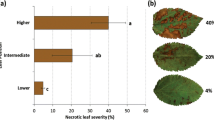
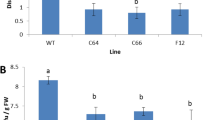
Abstract
Main conclusion
Resistance against anthracnose fungi was enhanced in transgenic pepper plants that accumulated high levels of a carboxylesterase, PepEST in anthracnose-susceptible fruits, with a concurrent induction of antioxidant enzymes and SA-dependent PR proteins.
A pepper esterase gene (PepEST) is highly expressed during the incompatible interaction between ripe fruits of pepper (Capsicum annuum L.) and a hemibiotrophic anthracnose fungus (Colletotrichum gloeosporioides). In this study, we found that exogenous application of recombinant PepEST protein on the surface of the unripe pepper fruits led to a potentiated state for disease resistance in the fruits, including generation of hydrogen peroxide and expression of pathogenesis-related (PR) genes that encode mostly small proteins with antimicrobial activity. To elucidate the role of PepEST in plant defense, we further developed transgenic pepper plants overexpressing PepEST under the control of CaMV 35S promoter. Molecular analysis confirmed the establishment of three independent transgenic lines carrying single copy of transgenes. The level of PepEST protein was estimated to be approximately 0.002 % of total soluble protein in transgenic fruits. In response to the anthracnose fungus, the transgenic fruits displayed higher expression of PR genes, PR3, PR5, PR10, and PepThi, than non-transgenic control fruits did. Moreover, immunolocalization results showed concurrent localization of ascorbate peroxidase (APX) and PR3 proteins, along with the PepEST protein, in the infected region of transgenic fruits. Disease rate analysis revealed significantly low occurrence of anthracnose disease in the transgenic fruits, approximately 30 % of that in non-transgenic fruits. Furthermore, the transgenic plants also exhibited resistance against C. acutatum and C. coccodes. Collectively, our results suggest that overexpression of PepEST in pepper confers enhanced resistance against the anthracnose fungi by activating the defense signaling pathways.






Similar content being viewed by others
Abbreviations
- APX:
-
Ascorbate peroxidase
- CaMV:
-
Cauliflower Mosaic Virus
- CXE:
-
Carboxylesterase
- gDNA:
-
Genomic DNA
- HR:
-
Hypersensitive response
- JA:
-
Jasmonic acid
- LB:
-
Left border
- MeSA:
-
Methyl SA
- PepEST:
-
Pepper esterase
- PR:
-
Pathogenesis related
- RB:
-
Right border
- SA:
-
Salicylic acid
References
Alkan N, Fluhr R, Prusky D (2012) Ammonium secretion during Colletotrichum coccodes infection modulates salicylic and jasmonic acid pathways of ripe and unripe tomato fruit. Mol Plant Microbe Interact 25:85–96
Apostol I, Heinstein PF, Low PS (1989) Rapid stimulation of an oxidative burst during elicitation of cultured plant cells: role in defense and signal transduction. Plant Physiol 90:109–116
Aranda J, Cerqueira NM, Fernandes PA, Roca M, Tunon I, Ramos MJ (2014) The catalytic mechanism of carboxylesterases: a computational study. Biochemistry 53:5820–5829
Bezier A, Lambert B, Baillieul F (2002) Cloning of a grapevine Botrytis-responsive gene that has homology to the tobacco hypersensitivity-related hsr203 J. J Exp Bot 53:2279–2280
Bowling SA, Guo A, Cao H, Gordon AS, Klessig DF, Dong X (1994) A mutation in Arabidopsis that leads to constitutive expression of systemic acquired resistance. Plant Cell 6:1845–1857
Chilton MD, Que Q (2003) Targeted integration of T-DNA into the tobacco genome at double-stranded breaks: new insights on the mechanism of T-DNA integration. Plant Physiol 133:956–965
Cho KC, Han YJ, Kim SJ, Lee SS, Hwang OJ, Song PS, Kim YS, Kim JI (2011) Resistance to Rhizoctonia solani AG-2-2 (IIIB) in creeping bentgrass plants transformed with pepper esterase gene PepEST. Plant Pathol 60:631–639
Cui F, Li MX, Chang HJ, Mao Y, Zhang HY, Lu LX, Yan SG, Lang ML, Liu L, Qiao CL (2015) Carboxylesterase-mediated insecticide resistance: quantitative increase induces broader metabolic resistance than qualitative change. Pest Biochem Physiol 121:88–96
