Abstract
Purpose
Despite mobilization is highly recommended in the ERAS® colorectal guideline, studies suggest that more than half of patients don’t reach the daily goal of 360 min out of bed. However, data used to quantify mobilization are predominantly based on self-assessments, for which the accuracy is uncertain. This study aims to accurately measure postoperative mobilization in ERAS®-patients by validated motion data from body sensors.
Methods
ERAS®-patients with elective bowel resections were eligible. Self-assessments and motion sensors (movisens: ECG-Move 4 and Move 4; Garmin: Vivosmart4) were used to record mobilization parameter from surgery to postoperative day 3 (POD3): Time out of bed, time on feet and step count.
Results
97 patients were screened and 60 included for study participation. Self-assessment showed a median out of bed duration of 215 min/day (POD1: 135 min, POD2: 225 min, POD3: 225 min). The goal of 360 min was achieved by 16.67% at POD1, 21.28% at POD2 and 20.45% at POD3. Median time on feet objectively measured by Move 4 was 109 min/day. During self-assessment, patients significantly underestimated their “time on feet”-duration with 85 min/day (p = 0.008). Median number of steps was 933/day (Move 4).
Conclusion
This study confirmed with objectively supported data, that most patients don’t reach the daily mobilization goal of 360 min despite being treated by an ERAS®-pathway with ERAS®-nurse. Even considering an empirically approximated underestimation, the ERAS®-target isn’t achieved by more than 75% of patients. Therefore, we propose an adjustment of the general ERAS®-goals into more patient-centered, individualized and achievable goals.
Registration
This study is part of the MINT-ERAS-project and was registered prospectively in the German Clinical Trials Register on 25.02.2022. Trial registration number is “DRKS00027863”.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Enhanced Recovery after Surgery (ERAS®) is an evidence-based and multimodal perioperative treatment strategy to improve the recovery process after surgery [1, 2]. In colorectal surgery, compliant ERAS®-treatment can achieve a halved postoperative complication rate [3] and a shortened hospital stay of ~ 2.5 days [4]. Early postoperative mobilization is one of the core components in the ERAS® guidelines for colorectal surgery [5, 6]. Comprehensive mobilization is an independent predictor for reduced moderate to severe complications [3] and a reduction of inpatient length of stay [7]. The most recent duration-specific ERAS® mobilization goals were published in the 3rd updated guidelines from 2013: a cumulative duration of 2 h out of bed on the day of surgery and 6 h from post-op day 1 (POD1) [8, 9]. In the latest 4th updated ERAS® guidelines from 2018 for colorectal surgery, mobilization is still declared with a “strong” recommendation-level, but without specification regarding the extent [6]. The ERAS® Interactive Audit System (EIAS) still uses the compliance target of 2 h on the day of surgery and 6 h from POD1 to assess protocol adherence [10, 11]. Studies have shown that mobilization is the ERAS® component with the poorest adherence [12, 13]. Grass et al. showed that 58% of 1170 ERAS® patients did not reach the 360 min out of bed on POD1 [14], in the study of Gustafsson et al. (n = 664) it was even higher at 72.5% [15]. Therefore, it can be assumed that at least half of the ERAS® patients did not reach the 360 min out of bed daily target.
There is evidence in goal-setting research that unrealistically high goals can be counterproductive [16,17,18]. Höpfner and Keith showed experimentally that failure to achieve an excessively high target was associated with a decrease in self-confidence and motivation [19]. This study therefore takes a critical look at whether the general ERAS® mobilization target is useful for the majority of patients.
However, the fact that the latest 4th ERAS® guidelines avoid specifying targets is also problematic, because there is strong evidence that specific targets positively influence patient behavior [17, 20]. In addition, the lack of specific mobilization-instructions in ERAS® guidelines contributes to clinicians interpreting “early mobilization” differently [21]. Hence, the mobilization target of the EIAS database (360 min out of bed/day) is not used consistently in the ERAS® literature [22]. For example, some ERAS®-studies used to assess adherence the ability to walk at POD1 [12], active mobilization on POD1 [23] or successful mobilization to a chair within 12 h after surgery [3].
A general problem in assessing mobilization adherence is that data are predominantly based on subjective assessments by nurses or patients [24]. Since no objective data were collected, it is unclear how accurate subjective assessments really are.
Hence, in the present study evaluation we focused on accurate, multi-sensor patient activity assessment to analyze postoperative mobilization more objectively. Finally, those data should allow to critically discuss the feasibility and patient-relevance of the ambitious ERAS® postoperative mobilization goal of 360 min/day.
Material & methods
Participants
Patients undergoing elective bowel surgery at the University Hospital Mannheim were eligible.
The inclusion criteria were ≥ 18 years, cognitive abilities to understand the study and informed consent, consent to treatment according to the ERAS® protocol, Barthel index ≥ 10 points for mobility (assisted walking > 50 m). Reasons for exclusion were inability to have a fluent conversation (language barrier, hearing problems, mental states), previous study participation, PEG or parenteral nutrition, cardiac devices and expected incompliance concerning the protocol requirements (especially handling with technical devices).
