Abstract
Purpose
Sperm quality plays a vital role in successful fertilization and pregnancy. Patients with fertilization failure (total failure or low-fertilization rate) despite having normal semen parameters are a challenging group whose sperm cannot fertilize the oocyte via the intracytoplasmic sperm injection (ICSI) technique. Microfluidics is offered as a new method for proper sperm sorting.
Methods
This study aimed to evaluate sperm parameters, DNA fragmentation index (DFI), expression of phospholipase C zeta 1 (PLCZ1), and transition nuclear proteins 1 (TNP1) mRNAs in sperm selected by microfluidic sperm sorting (MSS) chip compared with conventional density gradient centrifugation technique in patients with fertilization failure following ICSI. Subsequence fertilization rate and embryo quality were assayed.
Results
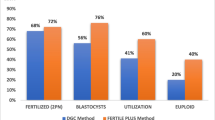
Normal morphology and total motility were significantly higher, and DFI was significantly lower in sperm selected by the MSS chip in fertilization failure and control groups. The RT-PCR results demonstrated a significant increase in the expression of PLCZ1 and TNP1 genes in sperm of both groups selected by MSS chips compared to the DGC method. In addition, with the selected sperm by MSS chip, an increase in fertilization rate and improvement of embryo quality was obtained.
Conclusion
The present study findings show that sperm sorting by the microfluidic method improves fertilization rate in patients with poor fertilization outcomes following ICSI.





Similar content being viewed by others
References
Yildiz K, Yuksel S (2019) Use of microfluidic sperm extraction chips as an alternative method in patients with recurrent in vitro fertilisation failure. J Assist Reprod Genet 36(7):1423–1429
Azizi E, Naji M, Nazarian H, Salehpour S, Borumandnia N, Shams MZ (2020) Does timing in ICSI cycle affect oocyte quality and reproductive outcomes? A prospective study. Arch Gynecol Obstet 302(2):505–513
Torra-Massana M, Cornet-Bartolomé D, Barragán M, Durban M, Ferrer-Vaquer A, Zambelli F et al (2019) Novel phospholipase C zeta 1 mutations associated with fertilization failures after ICSI. Hum Reprod 34(8):1494–1504
Tang L, Rao M, Yang W, Yao Y, Luo Q, Lu L et al (2021) Predictive value of the sperm DNA fragmentation index for low or failed IVF fertilization in men with mild-to-moderate asthenozoospermia. J Gynecol Obstet Hum Reprod 50(6):101868
Kahyaoglu I, Demir B, Turkkanı A, Cınar O, Dilbaz S, Dilbaz B et al (2014) Total fertilization failure: is it the end of the story? J Assist Reprod Genet 31(9):1155–1160
Yan Z, Fan Y, Wang F, Yan Z, Li M, Ouyang J et al (2020) Novel mutations in PLCZ1 cause male infertility due to fertilization failure or poor fertilization. Hum Reprod 35(2):472–481
Aghajanpour S, Ghaedi K, Salamian A, Deemeh M, Tavalaee M, Moshtaghian J et al (2011) Quantitative expression of phospholipase C zeta, as an index to assess fertilization potential of a semen sample. Hum Reprod 26(11):2950–2956
Haghighat S, Tavalaee M, Zakeri Z, Noureddini M, Shahverdi A, Esfahani MHN (2019) Reduction of truncated Kit Expression in men with abnormal semen parameters, globozoospermia and history of low or fertilization failure. Cell J (Yakhteh) 21(3):314
Amdani SN, Jones C, Coward K (2013) Phospholipase C zeta (PLCζ): oocyte activation and clinical links to male factor infertility. Adv Biol Regul 53(3):292–308
Yoon S-Y, Jellerette T, Salicioni AM, Lee HC, Yoo M-S, Coward K et al (2008) Human sperm devoid of PLC, zeta 1 fail to induce Ca 2+ release and are unable to initiate the first step of embryo development. J Clin Investig 118(11):3671–3681
Dai J, Dai C, Guo J, Zheng W, Zhang T, Li Y et al (2020) Novel homozygous variations in PLCZ1 lead to poor or failed fertilization characterized by abnormal localization patterns of PLCζ in sperm. Clin Genet 97(2):347–351
Kashir J, Konstantinidis M, Jones C, Lemmon B, Chang Lee H, Hamer R et al (2012) A maternally inherited autosomal point mutation in human phospholipase C zeta (PLCζ) leads to male infertility. Hum Reprod 27(1):222–231
Escoffier J, Lee HC, Yassine S, Zouari R, Martinez G, Karaouzène T et al (2016) Homozygous mutation of PLCZ1 leads to defective human oocyte activation and infertility that is not rescued by the WW-binding protein PAWP. Hum Mol Genet 25(5):878–891
Khattri A, Bhushan S, Sireesha V, Gupta N, Chakravarty B, Deendayal M et al (2011) The TNP1 haplotype–GCG is associated with azoospermia. Int J Androl 34(2):173–182
Della-Maria J, Gerard A, Franck P, Gerard H (2002) Effects of androgen-binding protein (ABP) on spermatid Tnp1 gene expression in vitro. Mol Cell Endocrinol 198(1–2):131–141
