of the maize transcription factor genes LC and C1. Plant Cell 14:2509–2526
Butelli E, Titta L, Giorgio M, Mock HP, Matros A, Peterek S, Schijlen EG, Hall RD, Bovy AG, Luo J, Martin C (2008) Enrichment of tomato fruit with health-promoting anthocyanins by expression of select transcription factors. Nat Biotechnol 26(11):1301–1308
Calvenzani V, Martinelli M, Lazzeri V, Giuntini D, Dall’Asta C, Galaverna G, Tonelli C, Ranieri A, Petroni K (2010) Response of wild-type and high pigment-1 tomato fruit to UV-B depletion: flavonoid profiling and gene expression. Planta 231(3):755–765
Castagna A, Chiavaro E, Dall’Asta C, Rinaldi M, Galaverna G, Ranieri A (2013) Effect of Postharvest UV-B irradiation on nutraceutical quality and physical properties of tomato fruits. Food Chem 137(1–4):151–158
Castagna A, Dall’Asta C, Chiavaro E, Galaverna G, Ranieri A (2014) Effect of post-harvest UV-B irradiation on polyphenol profile and antioxidant activity in flesh and peel of tomato fruits. Food Bioprocess Technol 7(8):2241–2250
Giuntini D, Lazzeri V, Calvenzani V, Dall’Asta C, Galaverna G, Tonelli C, Petroni C, Ranieri A (2008) Flavonoid profiling and biosynthetic gene expression in flesh and peel of two tomato genotypes grown under UV-B-depleted conditions during ripening. J Agric Food Chem 56(14):5905–5915
Gould K (2004) Nature’s Swiss army knife: the diverse protective roles of anthocyanins in leaves. J Biomed Biotechnol 5(5):314–320
Grotewold E (2006) The genetics and biochemistry of floral pigments. Annu Rev Plant Biol 57:761–780
Guo J, Han W, Wang M-H (2008) Ultraviolet and environmental stresses involved in the induction and regulation of anthocyanin biosynthesis: a review. Afr J Biotechnol 7(25):4966–4972
Gutierres JM, Carvalho FB, Schetinger MRC, Marisco P, Agostinho P, Rodrigues M, Rubin MA, Schmatz R, da Silva CR, Cognato GdP, Farias JG, Signor C, Morscha VM, Mazzanti CM, Bogo M, Bonan CD, Spanevello R (2014) Anthocyanins restore behavioral and biochemical changes caused by streptozotocin-induced sporadic dementia of Alzheimer’s type. Life Sci 96(1–2):7–17
Hagen SF, Borge GIA, Bengtsson GB, Bilger W, Berge A, Haffner K, Solhaug KA (2007) Phenolic contents and other health and sensory related properties of apple fruit (Malus domestica., Borkh cv. Aroma): effect of postharvest UV-B irradiation. Postharvest Biol Technol 45(1):1–10
Hemm MR, Herrmann KM, Chapple C (2001) AtMYB4: a transcription factor general in the battle against UV. Trends Plant Sci 6:135–136
Hidalgo M, Oruna-Concha MJ, Walton GE, Kallithraka S, Spencer JPE, Gibson GR, de Pascual-Teresa S (2012) Metabolism of anthocyanins by human gut micro flora and their influence on gut bacterial growth. J Agric Food Chem 60:3882–3890
Huang X, Ouyang X, Yang P, Lau OS, Chen L, Wei N, Deng XW (2013) Conversion from CUL4-based COP1–SPA E3 apparatus to UVR8–COP1–SPA complexes underlies a distinct biochemical function of COP1 under UV-B. Proc Natl Acad Sci USA 110(41):16669–16674
Jenkins GI (2014) Structure and function of the UV-B photoreceptor UVR8. Curr Opin Struct Biol 29:52–57
Jones CM, Mes P, Myers JR (2003) Characterization and inheritance of the Anthocyanin fruit (Aft) tomato. J Hered 94(6):449–456
Kaiserli E, Jenkins GI (2007) UV-B promotes rapid nuclear translocation of the Arabidopsis UV-B specific signaling component UVR8 and activates its function in the nucleus. Plant Cell 19(8):2662–2673
Kiferle C, Fantini E, Bassolino L, Povero G, Spelt C, Buti S, Giuliano G, Quattrocchio F, Koes R, Perata P, Gonzali S (2015) Tomato R2R3-MYB proteins SlANT1 and SlAN2: same protein activity, different roles. PLoS One 10(8):1–20
