Abstract
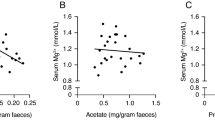
Dietary fibers have been shown to increase the intestinal absorption of calcium (Ca2+) and magnesium (Mg2+). However, the mechanisms that explain the enhanced electrolyte absorption remain unknown. Therefore, this study aims to investigate the short-term and long-term effects of 5% (w/w) sodium butyrate (Na-butyrate), an important end-metabolite of bacterial fermentation of dietary fibers, on Ca2+ and Mg2+ homeostasis in mice. Serum Ca2+ levels were only significantly increased in mice treated with Na-butyrate for 1 day. This was associated with a twofold increase in the mRNA expression levels of Trpv6 in the proximal and distal colon. Contrary, Na-butyrate did not affect serum Mg2+ concentrations at either of the intervention periods. However, we observed a reduction in urinary Mg2+ excretion, although not significantly, after 1 day of treatment. A significant reduction of 2.5–fold in urinary Mg2+ excretion was observed after 14 days of treatment. Indeed, 14-day Na-butyrate supplementation increased colonic Trpm7 expression by 1.2-fold compared to control mice. In conclusion, short-term Na-butyrate supplementation increases serum Ca2+ levels in mice. This was associated with increased mRNA expression levels of Trpv6 in the colon, suggesting that Na-butyrate regulates the expression of genes involved in active intestinal Ca2+ absorption.




Similar content being viewed by others
Data availability statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Alexander C, Swanson KS, Fahey GC, Garleb KA (2019) Perspective: Physiologic importance of short-chain fatty acids from nondigestible carbohydrate fermentation. Adv Nutr (Bethesda, Md) 10:576–589. https://doi.org/10.1093/advances/nmz004
Bedford A, Gong J (2018) Implications of butyrate and its derivatives for gut health and animal production. Anim Nutr 4:151–159. https://doi.org/10.1016/j.aninu.2017.08.010
Blaine J, Chonchol M, Levi M (2015) Renal control of calcium, phosphate, and magnesium homeostasis. Clin J Am Soc Nephrol 10:1257–1272
Bronner F (2003) Mechanisms of intestinal calcium absorption. J Cell Biochem 88:387–393. https://doi.org/10.1002/jcb.10330
Chonan O, Matsumoto K, Watanuki M (1995) Effect of galactooligosaccharides on calcium absorption and preventing bone loss in ovariectomized rats. Biosci Biotechnol Biochem 59:236–239. https://doi.org/10.1271/bbb.59.236
Chubanov V, Ferioli S, Wisnowsky A, Simmons DG, Leitzinger C, Einer C, Jonas W, Shymkiv Y, Bartsch H, Braun A, Akdogan B, Mittermeier L, Sytik L, Torben F, Jurinovic V, van der Vorst EP, Weber C, Yildirim OA, Sotlar K, Schurmann A, Zierler S, Zischka H, Ryazanov AG, Gudermann T (2016) Epithelial magnesium transport by TRPM6 is essential for prenatal development and adult survival. eLife 5. https://doi.org/10.7554/eLife.20914
Coudray C, Bellanger J, Vermorel M, Sinaud S, Wils D, Feillet-Coudray C, Brandolini M, Bouteloup-Demange C, Rayssiguier Y (2003) Two polyol, low digestible carbohydrates improve the apparent absorption of magnesium but not of calcium in healthy young men. J Nutr 133:90–93. https://doi.org/10.1093/jn/133.1.90
Coudray C, Demigne C, Rayssiguier Y (2003) Effects of dietary fibers on magnesium absorption in animals and humans. J Nutr 133:1–4. https://doi.org/10.1093/jn/133.1.1
Coudray C, Feillet-Coudray C, Tressol JC, Gueux E, Thien S, Jaffrelo L, Mazur A, Rayssiguier Y (2005) Stimulatory effect of inulin on intestinal absorption of calcium and magnesium in rats is modulated by dietary calcium intakes short- and long-term balance studies. Eur J Nutr 44:293–302. https://doi.org/10.1007/s00394-004-0526-7
de Baaij JH, Arjona FJ, van den Brand M, Lavrijsen M, Lameris AL, Bindels RJ, Hoenderop JG (2016) Identification of SLC41A3 as a novel player in magnesium homeostasis. Sci Rep 6:28565. https://doi.org/10.1038/srep28565
