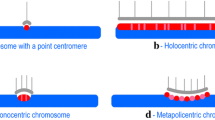
Abstract
An increasing number of observations suggest an evolutionary switch of centromere position on monocentric eukaryotic chromosomes which otherwise display a conserved sequence of genes and markers. Such observations are particularly frequent for primates and equidae (for review see Heredity 108:59–67, 2012) but occur also in marsupials (J Hered 96:217–224, 2005) and in plants (Chromosome Res 25:299–311, 2017 and references therein). The actual mechanism(s) behind remained unclear in many cases (Proc Natl Acad Sci USA 101:6542–6547, 2004; Trends Genet 30:66–74, 2014). The same is true for de novo centromere formation on chromosomes lacking an active centromere. This article focuses on recent reports on centromere repositioning and possible mechanisms behind and addresses open questions.

Similar content being viewed by others
References
Amor DJ, Choo KHA (2002) Neocentromeres: role in human disease, evolution, and centromere study. Am J Hum Genet 71:695–714. https://doi.org/10.1086/342730
Amor DJ, Bentley K, Ryan J, Perry J, Wong L, Slater H, Choo KH (2004) Human centromere repositioning “in progress”. Proc Natl Acad Sci U S A 101:6542–6547. https://doi.org/10.1073/pnas.0308637101
Bassett EA, Wood S, Saliman KJ, Ajith S, Foltz DR, Black BE (2010) Epigenetic centromere specification directs aurora B accumulation but is insufficient to efficiently correct mitotic errors. J Cell Biol 190:177–185. https://doi.org/10.1083/jcb.201001035
Bobkov GM, Gilbert N, Heun P (2018) Centormere transcription allows CENP-A to transit from chromatin association to stable incorporation. J Cell Biol. https://doi.org/10.1083/icb.201611087, jcb.201611087
Cardone MF, Lomiento M, Teti MG, Misceo D, Roberto R, Capozzi O, D’Addabbo P, Ventura M, Rocchi M, Archidiacono N (2007) Evolutionary history of chromosome 11 featuring four distinct centromere repositioning events in Catarrhini. Genomics 90:35–43. https://doi.org/10.1016/j.ygeno.2007.01.007
Chiatante G, Capozzi O, Svartman M, Perelman P, Centrone L, Romanenko SS, Ishida T, Valeri M, Roelke-Parker ME, Stanyon R (2017) Centromere repositioning explains fundamental number variability in the New World monkey genus Saimiri. Chromosoma 126:519–529. https://doi.org/10.1007/s00412-016-0619-0
Choo KH (1997) Centromere DNA dynamics: latent centromeres and neocentromere formation. Am J Hum Genet 61:1225–1233. https://doi.org/10.1086/301657
Das A, Smoak EM, Linares-Saldana R, Lampson MA, Black BE (2017) Centromere inheritance through the germline. Chromosoma 126:595–604. https://doi.org/10.1007/s00412-017-0640-y
Dawe RK, Hiatt EN (2004) Plant neocentromeres: fast, focused, and driven. Chromosom Res 12:655–669. https://doi.org/10.1023/B:CHRO.0000036607.74671.db
Dawe RK, Lowry EG, Gent JI, Stitzer MC, Swentowky KW, Higgins DM, Ross-Ibarra J, Wallace JG, Kanizay LB, Alabady M, Qiu W, Tseng K-F, Wang N, Gao Z, Birchler JA, Harkess AE, Hodges AL, Hiatt EN (2018) A kinesin-14 motor activates neocentroemeres to promote meiotic drive in maize. Cell 173:1–12. https://doi.org/10.1016/j.cell.2018.03.009
Dunleavy EM, Collins CM (2017) Centromere dynamics in male and female germ cells. Prog Mol Subcell Biol 56:357–375. https://doi.org/10.1007/978-3-319-58592-5_15
Dutrillaux B, Couturier J, Viegas-Péquignot E (1981) Chromosomal evolution in primates. Chromosom Today 7:176–191
Federico C, Pappalardo AM, Ferrito V, Tosi S, Saccone S (2017) Genomic properties of chromosomal bands are linked to evolutionary rearrangements and new centromere formation in primates. Chromosom Res 25:261–276. https://doi.org/10.1007/s10577-017-9560-1
Ferreri GC, Liscinsky DM, Mack JA, Eldridge MD, O’Neill RJ (2005) Retention of latent centromeres in the mammalian genome. J Hered 96:217–224. https://doi.org/10.1093/jhered/esi029
Fu S, Lv Z, Gao Z, Wu H, Pang J, Zhang B, Dong Q, Guo X, Wang XJ, Birchler JA, Han F (2013) De novo centromere formation on a chromosome fragment in maize. Proc Natl Acad Sci U S A 110:6033–6036. https://doi.org/10.1073/pnas.1303944110