Cummins I, Landrum M, Steel PG, Edwards R (2007) Structure activity studies with xenobiotic substrates using carboxylesterases isolated from Arabidopsis thaliana. Phytochemistry 68:811–818
De Buck S, Podevin N, Nolf J, Jacobs A, Depicker A (2009) The T-DNA integration pattern in Arabidopsis transformants is highly determined by the transformed target cell. Plant J 60:134–145
Falk A, Feys BJ, Frost LN, Jones JD, Daniels MJ, Parker JE (1999) EDS1, an essential component of R gene-mediated disease resistance in Arabidopsis has homology to eukaryotic lipases. Proc Natl Acad Sci USA 96:3292–3297
Feys BJ, Moisan LJ, Newman MA, Parker JE (2001) Direct interaction between the Arabidopsis disease resistance signaling proteins, EDS1 and PAD4. EMBO J 20:5400–5411
Forouhar F, Yang Y, Kumar D, Chen Y, Fridman E, Park SW, Chiang Y, Acton TB, Montelione GT, Pichersky E, Klessig DF, Tong L (2005) Structural and biochemical studies identify tobacco SABP2 as a methyl salicylate esterase and implicate it in plant innate immunity. Proc Natl Acad Sci USA 102:1773–1778
Fu ZQ, Dong X (2013) Systemic acquired resistance: turning local infection into global defense. Annu Rev Plant Biol 64:839–863
Gershater MC, Edwards R (2007) Regulating biological activity in plants with carboxylesterases. Plant Sci 173:579–588
Gershater MC, Cummins I, Edwards R (2007) Role of a carboxylesterase in herbicide bioactivation in Arabidopsis thaliana. J Biol Chem 282:21460–21466
Glazebrook J (2005) Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu Rev Phytopathol 43:205–227
Hong JK, Jung HW, Kim YJ, Hwang BK (2000) Pepper gene encoding a basic class II chitinase is inducible by pathogen and ethephon. Plant Sci 159:39–49
Ichinose Y, Hisayasu Y, Sanematsu S, Ishiga Y, Seki H, Toyoda K, Shiraishi T, Yamada T (2001) Molecular cloning and functional analysis of pea cDNA E86 encoding homologous protein to hypersensitivity-related hsr203J. Plant Sci 160:997–1006
Kim YS, Lee HH, Ko MK, Song CE, Bae CY, Lee YH, Oh BJ (2001) Inhibition of fungal appressorium formation by pepper (Capsicum annuum) esterase. Mol Plant Microbe Interact 14:80–85
Kim YS, Park JY, Kim KS, Ko MK, Cheong SJ, Oh BJ (2002) A thaumatin-like gene in nonclimacteric pepper fruits used as molecular marker in probing disease resistance, ripening, and sugar accumulation. Plant Mol Biol 49:125–135
Ko MK, Jeon WB, Kim KS, Lee HH, Seo HH, Kim YS, Oh BJ (2005) A Colletotrichum gloeosporioides-induced esterase gene of nonclimacteric pepper (Capsicum annuum) fruit during ripening plays a role in resistance against fungal infection. Plant Mol Biol 58:529–541
Ko MK, Soh H, Kim KM, Kim YS, Im K (2007) Stable production of transgenic pepper plants mediated by Agrobacterium tumefaciens. Hort Sci 42:1425–1430
Kordic S, Cummins I, Edwards R (2002) Cloning and characterization of an S-formylglutathione hydrolase from Arabidopsis thaliana. Arch Biochem Biophys 399:232–238
Kumar S, Fladung M (2002) Transgene integration in aspen: structures of integration sites and mechanism of T-DNA integration. Plant J 31:543–551
Laizure SC, Herring V, Hu Z, Witbrodt K, Parker RB (2013) The role of human carboxylesterases in drug metabolism: have we overlooked their importance? Pharmacotherapy 33:210–222
Marshall SD, Putterill JJ, Plummer KM, Newcomb RD (2003) The carboxylesterase gene family from Arabidopsis thaliana. J Mol Evol 57:487–500
Oakeshott JG, Claudianos C, Russell RJ, Robin GC (1999) Carboxyl/cholinesterases: a case study of the evolution of a successful multigene family. Bio Essays 21:1031–1042
Oh BJ, Ko MK, Kostenyuk I, Shin B, Kim KS (1999) Coexpression of a defensin gene and a thionin-like gene via different signal transduction pathways in pepper and Colletotrichum gloeosporioides interactions. Plant Mol Biol 41:313–319
Oh IS, Park AR, Bae MS, Kwon SJ, Kim YS, Lee JE, Kang NY, Lee S, Cheong H, Park OK (2005) Secretome analysis reveals an Arabidopsis lipase involved in defense against Alternaria brassicicola. Plant Cell 17:2832–2847