A sample size of 50 patients was planned for this pilot-study design.
Study design & procedure
This study evaluation is part of the MINT-ERAS® project, a monocenter pilot-RCT project investigating the effect of a specific patient conversation style (Motivational Interviewing/MI [25]) on postoperative ERAS® mobilization and nutrition goals. The results regarding the impact of MI on early mobilization are published in a separate detailed report [26]. In the present study evaluation – focusing on the overall mobilization data of ERAS®-patients recorded by three different body-motion sensors – data from both the intervention and the control group were analyzed together as a single-arm study.
The subjects were treated according to the certified ERAS®-pathway for colorectal surgery at the University Hospital Mannheim [27]. Prior to surgery, patients were educated as usually by the ERAS®-nurse on the objectives and process of ERAS®. The ERAS®-nurse is specifically responsible for the ERAS®-patients and cared for them daily after surgery (excluding weekends).
After study enrollment, participants were instructed on handling the technical equipment a few days before surgery (usually at the anesthesiologist appointment), and ERAS® objectives were revisited. At the daily postoperative visits from POD0 to POD3 (usually between 6:00 pm and 8:30 pm, see Table 1), the sensors were checked and general data were asked (pain, nausea, activity, etc.). At the following day’s visit, data from the previous evening were added (e.g., if a patient was still out of bed for 30 min after the visit at POD1, it was recorded at the POD2 visit). At POD0, the three motion sensors were attached to the study participants, and at POD3 after 8 pm, they were removed. A telephone follow-up was conducted 28–32 days after surgery to complete questionnaires.
Measuring devices
For objective activity profiling, three motion sensors (Garmin Vivosmart 4, Move 4 and ECG Move 4) were used and recorded data from 11:59 pm on day 0 for 68 h until 8:00 pm on POD3.
The commercial tool Garmin Vivosmart 4 (GVS4) [28] was worn on the wrist (see Fig. 1a). The GVS4 is waterproof, has a battery life of up to 7 days, and weighs between 16.5 g and 17.1 g. For analyzing step count, the GVS 4 was synchronized via Bluetooth with the “Garmin Connect” app [29] of a study smartphone.
The Move 4 [30] and EKG Move 4 [31] activity sensors were developed by movisens GmbH (Karlsruhe, Germany) and have been validated concerning step count (mean percentage deviation 0,6%) [32] and activity classes (accuracy 98,2%) [33] for scientific research purposes. The sensors are waterproof, the battery life is at least 3 days, they have a size of 62.3 mm x 38.6 mm x 11.5 mm and weigh 25–26 g. During the measurement, integrated sensors (3D acceleration, rotation, pressure, temperature) continuously record raw physical data, the ECG Move 4 also includes an ECG sensor. The configuration of the sensors was done by the software “Sensor Manager“ [34]. The sensors were attached with adhesive electrodes, the Move 4 vertically on the lateral right thigh (see Fig. 1b), the ECG Move 4 horizontally from the xiphoid to the underside of the left chest (see Fig. 1c). Body hair was shaved beforehand. The electrodes were checked for tightness during the daily visit and replaced if necessary. The “Data Analyzer” calculates physiological parameters such as activity classes (lying, sitting, standing, walking), number of steps, and cardiac parameters (heart rate, heart rate variability) from the raw data and presents them in analysis overviews [35]. To register steps and the activity class “walking”, at least three consecutive steps must be taken (time to take one step 0,2–2 s each) [36].
Endpoints
To measure mobilization, the ERAS® compliance target “time out of bed” POD0–POD3 (patient self-assessment) was recorded. To specify the activity profile and simultaneously support it with objective values, the number of steps (objective by Move 4, ECG Move 4 & GVS4) and the time on feet (subjective and objective by Move 4, sum of activity classes standing + walking) of POD1–POD3 were measured. By measuring “time on feet” subjectively as well as objectively, it was intended to draw conclusions about the accuracy of self-assessments by calculating the deviations. All data on POD3 refer to the period from 0:00 a.m. – 8:00 p.m. and all data on POD0 refer to the period after the end of surgery.
Further endpoints were mobilization at POD0 (arrival time on ward, mobilized yes/no, duration until first mobilization), metabolic equivalent (POD1–POD3), pain (maximum/average) as well as complications (POD0–POD3) and duration of hospital stay. Questionnaires were used to assess activity before surgery (IPAQ - International Physical Activity Score [37], pre-op), patient satisfaction (PEQ – Patient Experience Questionnaire [38], POD28–32), activity of daily living (Barthel-Index [39], pre-op und POD3) and Quality of Recovery (QoR-15 – Quality of Recovery [40], pre-op, POD 1/3/28–32). Minimum and maximum achievable scores are PEQ (0–100), Barthel (0–100), QoR-15 (0–150). In addition, correlation between postoperative “time on feet” (Move 4) and preoperative activity (IPAQ) or age was tested.
