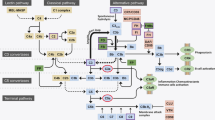
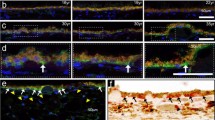
Abstract
Idiopathic epiretinal membrane (iERM) is a fibrocellular proliferation on the inner surface of the retina, which leads to decreased visual acuity and even central visual loss. As iERM is associated to advanced age and posterior vitreous detachment, a higher prevalence is expected with increasing life expectancy and aging of the global population. Although various cell types of retinal and extra-retinal origin have been described in iERMs (Müller glial cells, astrocytes, hyalocytes, retinal pigment epithelium cells, myofibroblasts, and fibroblasts), myofibroblasts have a central role in collagen production and contractile activity. Thus, myofibroblast differentiation is considered a key event for the iERM formation and progression, and fibroblasts, Müller glial cells, hyalocytes, and retinal pigment epithelium have been identified as myofibroblast precursors. On the other side, the different cell types synthesize growth factors, cytokines, and extracellular matrix, which have a crucial role in ERM pathogenesis. In the present review, the major cellular components and their functions are summarized, and their possible roles in the iERM formation are discussed. By exploring in detail the cellular and molecular aspects of iERM, we seek to contribute for better understanding of this fibrotic disease and the origin of myofibroblasts, which may eventually drive to more targeted therapeutic approaches.
Similar content being viewed by others
References
Gass JDM (1997) Macular dysfunction caused by vitreous and vitreomacular interface abnormalities. In: Gass JDM (ed) Stereoscopic atlas of macular diseases: diagnosis and treatment. Mosby, St Louis, pp 938–950
Joshi M, Agrawal S, Christoforidis JB (2013) Inflammatory mechanisms of idiopathic epiretinal membrane formation. Mediators Inflamm. https://doi.org/10.1155/2013/192582
Bringmann A, Wiedemann P (2009) Involvement of Müller glial cells in epiretinal membrane formation. Graefe’s Arch Clin Exp Ophthalmol. https://doi.org/10.1007/s00417-009-1082-x
Kanda A, Noda K, Hirose I, Ishida S (2019) TGF-β-SNAIL axis induces Müller glial-mesenchymal transition in the pathogenesis of idiopathic epiretinal membrane. Sci Rep. https://doi.org/10.1038/s41598-018-36917-9
Xiao W, Chen X, Yan W, Zhu Z, He M (2017) Prevalence and risk factors of epiretinal membranes : a systematic review and meta-analysis of population- based studies. BMJ Open. https://doi.org/10.1136/bmjopen-2016-014644
Bu SC, Kuijer R, Li XR, Hooymans JMM, Los LI (2014) Idiopathic epiretinal membrane. Retina. https://doi.org/10.1097/iae.0000000000000349
Cheung N, Tan SP, Lee SY, Cheung GCM, Tan G, Kumar N, Cheng CY, Wong TY (2017) Prevalence and risk factors for epiretinal membrane: the Singapore Epidemiology of Eye Disease study. Br J Ophthalmol. https://doi.org/10.1136/bjophthalmol-2016-308563
Meuer SM, Myers CE, Klein BEK, Swift MK, Huang Y, Gangaputra S, Pak JW, Danis RP, Klein R (2015) The epidemiology of vitreoretinal interface abnormalities as detected by spectral-domain optical coherence tomography: The Beaver Dam Eye study. Ophthalmology. https://doi.org/10.1016/j.ophtha.2014.10.014
Fung AT, Galvin J, Tran T (2021) Epiretinal membrane: a review. Clin Exp Ophthalmol. https://doi.org/10.1111/ceo.13914
Sebag J (2004) Anomalous posterior vitreous detachment : a unifying concept in vitreo-retinal disease. Graefe’s Arch Clin Exp Ophthalmol. https://doi.org/10.1007/s00417-004-0980-1
