Abstract
Background
In proliferative diabetic retinopathy (PDR), Müller glial cells (MGCs) acquire migratory ability and exhibit a fibroblast-like phenotype. These activated MGCs contribute to the formation of epiretinal membrane, which will stretch the retina, and cause retinal detachment and vitreous hemorrhage. Erythropoietin (Epo) is now found effective in ameliorating renal fibrosis by inhibiting epithelial-to-mesenchymal transition of tubular epithelial cells. This study is undertaken to determine whether Epo has an effect in inhibiting MGCs activation to attenuate epiretinal membrane formation in PDR.
Method
MIO-M1 cell line was used in this study. As a pilot test to determine the most efficient treatment time and concentration of Epo, levels of connective tissue growth factor (CTGF) and transforming growth factor-β (TGF-β) were measured by real-time PCR, after treatment with Epo on MGCs cultured in high glucose. MGCs were cultured in high glucose and normal glucose for 2 days, with or without TGF-β as a pro-fibrogenic cytokine. Epo was introduced at the same time. Immunofluorescence targeting α-smooth muscle actin (α-SMA), fibronectin, and glial fibrillary acidic protein (GFAP) was performed to explore the cell phenotype. Matrix metalloproteinase 9 (MMP9) mRNA level was detected by real-time PCR. Protein levels of CTGF and cytoskeletal proteins like α-SMA and fibronectin were measured by enzyme-linked immunosorbent assay (ELISA) and Western blot respectively. Wound-healing assay was applied to evaluate the migratory ability of MGCs, and actin-tracker green was used to draw the structure of F-actin in MGCs.
Results
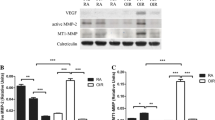
After being seeded into high-glucose medium containing TGF-β, MGCs expressed a larger amount of MMP9 mRNA as well as α-SMA, fibronectin at protein level. They secreted more CTGF, and their F-actin reorganized in a parallel manner and showed a stronger ability to migrate. In addition, these changes, including mRNA and protein expression, F-actin assembling, and cell migration, could be attenuated significantly by Epo treatment.
Conclusion
High glucose together with TGF-β promote MGCs to exhibit a fibroblast-like phenotype and develop a greater migratory ability. These changes can be inhibited by Epo, which therefore may contribute to the controlling of epiretinal membrane formation.






Similar content being viewed by others
References
Heng LZ, Comyn O, Peto T, Tadros C, Ng E, Sivaprasad S, Hykin PG (2013) Diabetic retinopathy: pathogenesis, clinical grading, management and future developments. Diabet Med 30(6):640–650. doi:10.1111/dme.12089
Campochiaro PA (2013) Ocular neovascularization. J Mol Med (Berl) 91(3):311–321. doi:10.1007/s00109-013-0993-5
Walshe R, Esser P, Wiedemann P, Heimann K (1992) Proliferative retinal diseases: myofibroblasts cause chronic vitreoretinal traction. Br J Ophthalmol 76(9):550–552
Abu El-Asrar AM, De Hertogh G, van den Eynde K, Alam K, Van Raemdonck K, Opdenakker G, Van Damme J, Geboes K, Struyf S (2015) Myofibroblasts in proliferative diabetic retinopathy can originate from infiltrating fibrocytes and through endothelial-to-mesenchymal transition (EndoMT). Exp Eye Res 132:179–189. doi:10.1016/j.exer.2015.01.023
Guidry C (2005) The role of Muller cells in fibrocontractive retinal disorders. Prog Retin Eye Res 24(1):75–86. doi:10.1016/j.preteyeres.2004.07.001
Guidry C, King JL, Mason JO 3rd (2009) Fibrocontractive Muller cell phenotypes in proliferative diabetic retinopathy. Invest Ophthalmol Vis Sci 50(4):1929–1939. doi:10.1167/iovs.08-2475
