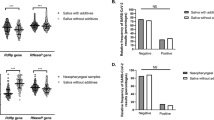
Abstract
Body fluids like blood and saliva are commonly encountered during investigations of high volume crimes like homicides. The identification of the cellular origin and the composition of the trace can link suspects or victims to a certain crime scene and provide a probative value for criminal investigations. To erase all traces from the crime scene, perpetrators often wash away their traces. Characteristically, items that show exposed stains like blood are commonly cleaned or laundered to free them from potential visible leftovers. Mostly, investigators do not delegate the DNA analysis of laundered items. However, some studies have already revealed that items can still be used for DNA analysis even after they have been laundered. Nonetheless, a systematical evaluation of laundered blood and saliva traces that provides a comparison of different established and newly developed methods for body fluid identification (BFI) is still missing. Herein, we present the results of a comprehensive study of laundered blood- and saliva-stained pieces of cloths that were applied to a broad range of methods for BFI including conventional approaches as well as molecular mRNA profiling. The study included the evaluation of cellular origin as well as DNA profiling of blood- and saliva-stained (synthetic fiber and cotton) pieces of cloths, which have been washed at various washing temperatures for one or multiple times. Our experiments demonstrate that, while STR profiling seems to be sufficiently sensitive for the individualization of laundered items, there is a lack of approaches for BFI with the same sensitivity and specificity allowing to characterize the cellular origin of challenging, particularly laundered, blood and saliva samples.








Similar content being viewed by others
References
Bond JW, Hammond C (2008) The value of DNA material recovered from crime scenes. J Forensic Sci 53:797–801. https://doi.org/10.1111/j.1556-4029.2008.00746.x
Virkler K, Lednev IK (2009) Analysis of body fluids for forensic purposes: from laboratory testing to non-destructive rapid confirmatory identification at a crime scene. Forensic Sci Int 188:1–17. https://doi.org/10.1016/j.forsciint.2009.02.013
Harbison SA, Fleming RI (2016) Forensic body fluid identification: state of the art. Res Reports Forensic Med Sci 6:11–23. https://doi.org/10.2147/RRFMS.S57994
Muro CK, Doty KC, de Souza FL, Lednev IK (2016) Forensic body fluid identification and differentiation by Raman spectroscopy. Forensic Chem 1:31–38. https://doi.org/10.1016/j.forc.2016.06.003
Li R (2015) Forensic serology. CRC Press, Boca Raton
Cox M (1991) A study of the sensitivity and specificity of four presumptive tests for blood. J Forensic Sci:1503–1511
Schweers BA, Old J, Boonlayangoor PW, Reich KA (2008) Developmental validation of a novel lateral flow strip test for rapid identification of human blood (rapid stain identification blood). Forensic Sci Int Genet 2:243–247. https://doi.org/10.1016/j.fsigen.2007.12.006
Bauer M, Patzelt D (2003) Protamine RNA as molecular marker for spermatozoa in semen stains. Int J Legal Med 117:175–179. https://doi.org/10.1007/s00414-002-0347-2
Juusola J, Ballantyne J (2005) Multiplex mRNA profiling for the identification of body fluids. Forensic Sci Int 152:1–12. https://doi.org/10.1016/j.forsciint.2005.02.020
Haas C, Klesser B, Maake C, Bär W, Kratzer A (2009) mRNA profiling for body fluid identification by reverse transcription endpoint PCR and realtime PCR. Forensic Sci Int Genet 3:80–88. https://doi.org/10.1016/j.fsigen.2008.11.003
Hanson EK, Lubenow H, Ballantyne J (2009) Identification of forensically relevant body fluids using a panel of differentially expressed microRNAs. Anal Biochem 387:303–314. https://doi.org/10.1016/j.ab.2009.01.037