Kim DO, Chun OK, Kim YJ, Moon HY, Lee CY (2003) Quantification of polyphenolics and their antioxidant capacity in fresh plums. J Agric Food Chem 51(22):6509–6515
Kliebenstein DJ, Lim JE, Landry LG, Last RL (2002) Arabidopsis UVR8 regulates Ultraviolet-B signal transduction and tolerance and contains sequence similarity to human regulator of chromatin condensation 1. Plant Physiol 130(1):234–243
Liu C, Han X, Cai L, Lu X, Ying T, Jiang Z (2011) Postharvest UV-B irradiation maintains sensory qualities and enhances antioxidant capacity in tomato fruit during storage. Postharvest Biol Technol 59(3):232–237
Liu Y, Li D, Zhang Y, Sun R, Xia M (2014) Anthocyanin increases adiponectin secretion and protects against diabetes-related endothelial dysfunction. Am J Physiol Endocrinol Metab 306:E975–E988
Mathews H, Clendennen SK, Caldwell CG, Liu XL, Connors K, Matheis N, Schuster DK, Menasco DJ, Wagoner W, Lightner J, Wagner DR (2003) Activation tagging in tomato identifies a transcriptional regulator of anthocyanin biosynthesis, modification, and transport. Plant Cell 15(8):1689–1703
Mazzucato A, Willems D, Bernini R, Picarella ME, Santangelo E, Ruiu F, Tilesi F, Soressi GP (2013) Novel phenotypes related to the breeding of purple-fruited tomatoes and effect of peel extracts on human cancer cell proliferation. Plant Physiol Biochem 72:125–133
Meng X, Yang D, Li X, Zhao S, Sui N, Meng Q (2015) Physiological changes in fruit ripening caused by overexpression of tomato SlAN2, an R2R3-MYB factor. Plant Physiol Biochem 89:24–30
Mes PJ, Boches P, Myers JR, Durst R (2008) Characterization of tomatoes expressing anthocyanin in the fruit. J Am Soc Hortic Sci 133(2):262–269
Peng T, Saito T, Honda C, Ban Y, Kondo S, Liu JH, Hatsuyama Y, Moriguchi T (2013) Screening of UV-B-induced genes from apple peels by SSH: possible involvement of MdCOP1-mediated signaling cascade genes in anthocyanin accumulation. Physiol Plant 148(3):432–444
Petroni K, Tonelli C (2011) Recent advances on the regulation of anthocyanin synthesis in reproductive organs. Plant Sci 181(3):219–229
Pojer E, Mattivi F, Johnson D, Stockley CS (2013) The case for anthocyanin consumption to promote human health: a review. Comp Rev Food Sci Food Saf 12:483–508
Procissi A, Dolfini S, Ronchi A, Tonelli C (1997) Light-dependent spatial and temporal expression of pigment regulatory genes in developing maize seeds. Plant Cell 9:1547–1557
Qian M, Zhang D, Yue X, Wang S, Li X, Teng Y (2013) Analysis of different pigmentation patterns in ‘Mantianhong’ (Pyrus pyrifolia Nakai) and ‘Cascade’ (Pyrus Communis L.) under bagging treatment and postharvest UV-B/visible irradiation conditions. Sci Hortic 151:75–82
Ravaglia D, Espley RV, Henry-Kirk RA, Andreotti C, Ziosi V, Hellens RP, Costa G, Allan AC (2013) Transcriptional regulation of flavonoid biosynthesis in nectarine (Prunus persica) by a set of R2R3 MYB transcription factors. BMC Plant Biol 13:68
Rizzini L, Favory JJ, Cloix C, Faggionato D, O’Hara A, Kaiserli E, Baumeister R, Schäfer E, Nagy F, Jenkins GI, Ulm R (2011) Perception of UV-B by the Arabidopsis UVR8 protein. Science 332:103–106
Ross S (2000) Functional foods: the food and drug administration perspective. Am J Clin Nutr 71(6):1735S–1738S
Scattino C, Castagna A, Neugart S, Chan HM, Schreiner M, Crisosto CH, Tonutti P, Ranieri A (2014) Post-harvest UV-B irradiation induces changes of phenol contents and corresponding biosynthetic gene expression in peaches and nectarines. Food Chem 163:51–60