de Baaij JH, Hoenderop JG, Bindels RJ (2012) Regulation of magnesium balance: lessons learned from human genetic disease. Clin Kidney J 5:i15–i24. https://doi.org/10.1093/ndtplus/sfr164
de Baaij JH, Hoenderop JG, Bindels RJ (2015) Magnesium in man: implications for health and disease. Physiol Rev 95:1–46. https://doi.org/10.1152/physrev.00012.2014
den Besten G, van Eunen K, Groen AK, Venema K, Reijngoud DJ, Bakker BM (2013) The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J Lipid Res 54:2325–2340. https://doi.org/10.1194/jlr.R036012
Dirks NF, Martens F, Vanderschueren D, Billen J, Pauwels S, Ackermans MT, Endert E, Heijer MD, Blankenstein MA, Heijboer AC (2016) Determination of human reference values for serum total 1,25-dihydroxyvitamin D using an extensively validated 2D ID-UPLC-MS/MS method. J Steroid Biochem Mol Biol 164:127–133
Donovan JD, Bauer L, Fahey GC Jr, Lee Y (2017) In vitro digestion and fermentation of microencapsulated tributyrin for the delivery of butyrate. J Food Sci 6:1491–1499
Ferioli S, Zierler S, Zaißerer J, Schredelseker J, Gudermann T, Chubanov V (2017) TRPM6 and TRPM7 differentially contribute to the relief of heteromeric TRPM6/7 channels from inhibition by cytosolic Mg(2+) and Mg·ATP. Sci Rep 7:8806
Fukushima A, Aizaki Y, Sakuma K (2009) Short-chain fatty acids induce intestinal transient receptor potential vanilloid type 6 expression in rats and Caco-2 cells. J Nutr 139:20–25. https://doi.org/10.3945/jn.108.096230
Fukushima A, Aizaki Y, Sakuma K (2012) Short-chain fatty acids increase the level of calbindin-D9k messenger RNA in Caco-2 cells. J Nutr Sci Vitaminol (Tokyo) 58:287–291. https://doi.org/10.3177/jnsv.58.287
Gao Z, Yin J, Zhang J, Ward RE, Martin RJ, Lefevre M, Cefalu WT, Ye J (2009) Butyrate improves insulin sensitivity and increases energy expenditure in mice. Diabetes 58:1509–1517
Gommers LMM, Ederveen THA, van der Wijst J, Overmars-Bos C, Kortman GAM, Boekhorst J, Bindels RJM, de Baaij JHF, Hoenderop JGJ (2019) Low gut microbiota diversity and dietary magnesium intake are associated with the development of PPI-induced hypomagnesemia. FASEB J 10:11235–11246
Gonçalves P, Martel F (2016) Regulation of colonic epithelial butyrate transport: Focus on colorectal cancer. Porto Biomed J 1:83–91
Hardwick LL, Jones MR, Brautbar N, Lee DB (1990) Site and mechanism of intestinal magnesium absorption. Miner Electrolyte Metab 16:174–180
Li Z, Yi CX, Katiraei S, Kooijman S, Zhou E, Chung CK, Gao Y, van den Heuvel JK, Meijer OC, Berbée JFP, Heijink M, Giera M, Willems van Dijk K, Groen AK, Rensen PCN, Wang Y (2018) Butyrate reduces appetite and activates brown adipose tissue via the gut-brain neural circuit. Gut 67:1269–1279. https://doi.org/10.1136/gutjnl-2017-314050
Liu H, Wang J, He T, Becker S, Zhang G, Li D, Ma X (2018) Butyrate: a double-edged sword for health? Adv Nutr (Bethesda, Md) 9:21–29. https://doi.org/10.1093/advances/nmx009
Lucas S, Omata Y, Hofmann J, Böttcher M, Iljazovic A, Sarter K, Albrecht O, Schulz O, Krishnacoumar B, Krönke G, Herrmann MA-O, Mougiakakos D, Strowig TA-O, Schett G, Zaiss MM (2018) Short-chain fatty acids regulate systemic bone mass and protect from pathological bone loss. Nat Commun 9(1):55. https://doi.org/10.1038/s41467-017-02490-4
Mineo H, Hara H, Tomita F (2001) Short-chain fatty acids enhance diffusional ca transport in the epithelium of the rat cecum and colon. Life Sci 69:517–526. https://doi.org/10.1016/s0024-3205(01)01146-8
Oh TJ, Sul WJ, Oh HN, Lee YK, Lim HL, Choi SH, Park KS, Jang HC (2019) Butyrate attenuated fat gain through gut microbiota modulation in db/db mice following dapagliflozin treatment. Sci Rep 9:20300