Foos RY (1974) Vitreoretinal juncture -simple epiretinal membranes. Graefe’s Arch Clin Exp Ophthalmol. https://doi.org/10.1007/bf02384852
Foos RY (1977) Vitreoretinal juncture; epiretinal membranes and vitreous. Invest Ophthalmol Vis Sci 16:416–422
Kohno RI, Hata Y, Kawahara S, Kita T, Arita R, Mochizuki Y, Aiello LP, Ishibashi T (2009) Possible contribution of hyalocytes to idiopathic epiretinal membrane formation and its contraction. Br J Ophthalmol. https://doi.org/10.1136/bjo.2008.155069
Kampik A (2012) Pathology of epiretinal membrane, idiopathic macular hole, and vitreomacular traction syndrome. Retina. https://doi.org/10.1097/iae.0b013e31825bc20a
Bu SC, Kuijer R, van der Worp RJ, Postma G, Renardel de Lavalette VW, Li XR, Hooymans JMM, Los LI (2015) Immunohistochemical evaluation of idiopathic epiretinal membranes and in vitro studies on the effect of TGF-β on Müller cells. Invest Ophthalmol Vis Sci. https://doi.org/10.1167/iovs.14-15971
Christakopoulos C, Cehofski LJ, Christensen SR, Vorum H, Honoré B (2019) Proteomics reveals a set of highly enriched proteins in epiretinal membrane compared with inner limiting membrane. Exp Eye Res. https://doi.org/10.1016/j.exer.2019.107722
Coltrini D, Belleri M, Gambicorti E, Romano D, Morescalchi F, Chandran AMK, Calza S, Semeraro F, Presta M (2020) Gene expression analysis identifies two distinct molecular clusters of idiopatic epiretinal membranes. Biochim Biophys Acta Mol Basis of Dis. https://doi.org/10.1016/j.bbadis.2020.165938
Yu J, Feng L, Wu Y, Wang H, Ba J, Zhu W, Xie C (2014) Vitreous proteomic analysis of idiopathic epiretinal membranes. Mol Biosyst. https://doi.org/10.1039/C4MB00240G
Öhman T, Tamene F, Göös H, Loukovaara S, Varjosalo M (2018) Systems pathology analysis identifies neurodegenerative nature of age-related vitreoretinal interface diseases. Aging Cell. https://doi.org/10.1111/acel.12809
Russo A, Ragusa M, Barbagallo C, Longo A, Avitabile T, Uva MG et al (2017) miRNAs in the vitreous humor of patients affected by idiopathic epiretinal membrane and macular hole. PLoS ONE. https://doi.org/10.1371/journal.pone.0174297
Usui-Ouchi A, Ouchi Y, Kiyokawa M, Sakuma T, Ito R, Ebihara N (2016) Upregulation of mir-21 levels in the vitreous humor is associated with development of proliferative vitreoretinal disease. PLoS ONE. https://doi.org/10.1371/journal.pone.0158043
Akamine PS, Lima CR, Lustoza-Costa GJ, Fuziwara CS, Del Debbio CB, Kimura ET, Santos MF, Hamassaki DE (2021) Age-related increase of let-7 family microRNA in rat retina and vitreous. Exp Eye Res. https://doi.org/10.1016/j.exer.2020.108434
Oberstein SYL, Byun J, Herrera D, Chapin EA, Fisher SK, Lewis GP (2011) Cell proliferation in human epiretinal membranes: characterization of cell types and correlation with disease condition and duration. Mol Vis 17:1794–1805
Zhao F, Gandorfer A, Haritoglou C, Scheler R, Schaumberger MM, Kampik A, Schumann RG (2013) Epiretinal cell proliferation in macular pucker and vitreomacular traction syndrome: analysis of flat-mounted internal limiting membrane specimens. Retina. https://doi.org/10.1097/iae.0b013e3182602087
Vogt D, Vielmuth F, Wertheimer C, Hagenau F, Guenther SR, Wolf A, Spindler V, Priglinger SG, Schumann RG (2018) Premacular membranes in tissue culture. Graefe’s Arch Clin Exp Ophthalmol. https://doi.org/10.1007/s00417-018-4033-6