Courts C, Madea B (2011) Specific microRNA signatures for the detection of saliva and blood in forensic bodyfluid identification. J Forensic Sci 56:1464–1470. https://doi.org/10.1111/j.1556-4029.2011.01894.x
Wang Z, Luo H, Pan X, Liao M, Hou Y (2012) A model for data analysis of microRNA expression in forensic body fluid identification. Forensic Sci Int Genet 6:419–423. https://doi.org/10.1016/j.fsigen.2011.08.008
Wang Z, Zhang J, Luo H, Ye Y, Yan J, Hou Y (2013) Screening and confirmation of microRNA markers for forensic body fluid identification. Forensic Sci Int Genet 7:116–123. https://doi.org/10.1016/j.fsigen.2012.07.006
Zubakov D, Boersma AWM, Choi Y, van Kuijk PF, Wiemer EA, Kayser M (2010) microRNA markers for forensic body fluid identification obtained from microarray screening and quantitative RT-PCR confirmation. Int J Legal Med 124:217–226. https://doi.org/10.1007/s00414-009-0402-3
Park JL, Park AM, Kwon OH, Lee HC, Kim JY, Seok HH, Lee WS, Lee SH, Kim YS, Woo KM, Kim SY (2014) Microarray screening and qRT-PCR evaluation of microRNA markers for forensic body fluid identification. Electrophoresis 35:362–3068. https://doi.org/10.1002/elps.201400075
Frumkin A, Wasserstrom A, Budowle B, Davidson A (2011) DNA methylation-based forensic tissue identification. Forensic Sci Int Genet 5:517–524. https://doi.org/10.1016/j.fsigen.2010.12.001
Lee HY, Jung SE, Lee EH, Yang WI, Shin KJ (2016) DNA methylation profiling for a confirmatory test for blood, saliva, semen, vaginal fluid and menstrual blood. Forensic Sci Int Genet 24:75–82. https://doi.org/10.1016/j.fsigen.2016.06.007
Poon H, Elliott J, Modler J, Frégeau C (2009) The use of Hemastix® and the subsequent lack of DNA recovery using the Promega DNA IQTM system. J Forensic Sci 54:1278–1286. https://doi.org/10.1111/j.1556-4029.2009.01173.x
Miranda GE, Prado FB, Delwing F, Darunge E Jr (2014) Analysis of the fluorescence of body fluids on different surfaces and times. Sci Justice 54:427–431. https://doi.org/10.1016/j.scijus.2014.10.002
Nelson DG, Santucci KA (2002) An alternate light source to detect semen. Acad Emerg Med 9:1045–1048
Vandenberg N, Orschoot RAH (2006) The use of polilight in the detection of seminal fluid, saliva, and bloodstains and comparison with conventional chemical-based screening tests. J Forensic Sci 51:361–370. https://doi.org/10.1111/j.1556-4029.2006.00065.x
Lindenbergh A, de Pagter M, Ramdayal G, Visser M, Zubakov D, Kayser M, Sijen T (2012) A multiplex (m)RNA-profiling system for the forensic identification of body fluids and contact traces. Forensic Sci Int Genet 6:565–577. https://doi.org/10.1016/j.fsigen.2012.01.009
Lindenbergh A, Maaskant P, Sijen T (2013) Implementation of RNA profiling in forensic casework. Forensic Sci Int Genet 7:159–166. https://doi.org/10.1016/j.fsigen.2012.09.003
van den Berge M, Bhoelai B, Harteveld J, Matai A, Sijen T (2016) Advancing forensic RNA typing: on non-target secretions, a nasal mucosa marker, a differential co-extraction protocol and the sensitivity of DNA and RNA profiling. Forensic Sci Int Genet 20:19–129. https://doi.org/10.1016/j.fsigen.2015.10.011
Andrews C, Coquoz R (1994) PCR DNA typing of washed stains. In: Bär W, Fiori A, Rossi U (eds) Advances in forensic haemogenetics 5. Springer Verlag, Heidelberg, pp 343–345. https://doi.org/10.1007/978-3-642-78782-9_90
Edler C, Gehl A, Kohwagner J, Walther M, Krebs O, Augustin C, Klein A (2017) Blood trace evidence on washed textiles—a systematic approach. Int J Legal Med 131:1179–1189. https://doi.org/10.1007/s00414-017-1549-y
Kamphausen T, Fandel SB, Gutmann JS, Bajanowski T, Poetsch M (2015) Everything clean? Transfer of DNA traces between textiles in the washtub. Int J Legal Med 129:709–714. https://doi.org/10.1007/s00414-015-1203-5