King JL, Mason JO 3rd, Cartner SC, Guidry C (2011) The influence of alloxan-induced diabetes on Muller cell contraction-promoting activities in vitreous. Invest Ophthalmol Vis Sci 52(10):7485–7491. doi:10.1167/iovs.11-7781
King JL, Guidry C (2012) Vitreous IGFBP-3 effects on Muller cell proliferation and tractional force generation. Invest Ophthalmol Vis Sci 53(1):93–99. doi:10.1167/iovs.11-8683
Friedlander M (2007) Fibrosis and diseases of the eye. J Clin Invest 117(3):576–586. doi:10.1172/jci31030
Guidry C (1997) Tractional force generation by porcine Muller cells. Development and differential stimulation by growth factors. Invest Ophthalmol Vis Sci 38(2):456–468
Guidry C, Bradley KM, King JL (2003) Tractional force generation by human Müller cells: growth factor responsiveness and integrin receptor involvement. Invest Ophthalmol Vis Sci 44(3):1355–1363
Sramek SJ, Wallow IH, Stevens TS, Nork TM (1989) Immunostaining of preretinal membranes for actin, fibronectin, and glial fibrillary acidic protein. Ophthalmology 96(6):835–841
Mizutani M, Gerhardinger C, Lorenzi M (1998) Muller cell changes in human diabetic retinopathy. Diabetes 47(3):445–449
Chu Y, Alder VA, Humphrey MF, Constable IJ (1999) Localization of IgG in the normal and dystrophic rat retina after laser lesions. Aust N Z J Ophthalmol 27(2):117–125
Kodal H, Weick M, Moll V, Biedermann B, Reichenbach A, Bringmann A (2000) Involvement of calcium-activated potassium channels in the regulation of DNA synthesis in cultured Muller glial cells. Invest Ophthalmol Vis Sci 41(13):4262–4267
Bringmann A, Pannicke T, Grosche J, Francke M, Wiedemann P, Skatchkov SN, Osborne NN, Reichenbach A (2006) Muller cells in the healthy and diseased retina. Prog Retin Eye Res 25(4):397–424. doi:10.1016/j.preteyeres.2006.05.003
Schuster SJ, Koury ST, Bohrer M, Salceda S, Caro J (1992) Cellular sites of extrarenal and renal erythropoietin production in anaemic rats. Br J Haematol 81(2):153–159
Erbayraktar S, Yilmaz O, Gokmen N, Brines M (2003) Erythropoietin is a multifunctional tissue-protective cytokine. Curr Hematol Rep 2(6):465–470
Hernandez C, Fonollosa A, Garcia-Ramirez M, Higuera M, Catalan R, Miralles A, Garcia-Arumi J, Simo R (2006) Erythropoietin is expressed in the human retina and it is highly elevated in the vitreous fluid of patients with diabetic macular edema. Diabetes Care 29(9):2028–2033. doi:10.2337/dc06-0556
Marti HH (2004) Erythropoietin and the hypoxic brain. J Exp Biol 207(Pt 18):3233–3242. doi:10.1242/jeb.01049
Garcia-Ramirez M, Hernandez C, Simo R (2008) Expression of erythropoietin and its receptor in the human retina: a comparative study of diabetic and nondiabetic subjects. Diabetes Care 31(6):1189–1194. doi:10.2337/dc07-2075
Villa P, Bigini P, Mennini T, Agnello D, Laragione T, Cagnotto A, Viviani B, Marinovich M, Cerami A, Coleman TR, Brines M, Ghezzi P (2003) Erythropoietin selectively attenuates cytokine production and inflammation in cerebral ischemia by targeting neuronal apoptosis. J Exp Med 198(6):971–975. doi:10.1084/jem.20021067
Hernandez C, Simo R (2012) Erythropoietin produced by the retina: its role in physiology and diabetic retinopathy. Endocrine 41(2):220–226. doi:10.1007/s12020-011-9579-6
Luo W, Hu L, Wang F (2015) The protective effect of erythropoietin on the retina. Ophthalmic Res 53(2):74–81. doi:10.1159/000369885