Guenther SR, Schumann RG, Hagenau F, Wolf A, Priglinger SG, Vogt D (2019) Comparison of surgically excised premacular membranes in eyes with macular pucker and proliferative vitreoretinopathy. Curr Eye Res. https://doi.org/10.1080/02713683.2018.1542006
Guidry C, King JL, Mason JO (2009) Fibrocontractive Müller cell phenotypes in proliferative diabetic retinopathy. Invest Ophthalmol Vis Sci. https://doi.org/10.1167/iovs.08-2475
Parapuram SK, Chang B, Li L, Hartung RA, Chalam KV, Nair-Menon JU, Hunt DM, Hunt RC (2009) Differential effects of TGFβ and vitreous on the transformation of retinal pigment epithelial cells. Invest Ophthalmol Vis Sci. https://doi.org/10.1167/iovs.09-3621
Shu DY, Lovicu FJ (2017) Myofibroblast transdifferentiation: the dark force in ocular wound healing and fibrosis. Prog Retin Eye Res. https://doi.org/10.1016/j.preteyeres.2017.08.001
Kritzenberger M, Junglas B, Framme C, Helbig H, Gabel VP, Fuchshofer R, Tamm ER, Hillenkamp J (2011) Different collagen types define two types of idiopathic epiretinal membranes. Histopathology. https://doi.org/10.1111/j.1365-2559.2011.03820.x
Bu SC, Kuijer R, van der Worp RJ, Huiskamp EA, Renardel de Lavalette VW, Li XR, Hooymans JMM, Los LI (2014) Glial cells and collagens in epiretinal membranes associated with idiopathic macular holes. Retina. https://doi.org/10.1097/iae.0000000000000013
Kampik A, Kenyon KR, Michels RG, Green WR, de la Cruz ZC (1981) Epiretinal and vitreous membranes. Comparative study of 56 cases. Arch Ophthalmol. https://doi.org/10.1001/archopht.1981.03930020319025
Vinores SA, Campochiaro PA, Conway BP (1990) Ultrastructural and electron-immunocytochemical characterization of cells in epiretinal membranes. Invest Ophthalmol Vis Sci 31:14–28
Mascharak S, des Jardins-Park HE, Longaker MT (2020) Fibroblast heterogeneity in wound healing: hurdles to clinical translation. Trends Mol Med. https://doi.org/10.1016/j.molmed.2020.07.008
Pakshir P, Noskovicova N, Lodyga M, Son DO, Schuster R, Goodwin A, Karvonen H, Hinz B (2020) The myofibroblast at a glance. J Cell Sci. https://doi.org/10.1242/jcs.227900
Gabbiani G, Hirschel BJ, Ryan GB, Statkov PR, Majno G (1972) Granulation tissue as a contractile organ: a study of structure and function. J Exp Med. https://doi.org/10.1084/jem.135.4.719
Zuchero JB, Barres BA (2015) Glia in mammalian development and disease. Development. https://doi.org/10.1242/dev.129304
Vecino E, Rodriguez FD, Ruzafa N, Pereiro X, Sharma SC (2016) Glia-neuron interactions in the mammalian retina. Prog Retin Eye Res. https://doi.org/10.1016/j.preteyeres.2015.06.003
Reichenbach A, Bringmann A (2020) Glia of the human retina. Glia. https://doi.org/10.1002/glia.23727
Edwards MM, McLeod DS, Bhutto IA, Villalonga MB, Seddon JM, Lutty GA (2016) Idiopathic preretinal glia in aging and age-related macular degeneration. Exp Eye Res. https://doi.org/10.1016/j.exer.2015.07.016
Bringmann A, Pannicke T, Grosche J, Francke M, Wiedemann P, Skatchkov SN, Osborne NN, Reichenbach A (2006) Müller cells in the healthy and diseased retina. Prog Retin Eye Res. https://doi.org/10.1016/j.preteyeres.2006.05.003
Escartin C, Galea E, Lakatos A, O’Callaghan JP, Petzold GC, Serrano-Pozo A (2021) Reactive astrocyte nomenclature, definitions, and future directions. Nat Neurosci. https://doi.org/10.1038/s41593-020-00783-4
Ponsioen TL, van Luyn MJA, van der Worp RJ, Pas HH, Hooymans JMM, Los LI (2008) Human retinal Müller cells synthesize collagens of the vitreous and vitreoretinal interface in vitro. Mol Vis 14:652–660