Statistical analysis
Only complete data sets were included for the statistical analysis of the individual days. For example, if patients were already discharged on POD3, they were included in the analyses from op-day to POD2, but not on POD3.
Continuous data are presented as median [quartile range = Q1-Q3] [range = minimum-maximum] and/or arithmetic mean ± standard deviation. For comparison of metric-scaled characteristics “time on feet” subjective & objective, Wilcoxon test for paired samples was used. Correlations were presented using Spearman’s rank correlation coefficient. A p-value < 0.05 (two-sided test) is considered statistically significant. SAS® software (SAS Institute, Inc., USA) was used for statistical analyses.
Results
Personal data & sample
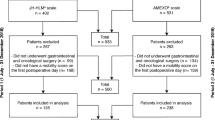
Between March 2022 and July 2022 97 patients were screened. 60 patients participated in the final study. Originally, only 50 study participants were planned as pilot study sample size, but due to some incomplete data sets, the sample size was increased to 60 during the recruitment period (approved by the ethics committee). According to intention-to-treat principle all 60 randomized study participants were included in the statistical analyses. The following deviations exist: The self-assessment “time out of bed” was collected from POD3 onwards by subjects 11 & 12. Reduced samples on individual days are due to one death on POD2 and five discharges on POD3. In addition, during the step count by GVS4 at POD3, the battery ran out for two subjects and once the fitness wristband was not worn continuously. Since the Move 4 fell of a few times during the initial phase (without data loss) when putting on / taking off pants, it was additionally covered with an adhesive film. Apart from this, all study participants were compliant with the protocol requirements and wore the motion sensors throughout day and night.
The study participants had an average age of 60.7 ± 13.3 years (range = 30–82 y.), BMI of 26.4 ± 5, and 56.67% were male (see Table 2). 75% of the subjects were classified ASA II, 21.67% ASA III, and 3.33% ASA I. The mean duration of surgery was 216.7 ± 110.8 min, and 35% of the patients received a stoma during surgery.
63.33% of procedures were minimal-invasive (laparoscopic or daVinci), 33.33% were primarily open, and 3.33% were converted (see Table 2). The two most common surgical indications were malignant neoplasms (46.67%) and stoma-reversal / restoration of bowel continuity after Hartmann’s Procedure (35%). The study included a wide range of bowel surgical procedures (see Table 2). Apart from one procedure (pelvic tumor debulking after rectal carcinoma), bowel resection was performed in all other operations.
Time out of bed POD1 – POD3 (Table 3)
Across POD1 to POD3, study participants spent a median of 215 min/day out of bed. At POD1, the median mobilization time was 135 min; at POD2 and POD3, it increased to 225 min. The ERAS® compliance target of 360 min was reached by less than 25% on all three days (POD1: 16.67%, POD2: 21.28%, POD3: 20.45%).
Time on feet (self-assessment & objective; Table 4)
Objectively measured by Move 4, study participants were median 109 min/day (POD 1–3) on their feet (standing and walking), compared with only 85 min/day in the self-assessment. Participants significantly underestimated themselves (⌀ / day, p < 0.008). Across POD1–3, the median deviation in self-assessment compared with the objective score was − 17.3 min/day (≙–15.87% of 109 min/day).
Step count (Table 5)
The motion sensors measured a median of 933 steps/day (Move 4), 964 steps/day (ECG Move 4), and 1800 steps/day (GVS4) across the period of POD1 to POD3. The GVS4 collected a higher number of steps than the two movisens sensors on all three days. The data of all three sensors are right-skewed distributed, recognizable by the higher mean values (compared to the median), the smaller distance of the median to Q1 than to Q3, and the high maxima (up to 21557 steps for GVS4 at POD3). The cumulative range (POD1–POD3) measured by Move 4 is 0–52632 steps.
Mobilization POD0
The median [Q1-Q3] time point at which the subjects arrived on the ward from the recovery room was 5:10 pm [2:28pm-7:05pm], minimum was 11:15 am and maximum 10:31 pm. 33 of the 60 subjects were mobilized to their feet on the day of surgery. Of the 33 subjects who were mobilized, a median [Q1-Q3] of 15 min [5–30] was spent out of bed on POD0, the maximum was 120 min. Based on all 60 study participants, the median [Q1-Q3] duration from the end of surgery (suture) to the first mobilization was 430 min [261–966]; only one person was mobilized for the first time on POD2 (after 2232 min).