Hu LM, Luo Y, Zhang J, Lei X, Shen J, Wu Y, Qin M, Unver YB, Zhong Y, Xu GT, Li W (2011) EPO reduces reactive gliosis and stimulates neurotrophin expression in Muller cells. Front Biosci (Elite Ed) 3:1541–1555
Imamura R, Isaka Y, Sandoval RM, Ichimaru N, Abe T, Okumi M, Yazawa K, Kitamura H, Kaimori J, Nonomura N, Rakugi H, Molitoris BA, Takahara S (2012) A nonerythropoietic derivative of erythropoietin inhibits tubulointerstitial fibrosis in remnant kidney. Clin Exp Nephrol 16(6):852–862. doi:10.1007/s10157-012-0647-x
Chen CL, Chou KJ, Lee PT, Chen YS, Chang TY, Hsu CY, Huang WC, Chung HM, Fang HC (2010) Erythropoietin suppresses epithelial to mesenchymal transition and intercepts Smad signal transduction through a MEK-dependent mechanism in pig kidney (LLC-PK1) cell lines. Exp Cell Res 316(7):1109–1118. doi:10.1016/j.yexcr.2010.02.022
Boulton M, Gregor Z, McLeod D, Charteris D, Jarvis-Evans J, Moriarty P, Khaliq A, Foreman D, Allamby D, Bardsley B (1997) Intravitreal growth factors in proliferative diabetic retinopathy: correlation with neovascular activity and glycaemic management. Br J Ophthalmol 81(3):228–233
Xie Z, Chen F, Wu X, Zhuang C, Zhu J, Wang J, Ji H, Wang Y, Hua X (2012) Safety and efficacy of intravitreal injection of recombinant erythropoietin for protection of photoreceptor cells in a rat model of retinal detachment. Eye (Lond) 26(1):144–152. doi:10.1038/eye.2011.254
Zhang J, Wu Y, Jin Y, Ji F, Sinclair SH, Luo Y, Xu G, Lu L, Dai W, Yanoff M, Li W, Xu GT (2008) Intravitreal injection of erythropoietin protects both retinal vascular and neuronal cells in early diabetes. Invest Ophthalmol Vis Sci 49(2):732–742. doi:10.1167/iovs.07-0721
Lagreze WA, Feltgen N, Bach M, Jehle T (2009) Feasibility of intravitreal erythropoietin injections in humans. Br J Ophthalmol 93(12):1667–1671. doi:10.1136/bjo.2008.156794
Abu El-Asrar AM, Missotten L, Geboes K (2010) Expression of advanced glycation end products and related molecules in diabetic fibrovascular epiretinal membranes. Clin Experiment Ophthalmol 38(1):57–64. doi:10.1111/j.1442-9071.2010.02194.x, quiz 87
Abu El-Asrar AM, Van den Steen PE, Al-Amro SA, Missotten L, Opdenakker G, Geboes K (2007) Expression of angiogenic and fibrogenic factors in proliferative vitreoretinal disorders. Int Ophthalmol 27(1):11–22. doi:10.1007/s10792-007-9053-x
Twigg SM, Joly AH, Chen MM, Tsubaki J, Kim HS, Hwa V, Oh Y, Rosenfeld RG (2002) Connective tissue growth factor/IGF-binding protein-related protein-2 is a mediator in the induction of fibronectin by advanced glycosylation end-products in human dermal fibroblasts. Endocrinology 143(4):1260–1269. doi:10.1210/endo.143.4.8741
Cicha I, Goppelt-Struebe M (2009) Connective tissue growth factor: context-dependent functions and mechanisms of regulation. Biofactors 35(2):200–208. doi:10.1002/biof.30
Lipson KE, Wong C, Teng Y, Spong S (2012) CTGF is a central mediator of tissue remodeling and fibrosis and its inhibition can reverse the process of fibrosis. Fibrogenesis Tissue Repair 5(Suppl 1):S24. doi:10.1186/1755-1536-5-s1-s24
Hinton DR, Spee C, He S, Weitz S, Usinger W, LaBree L, Oliver N, Lim JI (2004) Accumulation of NH2-terminal fragment of connective tissue growth factor in the vitreous of patients with proliferative diabetic retinopathy. Diabetes Care 27(3):758–764
Winkler JL, Kedees MH, Guz Y, Teitelman G (2012) Inhibition of connective tissue growth factor by small interfering ribonucleic acid prevents increase in extracellular matrix molecules in a rodent model of diabetic retinopathy. Mol Vis 18:874–886
