Abstract
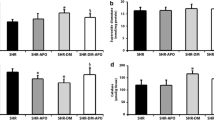
The intermediate-conductance Ca2+-activated K+ (KCa3.1) channels play a pivotal role in the cardiac fibroblast proliferation and inflammatory reaction during the progression of myocardial fibrosis. However, the relationship between KCa3.1 expression and oxidative stress, the important factor of promoting fibrosis, has not been clearly established. This study was designed to investigate whether the role of oxidative stress in promoting myocardial fibrosis is related to KCa3.1 channel by using biochemical approaches. It was found that mean blood pressure, plasma Ang II level, and myocardium malondialdehyde (MDA) content of angiotensinogen-renin (AGT-REN) double transgenic hypertension (dTH) mice were higher than those in wild-type (WT) mice of the same age (4, 8 and 12 months) and were significantly increased with age. However, plasma Ang (1–7) level and myocardium superoxide dismutase (SOD) activity showed a downward trend and were lower than those of the same-aged WT mice (4, 8 and 12 months). In addition, protein expression of myocardium KCa3.1 channel in 4-, 8-, and 12-month-old dTH mice were significantly higher than that of the same-aged WT mice and gradually increased with age. TRAM-34, a blocker of KCa3.1 channel, and losartan mitigated the myocardial structural and functional damage by inhibiting collagen deposition and decreasing the expression of β-MHC. After intervention of ROS scavenger N-acetyl cysteine (NAC) and NADPH inhibitor apocynin (Apo) in 6-month-old dTH mice for 4 weeks, myocardial oxidative stress level was reduced and KCa3.1 channel protein expression was decreased. Meanwhile, Apo inhibited the myocardium p-ERK1/2/T-ERK protein expression in dTH mice, and after blockage of ERK1/2 pathway with PD98059, the KCa3.1 protein expression was reduced. These results demonstrate for the first time that KCa3.1 channel is likely to be a critical target on the oxidative stress for its promoting role in myocardial fibrosis, and the ERK1/2 pathway may be involved in the regulation of oxidative stress to KCa3.1.





Similar content being viewed by others
References
Adebiyi OA, Adebiyi OO, Owira PM (2016) Naringin reduces hyperglycemia-induced cardiac fibrosis by relieving oxidative stress. PLoS One 11(3):e0149890
Albaqumi M, Srivastava S, Li Z, Zhdnova O, Wulff H, Itani O, Wallace DP, Skolnik EY (2008) KCa3.1 potassium channels are critical for cAMP dependent chloride secretion and cyst growth in autosomal-dominant polycystic kidney disease. Kidney Int 74(6):740–749
Asbun J, Villarreal FJ (2006) The pathogenesis of myocardial fibrosis in the setting of diabetic cardiomyopathy. J Am Coll Cardiol 47(4):693–700
Choi S, Kim JA, Na HY, Kim JE, Park S, Han KH, Kim YJ, Suh SH (2013) NADPH oxidase 2-derived superoxide downregulates endothelial KCa3.1 in preeclampsia. Free Radic Biol Med 57:10–21
Crosas-Molist E, Bertran E, Sancho P, López-Luque J, Fernando J, Sánchez A, Fernández M, Navarro E, Fabregat I (2014) The NADPH oxidase NOX4 inhibits hepatocyte proliferation and liver cancer progression. Free Radic Biol Med 69:338–347
Díez J (2007) Mechanisms of cardiac fibrosis in hypertension. The Journal of Clinical Hypertension 9(7):546–550
Freise C, Heldwein S, Erben U, Hoyer J, Köhler R, Jöhrens K, Patsenker E, Ruehl M, Seehofer D, Stickel F, Somasundaram R (2015) K+-channel inhibition reduces portal perfusion pressure in fibrotic rats and fibrosis associated characteristics of hepatic stellate cells. Liver Int 35(4):1244–1252
Fu RG, Zhang T, Wang L, Du Y, Jia LN, Hou JJ, Yao GL, Liu XD, Zhang L, Chen L, Gui BS, Xue RL (2014) Inhibition of the K(+) channel KCa3.1 reduces TGF-β1-induced premature senescence, myofibroblast phenotype transition and proliferation of mesangial cells. PLoS One 9(1):e87410
Huang C, Shen S, Ma Q, Chen J, Gill A, Pollock CA, Chen XM (2013) Blockade of KCa3.1 ameliorates renal fibrosis through the TGF-β1/Smad pathway in diabetic mice. Diabetes 62(8):2923–2934