Other outcome-variables (Tables 6 and 7)
On median, subjects had a MET level < 1.5 (inactive) at POD1 98.65% of the time ( ≙ 23 h + 19.5 min), then through POD2 (96.46%) and POD3 (95.54%) the “inactive” proportion of time decreased continuously (see Table 6). 65% of the subjects achieved the activity class “high” in the IPAQ, 30% “medium” and only 5% “low” (see Table 7). Absolute MET level in the IPAQ (r = − 0.133, p = 0.339) and age (r = + 0.251, p = 0.067) showed no significant relationship with cumulative (POD1–POD3) time on feet. In the questionnaire “Quality of Recovery”, the lowest median was reached after surgery at POD1 (106.5) and then increased through POD3 (114) to the highest value on POD28–32 (131), which was higher than the baseline preoperatively (128). Postoperative complications occurred once at POD0 (1x Clavien-Dindo (CD) Class I), three times at POD1 (2x CD I, 1x CD III), twice at POD2 (1x CD I, 1x CD V), and twice at POD3 (2x CD II). The median [Q1-Q3] regarding the patient-reported maximum pain peak (scale 0–10) was 5 [3–8] at POD0, 6.5 [4–8] at POD1, 4 [3–6] at POD2, and 4 [2–6] at POD3. Regarding the pain average over the whole day, the median [Q1-Q3] at POD0 was 4 [2–5] at POD1 3 [2.5-5], at POD2 3 [1–4], and at POD3 2 [1–4]. Postoperative hospital stay had a median of 5 days [Q1-Q3: 4–7; range: 3–20].
Discussion
Several previous studies have shown that a high percentage of patients fail to achieve the ambitious ERAS® compliance goal of 360 min/day out of bed [3, 12, 14, 15]. However, since the previous data were collected by subjective assessments, the accuracy of these data is unclear so far. To measure mobilization as accurately as possible in this study, we recorded activity with three different body sensors and compared it with self-assessment. With the data from the present study, we could show that less than a quarter of ERAS®-patients achieved the ERAS® compliance goal of 360 min/day out of bed. The median time out of bed across the first three postoperative days was 215 min/day, more than 2 h away from the general ERAS® mobilization target.
Within the ERAS®-program at the Universitätsmedizin Mannheim, there are specially trained physiotherapists and an ERAS®-nurse; additionally, all nursing staff on the ward have undergone ERAS®-training. Patients receive a detailed informational talk and an ERAS®-brochure preoperatively, which also include patient requirements (among other things to be 360 min out of bed from POD1). Furthermore, it should be considered that fitness wristbands can increase physical activity in hospitalized patients [41], especially when visual feedback is provided via a display [24]. Since we used fitness wristbands providing feedback in our study, it can be assumed that mobilization in this work tended to be even higher than in the broad range of ERAS®-treatments without study conditions. So, the patients had all the important information, the multidisciplinary staff was ERAS® trained and all patients were additionally equipped with mobilization-enhancing fitness wristbands.
Despite this described setting, we could show with objectively supported data that only about 20% achieved the ERAS® compliance target of 360 min out of bed. Objectively supported means that the accuracy of patient self-assessments has been checked with motion sensors. By recording “time on feet” (standing & walking) by self-assessment as well as objectively by the sensors, the accuracy of subjective data could be approximated in the present study. Due to the systematic underestimation (at all 3 PODs) of the parameter “time on feet”, it can be assumed that patients also underestimated themselves by about 15.87% for “time out of bed”. However, if 15.87% is added to Q3 of each POD1–3, the third quartile remains below 360 min at all three PODs. Thus, even taking underestimation into account, less than 25% met the ERAS® compliance target.
In our study, a mobilization adherence (≥ 360 min/day) of 16.67% was achieved on POD1. Compared with other studies, adherence was between 23.5% and 27.5% observed by Gustafsson et al. [15]. , 43,9% observed by Schwenk et al. [13]. and 42% observed by Grass et al. [14]. Adherence in Grass et al. increased to 58% at POD2 (vs. 21.28% in our study) and even to 69% at POD3 (vs. 20.45% in our study, cave: only until 8:00 pm). The higher values of Grass et al. might be surprising considering that in our study (in contrast to Grass et al.) the above-mentioned mobilization-promoting measures were applied. However, the 1170 records in the study of Grass et al. were analyzed retrospectively and are based only on subjective assessments of the nursing staff. In addition, when comparing individual monocentric studies, the workflow may vary clearly between the clinics.
55% of the subjects got up for the first time on the day of the operation, which is similar to the values from other studies with 45% [14], 48.39% [3], 54.5% [42] and 59,6% [13]. Subjects mobilized at POD0 spent a median of 15 min out of bed, with only one person reaching the ERAS® compliance target of 120 min. A main reason for the lack of mobilization at POD0 is probably the end of surgery respectively the time at which the patients arrive on the ward. With a median time point of 5:10 pm, more than half of the patients arrive on the ward from the recovery room after the usual working hours of the ERAS®-nurse and physiotherapists. Nevertheless, it should be noted, that despite poor adherence regarding the ERAS® mobilization goals, one of the main goals of ERAS® (early discharge) was successfully achieved in our study with a median postoperative hospital stay of 5 days.
Even though the ERAS® compliance target of the EIAS database only refers to “time out of bed”, it can be assumed that components of moderate activity on feet (standing & walking) are superior to sitting only [9]. For example, the number of steps has been shown to correlate negatively with length of hospital stay [24] and complications [43]. Time on feet (standing & walking) between POD1–POD3 in our study was 109 min/day (objective), comparatively higher than in an abdominal surgery study (n = 118; neoplasms colorectal, ovarian, or urinary bladder) with objective values ranging from 42 min (control) to 78 min (intervention) per day [44].