Klaassen I, van Geest RJ, Kuiper EJ, van Noorden CJ, Schlingemann RO (2015) The role of CTGF in diabetic retinopathy. Exp Eye Res 133:37–48. doi:10.1016/j.exer.2014.10.016
Leask A, Abraham DJ (2003) The role of connective tissue growth factor, a multifunctional matricellular protein, in fibroblast biology. Biochem Cell Biol 81(6):355–363. doi:10.1139/o03-069
Chintala H, Liu H, Parmar R, Kamalska M, Kim YJ, Lovett D, Grant MB, Chaqour B (2012) Connective tissue growth factor regulates retinal neovascularization through p53 protein-dependent transactivation of the matrix metalloproteinase (MMP)-2 gene. J Biol Chem 287(48):40570–40585. doi:10.1074/jbc.M112.386565
Grotendorst GR, Duncan MR (2005) Individual domains of connective tissue growth factor regulate fibroblast proliferation and myofibroblast differentiation. Faseb J 19(7):729–738. doi:10.1096/fj.04-3217com
Zhang X, Feng Z, Li C, Zheng Y (2012) Morphological and migratory alterations in retinal Muller cells during early stages of hypoxia and oxidative stress. Neural Regen Res 7(1):31–35. doi:10.3969/j.issn.1673-5374.2012.01.005
Brozzi F, Arcuri C, Giambanco I, Donato R (2009) S100B protein regulates astrocyte shape and migration via interaction with Src Kinase: implications for astrocyte development, activation, and tumor growth. J Biol Chem 284(13):8797–8811. doi:10.1074/jbc.M805897200
Limb GA, Daniels JT, Pleass R, Charteris DG, Luthert PJ, Khaw PT (2002) Differential expression of matrix metalloproteinases 2 and 9 by glial Muller cells: response to soluble and extracellular matrix-bound tumor necrosis factor-alpha. Am J Pathol 160(5):1847–1855
Barcelona PF, Jaldin-Fincati JR, Sanchez MC, Chiabrando GA (2013) Activated alpha2-macroglobulin induces Muller glial cell migration by regulating MT1-MMP activity through LRP1. Faseb J 27(8):3181–3197. doi:10.1096/fj.12-221598
Noda K, Ishida S, Inoue M, Obata K, Oguchi Y, Okada Y, Ikeda E (2003) Production and activation of matrix metalloproteinase-2 in proliferative diabetic retinopathy. Invest Ophthalmol Vis Sci 44(5):2163–2170
Wiercinska E, Naber HP, Pardali E, van der Pluijm G, van Dam H, ten Dijke P (2011) The TGF-beta/Smad pathway induces breast cancer cell invasion through the up-regulation of matrix metalloproteinase 2 and 9 in a spheroid invasion model system. Breast Cancer Res Treat 128(3):657–666. doi:10.1007/s10549-010-1147-x
Mo N, Li ZQ, Li J, Cao YD (2012) Curcumin inhibits TGF-beta1-induced MMP-9 and invasion through ERK and Smad signaling in breast cancer MDA- MB-231 cells. Asian Pac J Cancer Prev 13(11):5709–5714
Zhang H, Wang ZW, Wu HB, Li Z, Li LC, Hu XP, Ren ZL, Li BJ, Hu ZP (2013) Transforming growth factor-beta1 induces matrix metalloproteinase-9 expression in rat vascular smooth muscle cells via ROS-dependent ERK-NF-kappaB pathways. Mol Cell Biochem 375(1–2):11–21. doi:10.1007/s11010-012-1512-7
Zhu J, Nguyen D, Ouyang H, Zhang XH, Chen XM, Zhang K (2013) Inhibition of RhoA/Rho-kinase pathway suppresses the expression of extracellular matrix induced by CTGF or TGF-beta in ARPE-19. Int J Ophthalmol 6(1):8–14. doi:10.3980/j.issn.2222-3959.2013.01.02
Tsai HC, Su HL, Huang CY, Fong YC, Hsu CJ, Tang CH (2014) CTGF increases matrix metalloproteinases expression and subsequently promotes tumor metastasis in human osteosarcoma through down-regulating miR-519d. Oncotarget 5(11):3800–3812
Chen X, Guo Z, Wang P, Xu M (2014) Erythropoietin modulates imbalance of matrix metalloproteinase-2 and tissue inhibitor of metalloproteinase-2 in doxorubicin-induced cardiotoxicity. Heart Lung Circ 23(8):772–777. doi:10.1016/j.hlc.2014.02.015