Schreiber G, Reuveni M, Evenor D, Oren-Shamir M, Ovadia R, Sapir-Mir M, Bootbool-Man A, Nahon S, Shlomo H, Chen L, Levin I (2012) ANTHOCYANIN1 from Solanum chilense is more efficient in accumulating anthocyanin metabolites than its Solanum lycopersicum counterpart in association with the ANTHOCYANIN FRUIT phenotype of tomato. Theor Appl Genet 124(2):295–307
Schreiner M, Mewis I, Huyskens-Keil S, Jansen MAK, Zrenner R, Winkler JB, O’Brien N, Krumbein A (2012) UV-B-induced secondary plant metabolites—potential benefits for plant and human health. Crit Rev Plant Sci 31:229–240
Shang Y, Venail J, Mackay S, Bailey PC, Schwinn KE, Jameson PE, Martin CR, Davies KM (2011) The molecular basis for venation patterning of pigmentation and its effect on pollinator attraction in flowers of Antirrhinum. New Phytol 189(2):602–615
Shin DH, Choi MG, Kim K, Bang G, Cho M, Choi S-B, Choi G, Park Y-II (2013) HY5 regulates anthocyanin biosynthesis by inducing the transcriptional activation of the MYB75/PAP1 transcription factor in Arabidopsis. FEBS Lett 587:1543–1547
Stracke R, Favory J-J, Gruber H, Bartelniewoehner L, Bartels S, Binkert M, Funk M, Weisshaar B, Ulm R (2010) The Arabidopsis bZIP transcription factor HY5 regulates expression of the PFG1/MYB12 gene in response to light and ultraviolet-B radiation. Plant Cell Environ 33:88–103
Sun Y, Qian M, Wu R, Niu Q, Teng Y, Zhang D (2014) Postharvest pigmentation in red chinese sand pears (Pyrus pyrifolia Nakai) in response to optimum light and temperature. Postharvest Biol Technol 91:64–71
Titta L, Trinei M, Stendardo M, Berniakovich I, Petroni K, Tonelli C, Riso P, Porrini M, Minucci S, Pelicci PG, Rapisarda P, Reforgiato Recupero G, Giorgio M (2010) Blood orange juice inhibits fat accumulation in mice. Int J Obesity 34(3):578–588
Torres CA, Davies NM, Yañez JA, Andrews PK (2005) Disposition of selected flavonoids in fruit tissues of various tomato (Lycopersicon esculentum Mill.) genotypes. J Agric Food Chem 53(24):9536–9543
Toufektsian MC, de Lorgeril M, Nagy N, Salen P, Donati MB, Giordano L, Mock HP, Peterek S, Matros A, Petroni K, Pilu R, Rotilio D, Tonelli C, de Leiris J, Boucher F, Martin C (2008) Chronic dietary intake of plant-derived anthocyanins protects the rat heart against ischemia–reperfusion injury. J Nutr 138(4):747–752
Wallace TC (2011) Anthocyanins in cardiovascular disease. Adv Nutr 2(7):1–7
Wang LS, Stoner GD (2008) Anthocyanins and their role in cancer prevention. Cancer Lett 269(2):281–290
Wang S, Liu J, Feng Y, Niu X, Giovannoni J, Liu Y (2008) Altered plastid levels and potential for improved fruit nutrient content by downregulation of the tomato DDB1-interacting protein CUL4. Plant J 55:89–103
Acknowledgements
The research was supported by funds of the University of Pisa and the University of Milan. We are indebted with Prof. Gian Piero Soressi (University of Tuscia) for sharing genetic material and Prof. Chiara Tonelli (University of Milan) for helpful discussions. We kindly acknowledge Prof. Åke Strid (School of Science and Technology, Orebro University) for measurement of light transmittance across tomato peel.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Special topic: Polyphenols II: biosynthesis and function in plants and ecosystems. Guest editor: Stefan Martens.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Catola, S., Castagna, A., Santin, M. et al. The dominant allele Aft induces a shift from flavonol to anthocyanin production in response to UV-B radiation in tomato fruit. Planta 246, 263–275 (2017). https://doi.org/10.1007/s00425-017-2710-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-017-2710-z