It is striking that the commercial tracker GVS 4 counted considerably more steps than the two scientific research sensors from Movisens (∑ POD1–3: 4420 vs. 2758/2566). The reason for this are programming rules in movisens: steps are only counted when three consecutive steps are taken [36]. This minimizes noise and avoids counting “false steps”; however, in the postoperative setting, it can also cause that very “careful steps” (slow, small, isolated) are not registered adequately. The GVS4 is more sensitive, but in return the noise is much higher. For example, GVS4 already registered a few steps of participants, even though they had not yet stood up at that time.
In this study, unfortunately, it was not possible to calculate the time out of bed completely objectively (by the sum of the activity classes sitting, standing and walking) for two reasons: Firstly, the ECG Move 4 registers the activity class “sitting” at a horizontal upper body angle of > 30 degrees, and therefore periods with the backrest raised in bed were also registered as “sitting” (e.g. for reading, with gastric tube, sometimes also during sleeping, etc.). Increasing the angle for registering “sitting” is also problematic, since in that case patients sitting slightly at an angle could be registered as lying down. Second, even with adequate “sitting” registration, it is not possible to distinguish whether a patient is really sitting outside the bed (ERAS®-target) or merely at the edge of the bed. For these reasons, self-assessment of “time out of bed” was recorded from the eleventh study participant onwards, which was the main reason for a reduced sample size together with early discharges on POD3. Flavio Fiore et al. measured time out of bed both by the ActiGraph (sitting + standing + walking) and by self-assessment [42]. In the results, the objective measurement by the ActiGraph is up to 100% higher in median than the self-assessment; here the authors also discussed an incorrect classification of activity classes due to an elevated backrest. There are two ideas on how time out of bed could be measured completely objectively in future studies. The first idea is a technical localization of the participants, so that time periods outside a pre-programmed area around the patient’s bed are measured as “time out of bed”. The second idea are pressure-sensitive overlays for the bed, which can record periods without weight influence (threshold e.g. at 15 kg, so that bedspread, books, bags, etc. are not registered).
In summary, the data from this study, in comparison to the relevant literature, demonstrated that the current ERAS® compliance target of the EIAS database (360 min out of bed) is not achieved by most patients. There is evidence that setting goals too high [16] and failure to achieve goals [19] have a negative impact on parameters such as self-confidence and motivation, which can negatively affect performance [18, 45]. It is noteworthy that the current ERAS®-guidelines of colorectal surgery do not specify daily mobilization goals at all. This may lead to inconsistent implementation between hospitals [21, 22]. Additionally, specific and achievable goals have positive effects on patient engagement [20]. It was shown, with a medium effect size (d = 0.42 to d = 0.56), that more specific goals lead to better performance than no specification or “do your best” goals [17]. In the literature, the best results are achieved when goals are difficult but reachable [17, 18].
In this paper, we will propose individualization of daily ERAS® mobilization goals as a possible effective strategy. Since in our study postoperative activity did not correlate significantly with the age or subjective preoperative activity (IPAQ), these parameters should only play a minor role in individualization. Interestingly, the positive correlation between “age” and “time on feet” just missed significance (p = 0.067), which means that in tendency older people were even slightly longer on feet than younger people. A meaningful individualization could be done by the ERAS®-nurse in cooperation with the patients in line with the shared decision making (SDM) approach [46]. In their review, Rose et al. provide an overview of the positive effects of SDM in goal setting in a rehabilitation context; increased patient satisfaction and motivation were identified as important mechanisms [47].
Considering our lessons learned, the individualization of the ERAS® mobilization goal and subsequent patient motivation could proceed as follows: In the preoperative ERAS® consultation, patients are informed that spending as much time as possible out of bed has a positive effect on the recovery process. Subsequently, patients should set their individual goal for POD1, which they personally consider realistic and also are willing to achieve. The ERAS®-nurse provides support and ensures that the goals are specific (in hours), not too low/high and documented (e.g. ERAS®-diary). Patients who struggle with the specification of goals could be informed that on average about 2.5 h (150 min) are achieved out of bed on POD1 (which is about the median of our study of 135 min). After the operation (in the evening at POD0 or in the morning at POD1), the preoperative goal set for POD1 should be discussed again (and adjusted if necessary). At POD2 early in the morning (or at POD 1 in the evening), the mobilization at POD1 should be evaluated and based on this, a new individual daily target for POD2 would be set via SDM. The new daily target should be higher than the achieved value of POD1. In this way, the principle is similar to the mechanism of fitness trackers, which adjust the target for the following day depending on the achievement of a daily target (e.g. steps) [48]. The same procedure can be used for the following postoperative days. Although a goal for surgery day would also be useful, it is difficult to assess due to the uncertain end of surgery and dependence on staff for initial mobilization. To avoid early failure, we recommend the general goal of “first time standing up” for surgery day (currently achieved by about 50% [3, 13, 14]). In addition to time out of bed, walking goals (e.g., walking down the ward x times) are also possible within the individualized goals. It should be empirically tested whether individualized and participatory ERAS® mobilization goals lead to better mobilization than the general daily goal of 360 min or no specific goal at all (as in the current ERAS® guidelines). With new research, the intention is to close the current evidence gap regarding the imprecise mobilization recommendation within the next update of the ERAS® colorectal guideline.