Gong Z, Wu Y, Koblizkova A, Torres GA, Wang K, Iovene M, Neumann P, Zhang W, Novak P, Buell CR, Macas J, Jiang J (2012) Repeatless and repeat-based centromeres in potato: implications for centromere evolution. Plant Cell 24:3559–3574. https://doi.org/10.1105/tpc.112.100511
Han F, Gao Z, Birchler JA (2009a) Reactivation of an inactive centromere reveals epigenetic and structural components for centromere specification in maize. Plant Cell 21:1929–1939. https://doi.org/10.1105/tpc.109.066662
Han Y, Zhang Z, Liu C, Liu J, Huang S, Jiang J, Jin W (2009b) Centromere repositioning in cucurbit species: implication of the genomic impact from centromere activation and inactivation. Proc Natl Acad Sci U S A 106:14937–14941. https://doi.org/10.1073/pnas.0904833106
Hasson D, Alonso A, Cheung F, Tepperberg JH, Papenhausen PR, Engelen JJ, Warburton PE (2011) Formation of novel CENP-A domains on tandem repetitive DNA and across chromosome breakpoints on human chromosome 8q21 neocentromeres. Chromosoma 120:621–632. https://doi.org/10.1007/s00412-011-0337-6
Huang S, Li R, Zhang Z, Li L, Gu X, Fan W, Lucas WJ, Wang X, Xie B, Ni P, Ren Y, Zhu H, Li J, Lin K, Jin W, Fei Z, Li G, Staub J, Kilian A, van der Vossen EAG, Wu Y, Guo J, He J, Jia Z, Ren Y, Tian G, Lu Y, Ruan J, Qian W, Wang M, Huang Q, Li B, Xuan Z, Cao J, Asan, Wu Z, Zhang J, Cai Q, Bai Y, Zhao B, Han Y, Li Y, Li X, Wang S, Shi Q, Liu S, Cho WK, Kim JY, Xu Y, Heller-Uszynska K, Miao H, Cheng Z, Zhang S, Wu J, Yang Y, Kang H, Li M, Liang H, Ren X, Shi Z, Wen M, Jian M, Yang H, Zhang G, Yang Z, Chen R, Liu S, Li J, Ma L, Liu H, Zhou Y, Zhao J, Fang X, Li G, Fang L, Li Y, Liu D, Zheng H, Zhang Y, Qin N, Li Z, Yang G, Yang S, Bolund L, Kristiansen K, Zheng H, Li S, Zhang X, Yang H, Wang J, Sun R, Zhang B, Jiang S, Wang J, du Y, Li S (2009) The genome of the cucumber, Cucumis sativus L. Nat Genet 41:1275–1281. https://doi.org/10.1038/ng.475
Ishii K, Ogiyama Y, Chikashige Y, Soejima S, Masuda F, Kakuma T, Hiraoka Y, Takahashi K (2008) Heterochromatin integrity affects chromosome reorganization after centromere dysfunction. Science 321:1088–1091. https://doi.org/10.1126/science.1158699
Ketel C, Wang HS, McClellan M, Bouchonville K, Selmecki A, Lahav T, Gerami-Nejad M, Berman J (2009) Neocentromeres form efficiently at multiple possible loci in Candida albicans. PLoS Genet 5:e1000400. https://doi.org/10.1371/journal.pgen.1000400
Liu Y, Su H, Pang J, Gao Z, Wang XJ, Birchler JA, Han F (2015) Sequential de novo centromere formation and inactivation on a chromosomal fragment in maize. Proc Natl Acad Sci U S A 112:E1263–E1271. https://doi.org/10.1073/pnas.1418248112
Maggert KA, Karpen GH (2001) The activation of a neocentromere in Drosophila requires proximity to an endogenous centromere. Genetics 158:1615–1628
Marshall OJ, Chueh AC, Wong LH, Choo KHA (2008) Neocentromeres: new insights into centromere structure, disease development, and karyotype evolution. Am J Hum Genet 82:261–282. https://doi.org/10.1016/j.ajhg.2007.11.009
Montefalcone G, Tempesta S, Rocchi M, Archidiacono N (1999) Centromere repositioning. Genome Res 9:1184–1188. https://doi.org/10.1101/gr.9.12.1184
Nasuda S, Hudakova S, Schubert I, Houben A, Endo TR (2005) Stable barley chromosomes without centromeric repeats. Proc Natl Acad Sci U S A 102:9842–9847. https://doi.org/10.1073/pnas.0504235102
Rhoades MM, Vilkomerson H (1942) On the anaphase movement of chromosomes. Proc Natl Acad Sci U S A 28:433–436
Rocchi M, Archidiacono N, Schempp W, Capozzi O, Stanyon R (2012) Centromere repositioning in mammals. Heredity 108:59–67. https://doi.org/10.1038/hdy.2011.101
Schneider KL, Xie Z, Wolfgruber TK, Presting GG (2016) Inbreeding drives maize centromere evolution. Proc Natl Acad Sci U S A 113:E987–E996. https://doi.org/10.1073/pnas.1522008113
Schubert I, Lysak MA (2011) Interpretation of karyotype evolution should consider chromosome structural constraints. Trends Genet 27:207–216. https://doi.org/10.1016/j.tig.2011.03.004
