Abstract
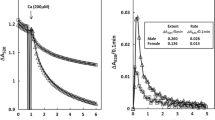
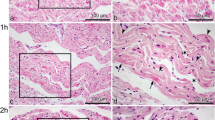
The immunohistochemical detection of dityrosine, troponins I (cTNI) and T (cTnT), and connexin 43 has been proposed as a tool for the diagnosis of myocardial infarction with short survival times. Results of clinical and experimental studies reveal that gender and/or ischemic preconditioning of the heart may have an influence on severity and magnitude of myocardial infarction. To clarify the question, if the above-mentioned markers are influenced by sex or ischemic preconditioning, experiments on isolated rat hearts using the Langendorff technique were performed. Using the hearts of 12 male and 12 female Wistar rats a local ischemia was induced through ligation of the left coronary artery. Furthermore, 12 male rat hearts underwent ischemic preconditioning of the heart by stopping the perfusion of the whole heart for 30 min and subsequently reperfusing the heart for another 60 min, before inducing local ischemia. The perfusion time after ligation varied from 10 to 60 min. A control group was comprised out of 6 male and 2 female rat hearts. These were placed in the Langendorff system for 60 min without further manipulation or received ischemic preconditioning without subsequent local ischemia or were excised without being mounted on the Langendorff system at all. All hearts were fixed in formalin and stained immunohistochemically. Depletion of the marker cTnT appeared to be less in females when compared to male hearts, for all other markers tested, no apparent difference in staining results were seen when comparing male and female rat hearts. Male rat hearts with ischemic preconditioning showed no difference compared to male rat hearts without ischemic preconditioning when stained fort dityrosine. Connexin 43 staining was less pronounced in hearts with ischemic preconditioning, whereas cTnI as well as cTnT depletion was more pronounced in preconditioned hearts. The presented findings indicate to some extent the vulnerability of the investigated markers for the influencing factors tested.



Similar content being viewed by others
References
Mayer F, Propper S, Ritz-Timme S (2014) Dityrosine, a protein product of oxidative stress, as a possible marker of acute myocardial infarctions. Int J Legal Med 128(5):787–794
Mayer F, Falk M, Huhn R, Behmenburg F, Ritz-Timme S (2016) Dityrosine as a marker of acute myocardial infarction? Experiments with the isolated Langendorff heart. Int J Legal Med 130:1053–1060
Campobasso CP, Dell’Erba AS, Addante A, Zotti F, Marzullo A, Colonna MF (2008) Sudden cardiac death and myocardial iaschemia indicators: a comparative study of four immunohistochemical markers. Am J Forensic Med Pathol 29:154–161
Sabatasso S, Mangin P, Fracasso T, Moretti M, Docquier M, Djonov V (2016) Early markers for myocardial ischemia and sudden cardiac death. Int J Legal Med 130:1265–1280
Isles CG, Hole DJ, Hawthorne VM, Lever AF (1992) Relation between coronary risk and coronary mortality in women of the Renfrew and Paisley survey- comparison with men. Lancet 339:702–706
Guerra S, Leri A, Wang X, Finato N, Di Loreto C, Beltrami CA et al (1999) Myocyte death in the failing human heart is gender dependent. Circ Res 85(9):856–866
Wang F, He Q, Sun Y, Dai X, Yang XP (2010) Female adult mouse cardiomyocytes are protected against oxidative stress. Hypertension 55(5):1172–1178
Bae S, Zhang L (2005) Gender differences in cardioprotection against ischemia/reperfusion injury in adult rat hearts: focus on Akt and protein kinase C signaling. J Pharmacol Exp Ther 315(3):1125–1135
Reiter R, Henry TD, Traverse JH (2013) Preinfarction angina reduces infarct size in ST-elevation myocardial infarction treated with percutaneous coronary intervention. Circ Cardiovasc Interv 6(1):52–58