Conclusion
In this study, postoperative mobilization of ERAS®-patients was measured as accurately as possible using three different motion sensors. Thereby it could be demonstrated that only about 20% of patients achieved the ERAS® compliance goal of 360 min/day out of bed. Evidence from goal setting research suggests that the best performance is achieved through specific goals that are challenging yet achievable. To provide optimal goal targets for as many patients as possible, this study proposes an adjustment of the general ERAS® mobilization goals to individualized goals defined participatively between ERAS® nurse and patient. In follow-up RCTs, it is needed to test whether individualized ERAS® goal setting and motivation techniques lead to improved mobilization. Additionally, the new research findings should help to adapt and solidify the recommendations for mobilization in future updates of the ERAS® guidelines.
Data availability
No datasets were generated or analysed during the current study.
References
Ljungqvist O, de Boer HD, Balfour A, Fawcett WJ, Lobo DN, Nelson G et al (2021) Opportunities and challenges for the next phase of enhanced recovery after surgery: a review. JAMA Surg 156(8):775–784. https://doi.org/10.1001/jamasurg.2021.0586
Ljungqvist O, Scott M, Fearon KC (2017) Enhanced recovery after surgery: a review. JAMA Surg 152(3):292–298. https://doi.org/10.1001/jamasurg.2016.4952
Ripolles-Melchor J, Ramirez-Rodriguez JM, Casans-Frances R, Aldecoa C, Abad-Motos A, Logrono-Egea M et al (2019) Association between Use of enhanced recovery after surgery protocol and postoperative complications in colorectal surgery: the postoperative outcomes within enhanced recovery after surgery protocol (POWER) study. JAMA Surg 154(8):725–736. https://doi.org/10.1001/jamasurg.2019.0995
Greer NL, Gunnar WP, Dahm P, Lee AE, MacDonald R, Shaukat A et al (2018) Enhanced recovery protocols for adults undergoing colorectal surgery: a systematic review and Meta-analysis. Dis Colon Rectum 61(9):1108–1118. https://doi.org/10.1097/DCR.0000000000001160
Kehlet H (2018) ERAS implementation-time to move Forward. Ann Surg 267(6):998–999. https://doi.org/10.1097/SLA.0000000000002720
Gustafsson UO, Scott MJ, Hubner M, Nygren J, Demartines N, Francis N et al (2019) Guidelines for Perioperative Care in Elective colorectal surgery: enhanced recovery after surgery (ERAS((R))) Society recommendations: 2018. World J Surg 43(3):659–695. https://doi.org/10.1007/s00268-018-4844-y
Turan A, Khanna AK, Brooker J, Saha AK, Clark CJ, Samant A et al (2023) Association between mobilization and composite postoperative complications following major elective surgery. JAMA Surg. https://doi.org/10.1001/jamasurg.2023.1122
Nygren J, Thacker J, Carli F, Fearon KC, Norderval S, Lobo DN et al (2013) Guidelines for perioperative care in elective rectal/pelvic surgery: enhanced recovery after surgery (ERAS((R))) Society recommendations. World J Surg 37(2):285–305. https://doi.org/10.1007/s00268-012-1787-6
Wilnerzon Thörn R-M, Forsberg A, Stepniewski J, Hjelmqvist H, Magnuson A, Ahlstrand R et al (2024) Immediate mobilization in post-anesthesia care unit does not increase overall postoperative physical activity after elective colorectal surgery: a randomized, double-blinded controlled trial within an enhanced recovery protocol. World J Surg 48(4):956–966. https://doi.org/10.1002/wjs.12102
Currie A, Soop M, Demartines N, Fearon K, Kennedy R, Ljungqvist O (2019) Enhanced recovery after surgery interactive audit system: 10 years’ experience with an International web-based clinical and Research Perioperative Care Database. Clin Colon Rectal Surg 32(1):75–81. https://doi.org/10.1055/s-0038-1673357
ERAS® interactive Audit System (EIAS) - Database for colorectal surgery. https://encare.net/colorectal-protocol/ Accessed 19.04.2024
van Zelm R, Coeckelberghs E, Sermeus W, De Buck A, Weimann A, Seys D et al (2017) Variation in care for surgical patients with colorectal cancer: protocol adherence in 12 European hospitals. Int J Colorectal Dis 32(10):1471–1478. https://doi.org/10.1007/s00384-017-2863-z
Schwenk W, Lang I, Huhn M (2022) [Elective colorectal fast-track resections-treatment adherence due to coordination by specialized nursing personnel]. Chirurg 93(5):499–508. https://doi.org/10.1007/s00104-021-01484-9
Grass F, Pache B, Martin D, Addor V, Hahnloser D, Demartines N et al (2018) Feasibility of early postoperative mobilisation after colorectal surgery: a retrospective cohort study. Int J Surg 56:161–166. https://doi.org/10.1016/j.ijsu.2018.06.024