Madro A, Kurzepa J, Czechowska G, Slomka M, Celinski K, Szymonik-Lesiuk S (2009) Erythropoietin inhibits liver gelatinases during galactosamine-induced hepatic damage in rats. Pharmacol Rep 61(5):917–923
Lei X, Zhang J, Shen J, Hu LM, Wu Y, Mou L, Xu G, Li W, Xu GT (2011) EPO attenuates inflammatory cytokines by Muller cells in diabetic retinopathy. Front Biosci (Elite Ed) 3:201–211
Park SL, Won SY, Song JH, Kambe T, Nagao M, Kim WJ, Moon SK (2015) EPO gene expression promotes proliferation, migration and invasion via the p38MAPK/AP-1/MMP-9 pathway by p21WAF1 expression in vascular smooth muscle cells. Cell Signal 27(3):470–478. doi:10.1016/j.cellsig.2014.12.001
Park SL, Won SY, Song JH, Kim WJ, Moon SK (2014) EPO gene expression induces the proliferation, migration and invasion of bladder cancer cells through the p21WAF1mediated ERK1/2/NF-kappaB/MMP-9 pathway. Oncol Rep 32(5):2207–2214. doi:10.3892/or.2014.3428
Spiering D, Hodgson L (2011) Dynamics of the Rho-family small GTPases in actin regulation and motility. Cell Adh Migr 5(2):170–180
Tsapara A, Luthert P, Greenwood J, Hill CS, Matter K, Balda MS (2010) The RhoA activator GEF-H1/Lfc is a transforming growth factor-beta target gene and effector that regulates alpha-smooth muscle actin expression and cell migration. Mol Biol Cell 21(6):860–870. doi:10.1091/mbc.E09-07-0567
Szczur K, Xu H, Atkinson S, Zheng Y, Filippi MD (2006) Rho GTPase CDC42 regulates directionality and random movement via distinct MAPK pathways in neutrophils. Blood 108(13):4205–4213. doi:10.1182/blood-2006-03-013789
Blajecka K, Marinov M, Leitner L, Uth K, Posern G, Arcaro A (2012) Phosphoinositide 3-kinase C2beta regulates RhoA and the actin cytoskeleton through an interaction with Dbl. PLoS One 7(9), e44945. doi:10.1371/journal.pone.0044945
Zhang LJ, Tao BB, Wang MJ, Jin HM, Zhu YC (2012) PI3K p110alpha isoform-dependent Rho GTPase Rac1 activation mediates H2S-promoted endothelial cell migration via actin cytoskeleton reorganization. PLoS One 7(9), e44590. doi:10.1371/journal.pone.0044590
Lin Y, Rao J, Zha XL, Xu H (2013) Angiopoietin-like 3 induces podocyte F-actin rearrangement through integrin alpha(V)beta(3)/FAK/PI3K pathway-mediated Rac1 activation. Biomed Res Int 2013:135608. doi:10.1155/2013/135608
Acknowledgments
The authors thank H Li and Ch Sun for their assistance throughout the study and JF Zhang for critical review of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The National Nature Science Foundation of China provided financial support in the form of the National Nature Science Funding (Grant No.81200693).
The sponsor had no role in the design or conduct of this research.
Conflict of Interest
All authors certify that they have NO affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge, or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Wentao Luo and Liumei Hu contributed equally to the work presented here and should therefore be regarded as equal authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
(JPG 7.93 mb)
Supplementary Fig. 2
(JPG 416 kb)
Rights and permissions
About this article
Cite this article
Luo, W., Hu, L., Li, W. et al. Epo inhibits the fibrosis and migration of Müller glial cells induced by TGF-β and high glucose. Graefes Arch Clin Exp Ophthalmol 254, 881–890 (2016). https://doi.org/10.1007/s00417-016-3290-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-016-3290-5