Huang C, Shen S, Ma Q, Gill A, Pollock CA, Chen XM (2014) KCa3.1 mediates activation of fibroblasts in diabetic renal interstitial fibrosis. Nephrol Dial Transplant 29(2):313–324
Huang C, Lin MZ, Cheng D, Braet F, Pollock CA, Chen XM (2016) KCa3.1 mediates dysfunction of tubular autophagy in diabetic kidneys via PI3k/Akt/mTOR signaling pathways. Sci Rep 6:23884
Ju CH, Wang XP, Gao CY, Zhang SX, Ma XH, Liu C (2015) Blockade of KCa3.1 attenuates left ventricular remodeling after experimental myocardial infarction. Cell Physiol Biochem 36(4):1305–1315
Kimura S, Mullins JJ, Bunnemann B, Metzger R, Hilgenfeldt U, Zimmermann F, Jacob H, Fuxe K, Ganten D, Kaling M (1992) High blood pressure in transgenic mice carrying the rat angiotensinogen gene. EMBOJ 11(3):821–827
Köhler R, Oliván-Viguera A, Wulff H (2016) Endothelial small- and intermediate-conductance K channels and endothelium-dependent hyperpolarization as drug targets in cardiovascular disease. Adv Pharmacol 77:65–104
Lijnen PJ, van Pelt JF, Fagard RH (2012) Stimulation of reactive oxygen species and collagen synthesis by angiotensin II in cardiac fibroblasts. Cardiovasc Ther 30(1):e1–e8
Liu XH, Pan LL, Deng HY, Xiong QH, Wu D, Huang GY, Gong QH, Zhu YZ (2013) Leonurine (SCM-198) attenuates myocardial fibrotic response via inhibition of NADPH oxidase 4. Int J Cardiol 168(4):3770–3778
Maki S, Miyauchi T, Sakai S, Kobayashi T, Maeda S, Takata Y, Sugiyama F, Fukamizu A, Murakami K, Goto K, Sugishita Y (1998) Endothelin-1 expression in hearts of transgenic hypertensive mice overexpressing angiotensin II. J Cardiovasc Pharmacol 31:S412–S416
Ogata T, Miyauchi T, Sakai S, Takanashi M, Irukayama-Tomobe Y, Yamaguchi I (2004) Myocardial fibrosis and diastolic dysfunction in deoxycorticosterone acetate-salt hypertensive rats is ameliorated by the peroxisome proliferator-activated receptor-alpha activator fenofibrate, partly by suppressing inflammatory responses associated with the nuclear factor-kappa-b pathway. J Am Coll Cardiol 43(8):1481–1488
Pan LL, Liu XH, Shen YQ, Wang NZ, Xu J, Wu D, Xiong QH, Deng HY, Huang GY, Zhu YZ (2012) Inhibition of NADPH oxidase 4-related signaling by sodium hydrosulfide attenuates myocardial fibrotic response. Molecules 17(3):2738–2751
Pan LL, Liu XH, Shen YQ, Wang NZ, Xu J, Wu D, Xiong QH, Deng HY, Huang GY, Zhu YZ (2013) Inhibition of NADPH oxidase 4-related signaling by sodium hydrosulfide attenuates myocardial fibrotic response. Int J Cardiol 168(4):3770–3778
Papaharalambus CA, Griendling KK (2007) Basic mechanisms of oxidative stress and reactive oxygen species in cardiovascular injury. Trends Cardiovasc med 17(2):48–54
Pitt B, Zannad F (2012) The detection of myocardial fibrosis: an opportunity to reduce cardiovascular risk in patients with diabetes mellitus? Circulation: Cardiovascular Imaging 5(1):9–11
Privratsky JR, Wold LE, Sowers JR, Quinn MT, Ren J (2003) AT1 blockade prevents glucose-induced cardiac dysfunction in ventricular myocytes: role of the AT1 receptor and NADPH oxidase. Hypertension 42(2):206–212
Roach KM, Duffy SM, Coward W, Feghali-Bostwick C, Wulff H, Bradding P (2013) The K+ channel KCa3.1 as a novel target for idiopathic pulmonary fibrosis. PLoS One 8(12):e85244
Roach KM, Wulff H, Feghali-Bostwick C, Amrani Y, Bradding P (2014) Increased constitutive αSMA and Smad2/3 expression in idiopathic pulmonary fibrosis myofibroblasts is KCa3.1-dependent. Respir Res 15:155
Roach KM, Feghali-Bostwick C, Wulff H, Amrani Y, Bradding P (2015) Fibrogenesis tissue repair. Human lung myofibroblast TGFβ1-dependent Smad2/3 signalling is Ca(2+)-dependent and regulated by KCa3.1K(+) channels. Fibrogenesis Tissue Repair 8:5
Sevelsted Møller L, Fialla AD, Schierwagen R, Biagini M, Liedtke C, Laleman W, Klein S, Reul W, Koch Hansen L, Rabjerg M, Singh V, Surra J, Osada J, Reinehr R, de Muckadell OB, Köhler R, Trebicka J (2016) The calcium-activated potassium channel KCa3.1 is an important modulator of hepatic injury. Sci Rep 6:28770
Sherrod M, Davis DR, Zhou X, Cassell MD, Sigmund CD (2005) Glial-specific ablation of angiotensinogen lowers arterial pressure in renin and angiotensinogen transgenic mice. Am J Physiol Regul Integr Comp Physiol 289(6):R1763–R1769