Radacher M, Dunkelmann B, Höckner G, Neuhuber F, Pölzgutter E, Breksler E, Baderer D, Steinletzberger N (2011) Luminol im Vergleich mit Fluorescein und Blue Star, Blue Star Forensic Magnum im Vergleich mit Lumiscene. Kriminalistik 3:180–184
Adair TW, Shaw RL (2005) Enhancement of bloodstains on washed clothing using luminol and LVC reagents. I.A.B.P.A, News
Horjan I, Barbaric L, Mrsic G (2016) Applicability of three commercially available kits for forensic identification of blood stains. J Forensic Leg Med 38:101–105. https://doi.org/10.1016/j.jflm.2015.11.021
Mushtaq S, Rasool N, Firiyal S (2016) Detection of dry bloodstains on different fabrics after washing with commercially available detergents. Aust J Forensic Sci 48:87–94. https://doi.org/10.1080/00450618.2015.1029971
Tobe SS, Watson N, Daeid NN (2007) Evaluation of six presumptive tests for blood, their specificity, sensitivity, and effect on high molecular-weight DNA. J Forensic Sci 52:102–109. https://doi.org/10.1111/j.1556-4029.2006.00324.x
Zubakov D, Hanekamp E, Kokshoorn M, van Ijcken W, Kayser M (2008) Stable RNA markers for identification of blood and saliva stains revealed from whole genome expression analysis of time-wise degraded samples. Int J Legal Med 122:135–142. https://doi.org/10.1007/s00414-007-0182-6
An JH, Shin KJ, Yang WI, Lee HY (2012) Body fluid identification in forensics. BMB Rep 45:545–553
Pang BCM, Cheung BKK (2008) Applicability of two commercially available kits for forensic identification of saliva stains. J Forensic Sci 53:1117–1122. https://doi.org/10.1111/j.1556-4029.2008.00814.x
Haas C, Hanson E, Kratzer A, Bär W, Ballantyne J (2011) Selection of highly specific and sensitive mRNA biomarkers for the identification of blood. Forensic Sci Int Genet 5:449–458. https://doi.org/10.1016/j.fsigen.2010.09.006
Haas C, Hanson E, Anjos MJ, Banemann R, Berti A, Borges E (2013) RNA/DNA co-analysis from human saliva and semen stains—results of a third collaborative EDNAP exercise. Forensic Sci Int Genet 7:230–239. https://doi.org/10.1016/j.fsigen.2012.10.011
Kohlmeier F, Schneider PM (2012) Successful mRNA profiling of 23 years old blood stains. Forensic Sci Int Genet 6:274–276. https://doi.org/10.1016/j.fsigen.2011.04.007
Raj PA, Edgerton M, Levine MJ (1990) Salivary histatin 5: dependence of sequence, chain length, and helical conformation for candidacidal activity. J Biol Chem 265:3898–3905
Sabatini LM, Carlock LR, Johnson GW, Azen EA (1987) cDNA cloning and chromosomal localization (4q1 1-13) of a gene for statherin, a regulator of calcium in saliva. Am J Hum Genet 41:1048–1060
Juusola J, Ballantyne J (2003) Messenger RNA profiling: a prototype method to supplant conventional methods for body fluid identification. Forensic Sci Int 135:85–96
Sakurada K, Ikegaya H, Fukushima H, Akutsu T, Watanabe K, Yoshino M (2009) Evaluation of mRNA-based approach for identification of saliva and semen. Legal Med 11:125–128. https://doi.org/10.1016/j.legalmed.2008.10.002
Setzer M, Juusola J, Ballantyne J (2008) Recovery and stability of RNA in vaginal swabs and blood, semen, and saliva stains. J Forensic Sci 53:296–305. https://doi.org/10.1111/j.1556-4029.2007.00652.x
Sirker M, Schneider PM, Gomes I (2016) A 17-month time course study of human RNA and DNA degradation in body fluids under dry and humid environmental conditions. Int J Legal Med 130:1431–1438. https://doi.org/10.1007/s00414-016-1373-9
Zubakov D, Kokshoorn M, Kloosterman A, Kayser M (2009) New markers for old stains: stable mRNA markers for blood and saliva identification from up to 16-year-old stains). Int J Legal Med 123:71–74. https://doi.org/10.1007/s00414-008-0249-z
van den Berge M, Ozcanhan G, Zijlstra SA, Lindenbergh A, Sijen T (2016) Prevalence of human cell material: DNA and RNA profiling of public and private objects and after activity scenarios. Forensic Sci Int Genet 21:81–89. doi:https://doi.org/10.1016/j.fsigen.2015.12.012