Ohta A, Motohashi Y, Sakai K, Hirayama M, Adachi T, Sakuma K (1998) Dietary fructooligosaccharides increase calcium absorption and levels of mucosal calbindin-D9k in the large intestine of gastrectomized rats. Scand J Gastroenterol 33:1062–1068. https://doi.org/10.1080/003655298750026769
Ohta A, Ohtsuki M, Baba S, Hirayama M, Adachi T (1998) Comparison of the nutritional effects of fructo-oligosaccharides of different sugar chain length in rats. Nutr Res 18:109–120. https://doi.org/10.1016/S0271-5317(97)00205-4
Scharrer E, Lutz T (1990) Effects of short chain fatty acids and K on absorption of Mg and other cations by the colon and caecum. Z Ernahrungswiss 29:162–168. https://doi.org/10.1007/bf02021554
Schuchardt JP, Hahn A (2017) Intestinal absorption and factors influencing bioavailability of magnesium-an update. Curr Nutr Food Sci 13:260–278. https://doi.org/10.2174/1573401313666170427162740
Tahiri M, Tressol JC, Arnaud J, Bornet F, Bouteloup-Demange C, Feillet-Coudray C, Ducros V, Pépin D, Brouns F, Rayssiguier AM, Coudray C (2001) Five-week intake of short-chain fructo-oligosaccharides increases intestinal absorption and status of magnesium in postmenopausal women. J Bone Miner Res 16:2152–2160. https://doi.org/10.1359/jbmr.2001.16.11.2152
Tang X, Ma S, Li Y, Sun Y, Zhang K, Zhou Q, Yu RA-O (2020) Evaluating the activity of sodium butyrate to prevent osteoporosis in rats by promoting Osteal GSK-3β/Nrf2 signaling and mitochondrial function. J Agric Food Chem 68:6588–6603
van den Heuvel EG, Muijs T, Brouns F, Hendriks HF (2009) Short-chain fructo-oligosaccharides improve magnesium absorption in adolescent girls with a low calcium intake. Nutr Res 29:229–237. https://doi.org/10.1016/j.nutres.2009.03.005
Whisner CM, Martin BR, Schoterman MH, Nakatsu CH, McCabe LD, McCabe GP, Wastney ME, van den Heuvel EG, Weaver CM (2013) Galacto-oligosaccharides increase calcium absorption and gut bifidobacteria in young girls: a double-blind cross-over trial. Br J Nutr 110:1292–1303. https://doi.org/10.1017/s000711451300055x
Yamazaki D, Funato Y, Miura J, Sato S, Toyosawa S, Furutani K, Kurachi Y, Omori Y, Furukawa T, Tsuda T, Kuwabata S, Mizukami S, Kikuchi K, Miki H (2013) Basolateral Mg2+ extrusion via CNNM4 mediates transcellular Mg2+ transport across epithelia: a mouse model. PLoS Genet 9:e1003983
Acknowledgements
The authors thank H. Janssen-Wagener (Radboud university medical center, Nijmegen, the Netherlands) and S. van Boxtel (Radboud University Medical Center, Nijmegen, the Netherlands) for their excellent technical support with the animal study.
Funding
This work was supported by grants from the Netherlands Organization for Scientific Research (J. Hoenderop, NWO VICI 016.130.668 and J. de Baaij, NWO VENI 016.186.012).
Author information
Authors and Affiliations
Contributions
L.M.M.G., J.H.Fd.B., Jvd.W., and J.G.J.H. conceived the study, and L.M.M.G., J.H.Fd.B., Jvd.W., C.B., R.J.M.B., and J.G.J.H. designed the experiments and interpreted the data. L.M.M.G., J.H.Fd.B., C.B., and L.J. performed the experiments and analyzed the data. L.M.M.G., J.H.Fd.B., Jvd.W., and J.G.J.H. wrote the paper. All authors edited and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
This animal study was approved by the animal ethics board of the Radboud University Nijmegen (RU DEC 2017–0024) and by the Dutch Central Commission for Animal Experiments (CCD, AVD1030020173224).
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gommers, L.M.M., van der Wijst, J., Bos, C. et al. Colonic expression of calcium transporter TRPV6 is regulated by dietary sodium butyrate. Pflugers Arch - Eur J Physiol 474, 293–302 (2022). https://doi.org/10.1007/s00424-021-02648-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-021-02648-6