Gustafsson UO, Hausel J, Thorell A, Ljungqvist O, Soop M, Nygren J (2011) Adherence to the enhanced recovery after surgery protocol and outcomes after colorectal cancer surgery. Arch Surg 146(5):571–577. https://doi.org/10.1001/archsurg.2010.309
Swann C, Rosenbaum S, Lawrence A, Vella SA, McEwan D, Ekkekakis P (2021) Updating goal-setting theory in physical activity promotion: a critical conceptual review. Health Psychol Rev 15(1):34–50. https://doi.org/10.1080/17437199.2019.1706616
Epton T, Currie S, Armitage CJ (2017) Unique effects of setting goals on behavior change: systematic review and meta-analysis. J Consult Clin Psychol 85(12):1182–1198. https://doi.org/10.1037/ccp0000260
Bar-Eli M, Tenenbaum G, Pie JS, Btesh Y, Almog A (1997) Effect of goal difficulty, goal specificity and duration of practice time intervals on muscular endurance performance. J Sports Sci 15(2):125–135. https://doi.org/10.1080/026404197367407
Hopfner J, Keith N (2021) Goal missed, self hit: Goal-Setting, Goal-Failure, and their affective, motivational, and behavioral consequences. Front Psychol 12:704790. https://doi.org/10.3389/fpsyg.2021.704790
McEwan D, Harden SM, Zumbo BD, Sylvester BD, Kaulius M, Ruissen GR et al (2016) The effectiveness of multi-component goal setting interventions for changing physical activity behaviour: a systematic review and meta-analysis. Health Psychol Rev 10(1):67–88. https://doi.org/10.1080/17437199.2015.1104258
Tazreean R, Nelson G, Twomey R (2022) Early mobilization in enhanced recovery after surgery pathways: current evidence and recent advancements. J Comp Eff Res 11(2):121–129. https://doi.org/10.2217/cer-2021-0258
Pisarska M, Pedziwiatr M, Malczak P, Major P, Ochenduszko S, Zub-Pokrowiecka A et al (2016) Do we really need the full compliance with ERAS protocol in laparoscopic colorectal surgery? A prospective cohort study. Int J Surg 36. https://doi.org/10.1016/j.ijsu.2016.11.088. Pt A):377 – 82
Catarci M, Ruffo G, Viola MG, Pirozzi F, Delrio P, Borghi F et al (2022) ERAS program adherence-institutionalization, major morbidity and anastomotic leakage after elective colorectal surgery: the iCral2 multicenter prospective study. Surg Endosc 36(6):3965–3984. https://doi.org/10.1007/s00464-021-08717-2
Wolk S, Linke S, Bogner A, Sturm D, Meissner T, Mussle B et al (2019) Use of Activity Tracking in Major visceral surgery-the enhanced perioperative mobilization trial: a Randomized Controlled Trial. J Gastrointest Surg 23(6):1218–1226. https://doi.org/10.1007/s11605-018-3998-0
Miller WR, Rollnick S (1991) Motivational interviewing: preparing people to change addictive behavior. Motivational interviewing: preparing people to change addictive behavior. The Guilford Press, New York, NY, US
Wiesenberger R, Müller J, Kaufmann M, Weiß C, Ghezel-Ahmadi D, Hardt J et al (2024) Influence of motivational interviewing on postoperative mobilization in the enhanced recovery after surgery (ERAS®) pathway in elective colorectal surgery - a randomized patient-blinded pilot study. Langenbeck’s Archives Surg 409(1):134. https://doi.org/10.1007/s00423-024-03321-z
Seyfried S, Herrle F, Schroter M, Hardt J, Betzler A, Rahbari NN et al (2021) [Initial experiences with the implementation of the enhanced recovery after surgery (ERAS(R)) protocol]. Chirurg 92(5):428–433. https://doi.org/10.1007/s00104-020-01341-1
Garmin - Vivosmart® 4 technische Daten. https://www.garmin.com/de-DE/p/125677 Accessed 19.04.2024.
Garmin Connect-App https://www.garmin.com/de-DE/p/125677 Accessed 19.04.2024.
movisens - Move 4 Aktivitätssensor https://www.movisens.com/de/produkte/aktivitaetssensor/ Accessed 19.04.2024.
movisens - EcgMove 4 - EKG- und Aktivitätssensor. https://www.movisens.com/de/produkte/ekg-sensor/ Accessed 19.04.2024
Anastasopoulou P, Härtel S, Hey S (2013) A comparison of two commercial activity monitors for measuring step counts during different everyday life walking activities. Int J Sports Sci Eng 07:031–035
Anastasopoulou P, Tansella M, Stumpp J, Shammas L, Hey S (2012) Classification of human physical activity and energy expenditure estimation by accelerometry and barometry. Annu Int Conf IEEE Eng Med Biol Soc 2012:6451–6454. https://doi.org/10.1109/embc.2012.6347471
movisens - SensorManager https://www.movisens.com/de/produkte/sensormanager/ Accessed 19.04.2024.
movisens - DataAnalyzer – Analyse der Sensor-Daten. https://www.movisens.com/de/produkte/dataanalyzer/ Accessed 19.04.2024.