Nakanishi H, Hara M, Takahashi S, Takada A, Saito K (2014) Evaluation of forensic examination of extremely aged seminal stains. Legal Med 16:303–307. https://doi.org/10.1016/j.legalmed.2014.04.002
van Steendam K, de Ceuleneer M, Dhaenens M, van Hoofstat D, Deforce D (2013) Mass spectrometry-based proteomics as a tool to identify biological matrices in forensic sciences. Int J Legal Med 127:287–298. https://doi.org/10.1007/s00414-012-0747-x
Dammeier S, Nahnsen S, Veit J, Wehner F, Ueffing M, Kohlbach O (2016) Mass-spectrometry-based proteomics reveals organ-specific expression patterns to be used as forensic evidence. J Proteome Res 15:182–192. https://doi.org/10.1021/acs.jproteome.5b0070
Hanson E, Ingold S, Haas C, Ballantyne J (2015) Targeted multiplexed next generation RNA sequencing assay for tissue source determination of forensic samples. Forensic Sci Int Genet Supplement Series 5:e441–e443. https://doi.org/10.1016/j.fsigss.2015.09.175
Zubakov D, Kokmeijer I, Ralf A, Rajagopalan N, Calandro L, Wootton S, Langit R, Chang C, Lagace R, Kayser M (2015) Towards simultaneous individual and tissue identification: a proof-of-principle study on parallel sequencing of STRs, amelogenin, and mRNAs with the ion torrent PGM. Forensic Sci Int Genet 14:122–128. https://doi.org/10.1016/j.fsigen.2015.04.002
Lin MH, Albani PP, Fleming R (2016) Degraded RNA transcript stable regions (StaRs) as targets for enhanced forensic RNA body fluid identification. Forensic Sci Int Genet 20:61–70. https://doi.org/10.1016/j.fsigen.2015.09.012
Winter J, Diederichs S (2011) Argonaute proteins regulate microRNA stability: increased microRNA abundance by Argonaute proteins is due to microRNA stabilization. RNA Biol 8:1149–1157. https://doi.org/10.4161/rna.8.6.17665
Hutvagner G, Simard MJ (2008) Argonaute proteins: key players in RNA silencing. Nat Rev Mol Cell Biol 9:22–32. https://doi.org/10.1038/nrm2321
Goray M, Mitchell JR, van Oorschot RAH (2012) Evaluation of multiple transfer of DNA using mock case scenarios. Legal Med 14:40–46. https://doi.org/10.1016/j.legalmed.2011.09.006
Wiegand P, Heimbold C, Klein R, Immel U, Stiller D, Klintschar M (2010) Transfer of biological stains from different surfaces. Int J Legal Med 125:727–731. https://doi.org/10.1007/s00414-010-0424-x
Goray M, Eken E, Robert J, Mitchell RJ, van Oorschot RAH (2010) Secondary DNA transfer of biological substances under varying test conditions. Forensic Sci Int Genet 4:62–67. https://doi.org/10.1016/j.fsigen.2009.05.001
Phipps M, Petricevic S (2007) The tendency of individuals to transfer DNA to handled items. Forensic Sci Int 168:162–168. https://doi.org/10.1016/j.forsciint.2006.07.010
Brayley-Morris H, Sorrell A, Revoir AP, Meakin GE, Courts DS, Morgan RM (2015) Persistence of DNA from laundered semen stains: implications for child sex trafficking cases. Forensic Sci Int Genet 19:165–171. https://doi.org/10.1016/j.fsigen.2015.07.016
Acknowledgements
We kindly thank our blood and saliva donors for supplying samples. We also want to acknowledge Titia Sijen and Margreet van den Berge from the NFI for their support and helping hand with the establishment of mRNA analysis in our laboratory.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material

Supplementary Fig. 1
RNA profile of 100 μl volume sample on cotton washed at 40 °C (GIF 441 kb)
Rights and permissions
About this article
Cite this article
Kulstein, G., Wiegand, P. Comprehensive examination of conventional and innovative body fluid identification approaches and DNA profiling of laundered blood- and saliva-stained pieces of cloths. Int J Legal Med 132, 67–81 (2018). https://doi.org/10.1007/s00414-017-1691-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00414-017-1691-6