Eastlake K, Banerjee PJ, Angbohang A, Charteris DG, Khaw PT, Limb GA (2016) Müller glia as an important source of cytokines and inflammatory factors present in the gliotic retina during proliferative vitreoretinopathy. Glia. https://doi.org/10.1002/glia.22942
Bu SC, Kuijer R, van der Worp RJ, Postma G, De Lavalette VWR, Li XR, Hooymans JMM, Los LI (2015) Immunohistochemical evaluation of idiopathic epiretinal membranes and in vitro studies on the effect of TGF-β on Müller cells. Invest Ophthalmol Vis Sci. https://doi.org/10.1167/iovs.14-15971
Kase S, Saito W, Yokoi M, Yoshida K, Furudate N, Muramatsu M, Saito A, Ohno S (2006) Expression of glutamine synthetase and cell proliferation in human idiopathic epiretinal membrane. Br J Ophthalmol. https://doi.org/10.1136/bjo.2005.078394
Minchiotti S, Stampachiacchiere B, Micera A, Lambiase A, Ripandelli G, Billi B, Bonini S (2008) Human idiopathic epiretinal membranes express NGF and NGF receptors. Retina. https://doi.org/10.1097/iae.0b013e31815ec275
Iannetti L, Accorinti M, Malagola R, Bozzoni-Pantaleoni F, da Dalt S, Nicoletti F, Gradini R, Traficante A, Campanella M, Pivetti-Pezzi P (2011) Role of the intravitreal growth factors in the pathogenesis of idiopathic epiretinal membrane. Invest Ophthalmol Vis Sci. https://doi.org/10.1167/iovs.10-7116
Wu D, Kanda A, Liu Y, Noda K, Murata M, Ishida S (2020) Involvement of Müller glial autoinduction of TGF-β in diabetic fibrovascular proliferation via glial-mesenchymal transition. Invest Ophthalmol Vis Sci. https://doi.org/10.1167/iovs.61.14.29
Zhang W, Kong Y (2020) YAP is essential for TGF-β-induced retinal fibrosis in diabetic rats via promoting the fibrogenic activity of Müller cells. J Cell Mol Med. https://doi.org/10.1111/jcmm.15739
Simón MV, Prado Spalm FH, Politi LE, Rotstein NP (2015) Sphingosine-1-phosphate is a crucial signal for migration of retina Müller glial cells. Invest Ophthalmol Vis Sci. https://doi.org/10.1167/iovs.14-16195
Vera MS, Simón MV, Prado Spalm FH, Ayala-Peña VB, German OL, Politi LE, Santiago Valtierra FX, Rotstein NP (2021) Ceramide-1-phosphate promotes the migration of retina Müller glial cells. Exp Eye Res. https://doi.org/10.1016/j.exer.2020.108359
Smiddy WE, Maguire AM, Green WR, Michels RG, De La Cruz Z, Enger C, Jaeger M, Rice TA (1989) Idiopathic epiretinal membranes: ultrastructural characteristics and clinicopathologic correlation. Ophthalmology. https://doi.org/10.1016/S0161-6420(89)32811-9
Sakamoto T, Ishibashi T (2011) Hyalocytes: essential cells of the vitreous cavity in vitreoretinal pathophysiology? Retina. https://doi.org/10.1097/iae.0b013e3181facfa9
Kita T, Sakamoto T, Ishibashi T (2014) II.D. Hyalocytes: essential vitreous cells in vitreoretinal health and disease. In: Sebag J (ed) Vitreous. Springer, New York. https://doi.org/10.1007/978-1-4939-1086-1_10
Yu C, Roubeix C, Sennlaub F, Saban DR (2020) Microglia versus monocytes: distinct roles in degenerative diseases of the retina. Trends Neurosci. https://doi.org/10.1016/j.tins.2020.03.012
Baek J, Park HY, Lee JH, Choi M, Lee JH, Ha M, Lee MY (2020) Elevated M2 macrophage markers in epiretinal membranes with ectopic inner foveal layers. Invest Ophthalmol Vis Sci. https://doi.org/10.1167/iovs.61.2.19
Prinz M, Erny D, Hagemeyer N (2017) Ontogeny and homeostasis of CNS myeloid cells. Nat Immunol. https://doi.org/10.1038/ni.3703