Iwasaka T, Nakamura S, Karakawa M, Sugiura T, Inada M (1994) Cardioprotective effect of unstable angina prior to acute anterior myocardial infarction. Chest 105(1):57–61
Ovize M, Kloner RA, Hale SL, Przyklenk K (1992) Coronary cyclic flow variations "precondition" ischemic myocardium. Circulation 85(2):779–789
Liu YG, Downey JM (1992) Ischemic preconditioning protects against infarction in rat-heart. Am J Physiol 263(4):H1107–H1111
Schott RJ, Rohmann S, Braun ER, Schaper W (1990) Ischemic preconditioning reduces infarct size in swine myocardium. Circ Res 66(4):1133–1142
Murry CE, Jennings RB, Reimer KA (1986) Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation 74(5):1124–1136
Jenkins DP, Pugsley WB, Yellon DM (1995) Ischaemic preconditioning in a model of global ischaemia: infarct size limitation, but no reduction of stunning. J Mol Cell Cardiol 27(8):1623–1632
Efstathiou A, Seraskeris S, Papakonstantinou C, Aidonopoulos A, Lazou A (2001) Differential effect of preconditioning on post-ischaemic myocardial performance in the absence of substantial infarction and in extensively infarcted rat hearts. Eur J Cardiothorac Surg 19:493–499
Korthuis RJ, Gute DC, Cepinska G, Kvietsy PR (1998) Cellular mechanisms of acute versus delayed preconditioning. Pathophysiology 5:35–48
Carden DL, Granger DN (2000) Pathophysiology of ischaemia-reperfusion injurry. J Pathol 200 190:255–266
Bell RM, Mocanu MM, Yellon DM (2011) Retrograde heart perfusion: the Langendorff technique of isolated heart perfusion. J Mol Cell Cardiol 50:940–950
Li GC, Vasquez JA, Gallagher KP, Lucchesi BR (1990) Myocardial protection with preconditioning. Circulation 82:609–619
Murry CE, Richard VJ, Jennings RB, Reimer KA (1991) Myocardial protection is lost before contractile funtion recovers from ischemic preconditioning. Am J Physiol 260(29):H796–H804
Miura T, Noto T, Adachi T, Endoh A, Goto M, Urabe K, Iimura O (1991) Does myocardial stunning contribute to infarct limitation by preconditioning? Circulation 84(6):2504–2512
Giulivi C, Traaseth NJ, Davies KJ (2003) Tyrosine oxidation products: analysis and biological relevance. Amino Acids 25(3–4):227–232
Beardslee MA, Lerner DL, Tadros PN, Laing JG, Beyer EC, Yamada KA, Kléber ÁG, Schuessler RB, Saffitz JE (2000) Dephosphorylation and intracellular redistribution of ventricular connexin43 during electrical uncoupling induced by Ischaemia. Circ Res 87:656–662
Brandenburger T, Huhn R, Galas A, Pannen BH, Keitel V, Barthel F, Bauer I, Heinen A (2014) Remote ischemic preconditioning preserves connexin 43 phosphorylation in the rat heart in vivo. J Transl Med 12(28):228
Ortmann C, Pfeiffer H, Brinkmann B (2000) A comparative study on the immunohistochemical detection of early myocardial damage. Int J Legal Med 113:215–220
Osuna E, Pèrez-Cárceles MD, Alvarez MV, Noguera J, Luna A (1998) Cardiac troponin I (cTn I) and the postmortem diagnosis of myocardial infarction. Int J Legal Med 111:173–176
Kobayashi C, Miura M, Miyaishi S (2017) Postmortem exudation of myoglobin from striated muscle during formalin fixation (abstract). Rechtsmedizin 27:311–404
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The experiments in this study were conducted in accordance with the German legislation on protection of animals and the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85-23, revised 1996). The protocol for the Langendorff experiments was approved by the local Animal Ethics Committee (Project No. O 27/11).
Rights and permissions
About this article
Cite this article
Scholl, K., Huhn, R., Ritz-Timme, S. et al. The impact of sex and myocardial ischemic preconditioning on immunohistochemical markers of acute myocardial infarction. Int J Legal Med 133, 529–538 (2019). https://doi.org/10.1007/s00414-018-1948-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00414-018-1948-8