Movisens Docs - Algorithms - Physical Activity. https://docs.movisens.com/Algorithms/physical_activity/#activity-class-activityclass Accessed 19.04.2024.
Craig CL, Marshall AL, Sjostrom M, Bauman AE, Booth ML, Ainsworth BE et al (2003) International physical activity questionnaire: 12-country reliability and validity. Med Sci Sports Exerc 35(8):1381–1395. https://doi.org/10.1249/01.MSS.0000078924.61453.FB
Gehrlach C, Altenhöner T, Schwappach D (2010) Der Patients’ Experience Questionnaire: Patientenerfahrungen vergleichbar machen. Verlag Bertelsmann Stiftung
Mahoney FI, Barthel DW (1965) FUNCTIONAL EVALUATION: THE BARTHEL INDEX. Md State Med J 14:61–65
Stark PA, Myles PS, Burke JA (2013) Development and psychometric evaluation of a postoperative quality of recovery score: the QoR-15. Anesthesiology 118(6):1332–1340. https://doi.org/10.1097/ALN.0b013e318289b84b
de Leeuwerk ME, Bor P, van der Ploeg HP, de Groot V, van der Schaaf M, van der Leeden M et al (2022) The effectiveness of physical activity interventions using activity trackers during or after inpatient care: a systematic review and meta-analysis of randomized controlled trials. Int J Behav Nutr Phys Act 19(1):59. https://doi.org/10.1186/s12966-022-01261-9
Fiore JF Jr., Castelino T, Pecorelli N, Niculiseanu P, Balvardi S, Hershorn O et al (2017) Ensuring early mobilization within an enhanced recovery program for colorectal surgery: a Randomized Controlled Trial. Ann Surg 266(2):223–231. https://doi.org/10.1097/SLA.0000000000002114
Nevo Y, Shaltiel T, Constantini N, Rosin D, Gutman M, Zmora O et al (2022) Activity tracking after surgery: does it correlate with postoperative complications? Am Surg 88(2):226–232. https://doi.org/10.1177/0003134820988818
Porserud A, Aly M, Nygren-Bonnier M, Hagströmer M (2019) Objectively measured mobilisation is enhanced by a new behaviour support tool in patients undergoing abdominal cancer surgery. Eur J Surg Oncol 45(10):1847–1853. https://doi.org/10.1016/j.ejso.2019.04.013
Brusso RC, Orvis KA (2013) The impeding role of initial unrealistic goal-setting on videogame-based training performance: identifying underpinning processes and a solution. Comput Hum Behav 29(4):1686–1694. https://doi.org/10.1016/j.chb.2013.01.006
Charles C, Gafni A, Whelan T (1997) Shared decision-making in the medical encounter: what does it mean? (or it takes at least two to tango). Soc Sci Med 44(5):681–692. https://doi.org/10.1016/s0277-9536(96)00221-3
Rose A, Rosewilliam S, Soundy A (2017) Shared decision making within goal setting in rehabilitation settings: a systematic review. Patient Educ Couns 100(1):65–75. https://doi.org/10.1016/j.pec.2016.07.030
Garmin - Auto Goal feature https://support.garmin.com/sv-SE/?faq=LbmCWXit2h5eY4ytvUUeT6 Accessed 19.04.2024.
Funding
This study was not externally supported. All costs were covered by the Universitätsmedizin Mannheim.
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
Authors’ Contributions (Initials as in the order of authorship): Study conception and design: RW, JM, MK, CW, CR, FH Acquisition of data: RW, JM Analysis and interpretation of data: RW, CW, DGA, JH, FH Drafting of manuscript: RW Critical revision of manuscript: RW, JM, MK, CW, DGA, JH, CR, FH.
Corresponding author
Ethics declarations
Compliance with ethical standards
This study was registered prospectively in the German Clinical Trials Register (DRKS00027863) and approved by the II Ethics Committee of the University of Heidelberg (number: 2021-698-AF 5). All participants have signed informed consent.
Publication history
The submitted manuscript has not yet been published. Only an abstract of the paper is available online as part of the ERAS World Congress (https://erascongress.com/program/2023/P8.cgi).
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wiesenberger, R., Müller, J., Kaufmann, M. et al. Feasibility and usefulness of postoperative mobilization goals in the enhanced recovery after surgery (ERAS®) clinical pathway for elective colorectal surgery. Langenbecks Arch Surg 409, 266 (2024). https://doi.org/10.1007/s00423-024-03442-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00423-024-03442-5