Eme-Scolan E, Dando SJ (2020) Tools and approaches for studying microglia in vivo. Front Immunol. https://doi.org/10.3389/fimmu.2020.583647
Boneva SK, Wolf J, Rosmus DD, Schlecht A, Prinz G, Laich Y et al (2020) Transcriptional profiling uncovers human hyalocytes as a unique innate immune cell population. Front Immunol. https://doi.org/10.3389/fimmu.2020.567274
Rathnasamy G, Fould WS, Ling EA, Kaur C (2019) Retinal microglia – a key player in healthy and diseased retina. Prog Neurobiol. https://doi.org/10.1016/j.pneurobio.2018.05.006
O’Koren EG, Yu C, Klingeborn M, Wong AYW, Prigge CL, Mathew R et al (2019) Microglial function is distinct in different anatomical locations during retinal homeostasis and degeneration. Immunity. https://doi.org/10.1016/j.immuni.2019.02.007
Reyes NJ, O’Koren EG, Saban DR (2017) New insights into mononuclear phagocyte biology from the visual system. Nat Rev Immunol. https://doi.org/10.1038/nri.2017.13
Castanos MV, Zhou DB, Linderman RE, Allison R, Milman T, Carroll J, Migacz J, Bosen RB, Chui TYP (2020) Imaging of macrophage-like cells in living human retina using clinical OCT. Invest Ophthalmol Vis Sci. https://doi.org/10.1167/iovs.61.6.48
Hammer DX, Agrawal A, Villanueva R, Saeedi O, Liu Z (2020) Label-free adaptive optics imaging of human retinal macrophage distribution and dynamics. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.2010943117
Balazs EA, Toth LZJ, Eckl EA, Mitchell AP (1964) Studies on the structure of the vitreous body. XII. Cytological and histochemical studies on the cortical tissue layer. Exp Eye Res. https://doi.org/10.1016/s0014-4835(64)80008-7
Qiao H, Hisatomi T, Sonoda KH, Kura S, Sassa Y, Kinoshita S, Nakamura T, Sakamoto T, Ishibashi T (2005) The characterisation of hyalocytes: the origin, phenotype, and turnover. Br J Ophthalmol. https://doi.org/10.1136/bjo.2004.050658
Haddad A, André JC (1998) Hyalocyte-like cells are more numerous in the posterior chamber than they are in the vitreous of the rabbit eye. Exp Eye Res. https://doi.org/10.1006/exer.1997.0476
Zhu M, Madigan MC, van Driel D, Maslim J, Billson FA, Provis JM, Penfold PL (2000) The human hyaloid system : cell death and vascular regression. Exp Eye Res. https://doi.org/10.1006/exer.2000.0844
Lazarus HS, Hageman GS (1994) In situ characterization of the human hyalocyte. Arch Ophthalmol. https://doi.org/10.1001/archopht.1994.01090220106031
Nuzzi R, Bergandi L, Zabetta LC, D’Errico L, Riscaldino F, Menegon S, Silvagno F (2020) In vitro generation of primary cultures of human hyalocytes. Mol Vis 26:818–829
Schumann RG, Gandorfer A, Ziada J, Scheler R, Schaumberger MM, Wolf A, Haritoglou C (2014) Hyalocytes in idiopathic epiretinal membranes: a correlative light and electron microscopic study. Graefe’s Arch Clin Exp Ophthalmol. https://doi.org/10.1007/s00417-014-2841-x
Sommer F, Pollinger K, Brandl F, Weiser B, Tessmar J, Blunk T, Göpferich A (2008) Hyalocyte proliferation and ECM accumulation modulated by bFGF and TGF-β1. Graefe’s Arch Clin Exp Ophthalmol. https://doi.org/10.1007/s00417-008-0846-z
Kita T, Hata Y, Kano K, Miura M, Nakao S, Noda Y, Shimokawa H, Ishibashi T (2007) Transforming growth factor-beta2 and connective tissue growth factor in proliferative vitreoretinal diseases: possible involvement of hyalocytes and therapeutic potential of Rho kinase inhibitor. Diabetes. https://doi.org/10.2337/db06-0581