Park CJ, Kim KJ, Shin R, Park JM, Shin YC, Paek KH (2004) Pathogenesis-related protein 10 isolated from hot pepper functions as a ribonuclease in an antiviral pathway. Plant J 37:186–198
Park SW, Kaimoyo E, Kumar D, Mosher S, Klessig DF (2007) Methyl salicylate is a critical mobile signal for plant systemic acquired resistance. Science 318:113–116
Paulert R, Ebbinghaus D, Urlass C, Moerschbacher BM (2010) Priming of the oxidative burst in rice and wheat cell cultures by ulvan, a polysaccharide from green macroalgae, and enhanced resistance against powdery mildew in wheat and barley plants. Plant Pathol 59:634–642
Penninckx IA, Eggermont K, Terras FR, Thomma BP, De Samblanx GW, Buchala A, Metraux JP, Manners JM, Broekaert WF (1996) Pathogen-induced systemic activation of a plant defensin gene in Arabidopsis follows a salicylic acid-independent pathway. Plant Cell 8:2309–2323
Pontier D, Godiard L, Marco Y, Roby D (1994) hsr203J, a tobacco gene whose activation is rapid, highly localized and specific for incompatible plant/pathogen interactions. Plant J 5:507–521
Pontier D, Tronchet M, Rogowsky P, Lam E, Roby D (1998) Activation of hsr203, a plant gene expressed during incompatible plant-pathogen interactions, is correlated with programmed cell death. Mol Plant Microbe Interact 11:544–554
Prusky D, Kobiler I, Ardi R, Beno-Moalem D, Yakoby N, Keen NT (2000) Resistance mechanisms of subtropical fruits to Colletotrichum gloeosporioides. In: Prusky D, Freeman S, Dickman M (eds) Colletotrichum: Host specificity, pathology, and host-pathogen interaction. American Phytopathological Society Press, St Paul, pp 232–244
Seo HH, Park S, Park S, Oh BJ, Back K, Han O, Kim JI, Kim YS (2014) Overexpression of a defensin enhances resistance to a fruit-specific anthracnose fungus in pepper. PLoS One 9:e97936
Shimizu K, Takahashi M, Goshima N, Kawakami S, Irifune K, Morikawa H (2001) Presence of an SAR-like sequence in junction regions between an introduced transgene and genomic DNA of cultured tobacco cells: its effect on transformation frequency. Plant J 26:375–384
Thordal-Christensen H, Zhang Z, Wei Y, Collinge DB (1997) Subcellular localization of H2O2 in plants. H2O2 accumulation in papillae and hypersensitive response during the barley-powdery mildew interaction. Plant J 11:1187–1194
Torres MA, Jones JDG, Dangl JL (2006) Reactive oxygen species signaling in response to pathogens. Plant Physiol 141:373–378
van Kan JA (2006) Licensed to kill: the lifestyle of a necrotrophic plant pathogen. Trend Plant Sci 11:247–253
Wei R, Oeser T, Zimmermann W (2014) Synthetic polyester-hydrolyzing enzymes from thermophilic actinomycetes. Adv Appl Microbiol 89:267–305
Zeng FS, Zhan YG, Zhao HC, Xin Y, Qi FH, Yang CP (2010) Molecular characterization of T-DNA integration sites in transgenic birch. Trees Struct Funct 24:753–762
Zhao Y, Park RD, Muzzarelli RA (2010) Chitin deacetylases: properties and applications. Mar Drugs 8:24–46
Zhu QH, Ramm K, Eamens AL, Dennis ES, Upadhyaya NM (2006) Transgene structures suggest that multiple mechanisms are involved in T-DNA integration in plants. Plant Sci 171:308–322
Zhu C, Wu J, He C (2010) Induction of chromosomal inversion by integration of T-DNA in the rice genome. J Genet Genomic 37:189–196
Acknowledgments
We thank the Kumho Life Science Laboratory in Chonnam National University for providing plant growth facilities. This work was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (Grant no. NRF-2014R1A1A2057739 to J.-I. Kim; Grant no. NRF-2014R1A1A2007413 to Y.S. Kim) and Next-Generation BioGreen 21 Program, Rural Development Administration, Republic of Korea (Grant no. PJ01104001 to J.-I. Kim).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ko, M., Cho, J.H., Seo, HH. et al. Constitutive expression of a fungus-inducible carboxylesterase improves disease resistance in transgenic pepper plants. Planta 244, 379–392 (2016). https://doi.org/10.1007/s00425-016-2514-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-016-2514-6