Somanna NK, Valente AJ, Krenz M, Fay WP, Delafontaine P, Chandrasekar B (2016) The Nox1/4 dual inhibitor GKT137831 or Nox4 knockdown inhibits angiotensin-II-induced adult mouse cardiac fibroblast proliferation and migration. AT1 physically associates with Nox4. J Cell Physiol 231(5):1130–1141
Tordjman KM, Semenkovich CF, Coleman T, Yudovich R, Bak S, Osher E, Vechoropoulos M, Stern N (2007) Absence of peroxisome proliferator-activated receptor-abolishes hypertension and attenuates atherosclerosis inthe Tsukuba hypertensive mouse. Hypertension 50(5):945–951
Touyz RM, Briones AM (2011) Reactive oxygen species and vascular biology: implications in human hypertension. Hypertens Res 34:5–14
Tsutsui H, Kinugawa S, Matsushima S (2011) Oxidative stress and heart failure. Am J Physiol Heart Circ Physiol 301(6):H2181–H2190
Wam NC, CroR KD (2006) Hypertension and oxidative stress. ClinicalExperimental Pharmacology Physiology 33(9):872–876
Wang LP, Wang Y, Zhao LM, Li GR, Deng XL (2013) Angiotensin II upregulates KCa3.1 channels and stimulates cell proliferation in rat cardiac fibroblasts. Biochem Pharmacol 85(10):1486–1494
Wang J, Hong Z, Zeng C, Yu Q, Wang H (2014) NADPH oxidase 4 promotes cardiac microvascular angiogenesis after hypoxia/reoxygenation in vitro. Free Radic Biol Med 69:278–288
Yang CM, Lee IT, Hsu RC, Chi PL, Hsiao LD (2013) NADPH oxidase/ROS-dependent PYK2 activation is invol ved in TNF-α-induced matrix metalloproteinase-9 expression in rat heart-derived H9c2 cells. Toxicol Appl Pharmacol 272(2):431–442
Yu M, Zheng Y, Sun HX, Yu DJ (2012) Inhibitory effects of enalaprilat on rat cardiac fibroblast proliferation via ROS/P38MAPK/TGF-β1 signaling pathway. Molecules 17(3):2738–2751
Zhao W, Zhao D, Yan R, Sun Y (2009) Cardiac oxidative stress and remodeling following infarction: role of NADPH oxidase. Cardiovasc Pathol 18(3):156–166
Zhao LM, Zhang W, Wang LP, Li GR, Deng XL (2012) Advanced glycation end products promote proliferation of cardiac fibroblasts by upregulation of KCa3.1 channels. Pflug Arch 464(6):613–621
Zhao LM, Wang LP, Wang HF, Ma XZ, Zhou DX, Deng XL (2015) The role of KCa3.1 channels in cardiac fibrosis induced by pressure overload in rats. Pfluger Arch 467:2275–2285
Zhuang Y, Feng Q, Ding G, Zhao M, Che R, Bai M, Bao H, Zhang A, Huang S (2014) Activation of ERK1/2 by NADPH oxidase-originated reactive oxygen species mediates uric acid-induced mesangial cell proliferation. Am J Physiol Renal Physiol 307(4):F396–F406
Acknowledgments
The work was supported by National Natural Science Foundation of China (grant nos. 81600316 and 81372029) and the Natural Science Foundation of Hebei Province (grant no. H2015209153).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Experimental protocols were approved by the Institutional Animal Care and Use Committee of North China University of Science and Technology and conformed to the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health, USA.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Ion channels, receptors, and transporters
Electronic supplementary material
Fig. S1
The PCR result of AGT-REN double transgenic hypertensive mice. 10–14 days after birth, postnatal mice underwent toe-clipping for numbering and clipping of the tip of tail for identification. Primer sequence: AGT (441 bp) L: CACCCCAGAGCACCATTACT; R: TGGAGCTGTAGCGTGTCATC. REN (545 bp) L: AACCACTGCTTCACCACCG; R: CACACTACCCTTTCCTTCCTACATC. Results are shown in S1 Fig; water was used as the blank control, WT as the negative control, 1–9 were AGT-REN double transgenic hypertensive mice and 10 as the positive control of AGT and REN, respectively. (GIF 60 kb)
Rights and permissions
About this article
Cite this article
Wang, LP., Fan, SJ., Li, SM. et al. Oxidative stress promotes myocardial fibrosis by upregulating KCa3.1 channel expression in AGT-REN double transgenic hypertensive mice. Pflugers Arch - Eur J Physiol 469, 1061–1071 (2017). https://doi.org/10.1007/s00424-017-1984-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-017-1984-0