Kita T, Hata Y, Arita R, Kawahara S, Miura M, Nakao S, Mochizuki Y, Enaida H, Goto Y, Shimokawa H, Hafezi-Moghadam A, Ishibashi T (2008) Role of TGF-β in proliferative vitreoretinal diseases and ROCK as a therapeutic target. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.0804054105
Lakkaraju A, Umapathy A, Tan LX, Daniele L, Philp NJ, Boesze-Battaglia K, Williams DS (2020) The cell biology of the retinal pigment epithelium. Prog Retin Eye Res. https://doi.org/10.1016/j.preteyeres.2020.100846
Feist RM, King JL, Morris R, Witherspoon CD, Guidry C (2014) Myofibroblast and extracellular matrix origins in proliferative vitreoretinopathy. Graefe’s Arch Clin Exp Ophthalmol. https://doi.org/10.1007/s00417-013-2531-0
Tamiya S, Kaplan HJ (2016) Role of epithelial-mesenchymal transition in proliferative vitreoretinopathy. Exp Eye Res. https://doi.org/10.1016/j.exer.2015.02.008
Chen Z, Shao Y, Li X (2015) The roles of signaling pathways in epithelial-to-mesenchymal transition of PVR. Mol Vis 21:706–710
Zhou M, Geathers JS, Grillo SL, Weber SR, Wang W, Zhao Y, Sundstrom JM (2020) Role of epithelial-mesenchymal transition in retinal pigment epithelium dysfunction. Front Cell Dev Biol. https://doi.org/10.3389/fcell.2020.00501
Choi K, Lee K, Ryu SW, Im M, Kook KH, Choi C (2012) Pirfenidone inhibits transforming growth factor- β1-induced fibrogenesis by blocking nuclear translocation of Smads in human retinal pigment epithelial cell line ARPE-19. Mol Vis 18:1010–1020
Saika S, Yamanaka O, Nishikawa-Ishida I, Kitano A, Flanders KC, Okada Y, Ohnishi Y, Nakajima Y, Ikeda K (2007) Effect of Smad7 gene overexpression on transforming growth factor beta-induced retinal pigment fibrosis in a proliferative vitreoretinopathy mouse model. Arch Ophthalmol. https://doi.org/10.1001/archopht.125.5.647
Saika S, Kono-Saika S, Tanaka T, Yamanaka O, Ohnishi Y, Sato M, Muragaki Y, Ooshima A, Yoo J, Flanders KC, Roberts AB (2004) Smad3 is required for dedifferentiation of retinal pigment epithelium following retinal detachment in mice. Lab Invest 84:1245–1258
Li H, Li M, Xu D, Zhao C, Liu G, Wang F (2014) Overexpression of Snail in retinal pigment epithelial triggered epithelial-mesenchymal transition. Biochem Biophys Res Commun 446(1):347–351. https://doi.org/10.1016/j.bbrc.2014.02.119
Chen X, Xiao W, Wang W, Luo L, Ye S, Liu Y (2014) The complex interplay between ERK1/2, TGFB/Smad, and Jagged/Notch signaling pathways in the regulation of epithelial-mesenchymal transition in retinal pigment epithelium cells. PLoS ONE. https://doi.org/10.1371/journal.pone.0096365
Wang FE, Zhang C, Maminishkis A, Dong L, Zhi C, Li R et al (2010) MicroRNA-204/211 alters epithelial physiology. FASEB. https://doi.org/10.1096/fj.08-125856
Funding
Supported in part by São Paulo Research Foundation- Brazil (FAPESP, Grant number 15/2150–2), National Council of Scientific and Technological Development-Brazil (CNPq, Grant number 310402/2017–4), and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior- Brazil (CAPES)- Finance code 001. RAS, VMPR, PVS, and GJLC were recipients of CAPES fellowships. The sponsors had no role in the design or conduct of this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with humans or animals performed by any of the authors.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
da Silva, R.A., Roda, V.M., Matsuda, M. et al. Cellular components of the idiopathic epiretinal membrane. Graefes Arch Clin Exp Ophthalmol 260, 1435–1444 (2022). https://doi.org/10.1007/s00417-021-05492-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-021-05492-7