Schubert I, Rieger R (1977) On the expressivity of aberration hot spots after treatment with mutagens showing delayed or non-delayed effects. Mutat Res 44:337–344. https://doi.org/10.1016/0027-5107(77)90093-8
Schubert V, Lermontova I, Schubert I (2014) Loading of the centromeric histone H3 variant during meiosis—how does it differ from mitosis? Chromosoma 123:491–497. https://doi.org/10.1007/s00412-014-0466-9
Scott KC, Sullivan BA (2014) Neocentromeres: a place for everything and everything in its place. Trends Genet 30:66–74. https://doi.org/10.1016/j.tig.2013.11.003
Shang WH, Hori T, Martins NM, Toyoda A, Misu S, Monma N, Hiratani I, Maeshima K, Ikeo K, Fujiyama A, Kimura H, Earnshaw WC, Fukagawa T (2013) Chromosome engineering allows the efficient isolation of vertebrate neocentromeres. Dev Cell 24:635–648. https://doi.org/10.1016/j.devcel.2013.02.009
Sullivan LL, Maloney KA, Towers AJ, Gregory SG, Sullivan BA (2016) Human centromere repositioning within euchromatin after partial chromosome deletion. Chromosom Res 24:451–466. https://doi.org/10.1007/s10577-016-9536-6
Teo CH, Lermontova I, Houben A, Mette FM, Schubert I (2013) De novo generation of plant centromeres at tandem repeats. Chromosoma 122:233–241. https://doi.org/10.1007/s00412-013-0406-0
Thakur J, Sanyal K (2013) Efficient neocentromere formation is suppressed by gene conversion to maintain centromere function at native physical chromosomal loci in Candida albicans. Genome Res 23:638–652. https://doi.org/10.1101/gr.141614.112
Tolomeo D, Capozzi O, Stanyon RR, Archidiacono N, D’Addabbo P, Catacchio CR, Purgato S, Perini G, Schempp W, Huddleston J, Malig M, Eichler EE, Rocchi M (2017) Epigenetic origin of evolutionary novel centromeres. Sci Rep 7:41980. https://doi.org/10.1038/srep41980
Ventura M, Mudge JM, Palumbo V, Burn S, Blennow E, Pierluigi M, Giorda R, Zuffardi O, Archidiacono N, Jackson MS, Rocchi M (2003) Neocentromeres in 15q24-26 map to duplicons which flanked an ancestral centromere in 15q25. Genome Res 13:2059–2068. https://doi.org/10.1101/gr.1155103
Vu GT, Cao HX, Watanabe K, Hensel G, Blattner FR, Kumlehn J, Schubert I (2014) Repair of site-specific DNA double-strand breaks in barley occurs via diverse pathways primarily involving the sister chromatid. Plant Cell 26:2156–2167. https://doi.org/10.1105/tpc.114.126607
Vu GTH, Cao HX, Reiss B, Schubert I (2017) Deletion-bias in DNA double-strand break repair differentially contributes to plant genome shrinkage. New Phytol 214:1712–1721. https://doi.org/10.1111/nph.14490
Wang K, Wu Y, Zhang W, Dawe RK, Jiang J (2014) Maize centrpmeres expand and adopt a uniform size in the geneticbackground of oat. Genome Res 24:107–116. https://doi.org/10.1101/gr.160887.113
Zeitlin SG, Baker NM, Chapados BR, Soutoglou E, Wang JY, Berns MW, Cleveland DW (2009) Double-strand DNA breaks recruit the centromeric histone CENP-A. Proc Natl Acad Sci U S A 106:15762–15767. https://doi.org/10.1073/pnas.0908233106
Zhang H, Koblizkova A, Wang K, Gong Z, Oliveira L, Torres GA, Wu Y, Zhang W, Novak P, Buell CR, Macas J, Jiang J (2014) Boom-bust turnovers of megabase-sized centromeric DNA in Solanum species: rapid evolution of DNA sequences associated with centromeres. Plant Cell 26:1436–1447. https://doi.org/10.1105/tpc.114.123877
Zhao H, Zeng Z, Koo DH, Gill BS, Birchler JA, Jiang J (2017) Recurrent establishment of de novo centromeres in the pericentromeric region of maize chromosome 3. Chromosom Res 25:299–311. https://doi.org/10.1007/s10577-017-9564-x
Acknowledgements
I thank Jörg Fuchs, Andreas Houben, Inna Lermontova, IPK Gatersleben, and Jiri Macas, C. Budejovice, for the critical reading, Karin Lipfert for the artwork, and Catrin Kaydamov for the help with the references.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schubert, I. What is behind “centromere repositioning”?. Chromosoma 127, 229–234 (2018). https://doi.org/10.1007/s00412-018-0672-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00412-018-0672-y