Ozcan P, Takmaz T, Yazici MGK, Alagoz OA, Yesiladali M, Sevket O et al (2021) Does the use of microfluidic sperm sorting for the sperm selection improve in vitro fertilization success rates in male factor infertility? J Obstet Gynaecol Res 47(1):382–388
Yetkinel S, Kilicdag EB, Aytac PC, Haydardedeoglu B, Simsek E, Cok T (2019) Effects of the microfluidic chip technique in sperm selection for intracytoplasmic sperm injection for unexplained infertility: a prospective, randomized controlled trial. J Assist Reprod Genet 36(3):403–409
Kishi K, Ogata H, Ogata S, Mizusawa Y, Okamoto E, Matsumoto Y et al (2015) Frequency of sperm DNA fragmentation according to selection method: comparison and relevance of a microfluidic device and a swim-up procedure. J Clin Diagn Res 9(11):14–16
Aitken RJ, Clarkson JS (1988) Significance of reactive oxygen species and antioxidants in defining the efficacy of sperm preparation techniques. J Androl 9(6):367–376
Dai X, Wang Y, Cao F, Yu C, Gao T, Xia X et al (2020) Sperm enrichment from poor semen samples by double density gradient centrifugation in combination with swim-up for IVF cycles. Sci Rep 10(1):1–8
Zini AFA, Phang D, Jarvi K (2000) Influence of semen processing technique on human sperm DNA integrity. Urology 56(6):1081–1084
Parrella A, Keating D, Cheung S, Xie P, Stewart JD, Rosenwaks Z et al (2019) A treatment approach for couples with disrupted sperm DNA integrity and recurrent ART failure. J Assist Reprod Genet 36(10):2057–2066
Quinn MM, Jalalian L, Ribeiro S, Ona K, Demirci U, Cedars MI et al (2018) Microfluidic sorting selects sperm for clinical use with reduced DNA damage compared to density gradient centrifugation with swim-up in split semen samples. Hum Reprod 33(8):1388–1393
Kashir J (2020) Increasing associations between defects in phospholipase C zeta and conditions of male infertility: not just ICSI failure? J Assist Reprod Genet 37(6):1273–1293
Jedrzejczak P, Kempisty B, Bryja A, Mostowska M, Depa-Martynow M, Pawelczyk L et al (2007) Quantitative assessment of transition proteins 1, 2 spermatid-specific linker histone H1-like protein transcripts in spermatozoa from normozoospermic and asthenozoospermic men. Arch Androl 53(4):199–205
WHO (2010) World Health Organization laboratory manual for the examination and processing of human semen
Van Royen E, Mangelschots K, De Neubourg D, Valkenburg M, Van de Meerssche M, Ryckaert G et al (1999) Characterization of a top quality embryo, a step towards single-embryo transfer. Hum Reprod 14(9):2345–2349
Kabukçu C, Çil N, Çabuş Ü, Alataş E (2021) Effect of ejaculatory abstinence period on sperm DNA fragmentation and pregnancy outcome of intrauterine insemination cycles: a prospective randomized study. Arch Gynecol Obstet 303(1):269–278
Rex A, Aagaard J, Fedder J (2017) DNA fragmentation in spermatozoa: a historical review. Andrology 5(4):622–630
Zini A, Fischer MA, Sharir S, Shayegan B, Phang D, Jarvi K (2002) Prevalence of abnormal sperm DNA denaturation in fertile and infertile men. Urology 60(6):1069–1072
Zhu WJ (2018) Preparation and observation methods can produce misleading artefacts in human sperm ultrastructural morphology. Andrologia 50(7):e13043
Marzano G, Chiriacò MS, Primiceri E, Dell’Aquila ME, Ramalho-Santos J, Zara V et al (2020) Sperm selection in assisted reproduction: a review of established methods and cutting-edge possibilities. Biotechnol Adv 40:107498
Yalcinkaya Kalyan E, Can Celik S, Okan O, Akdeniz G, Karabulut S, Caliskan E (2019) Does a microfluidic chip for sperm sorting have a positive add-on effect on laboratory and clinical outcomes of intracytoplasmic sperm injection cycles? A sibling oocyte study. Andrologia 51(10):e13403
Gode F, Gürbüz AS, Tamer B, Pala I, Isik AZ (2020) The effects of microfluidic sperm sorting, density gradient and swim-up methods on semen oxidation reduction potential. Urol J 17(4):397–401
Ješeta M, Franzová K, Žáková J, Ventruba P, Crha I (2020) Comparison of microfluidic and swim-up sperm separation methods for IVF. Med J Cell Biol 8(4):170–175
van der Westerlaken L, Helmerhorst F, Dieben S, Naaktgeboren N (2005) Intracytoplasmic sperm injection as a treatment for unexplained total fertilization failure or low fertilization after conventional in vitro fertilization. Fertil Steril 83(3):612–617
Xue L-T, Wang R-X, He B, Mo W-Y, Huang L, Wang S-K et al (2016) Effect of sperm DNA fragmentation on clinical outcomes for Chinese couples undergoing in vitro fertilization or intracytoplasmic sperm injection. J Int Med Res 44(6):1283–1291
Lee HC, Arny M, Grow D, Dumesic D, Fissore RA, Jellerette-Nolan T (2014) Protein phospholipase C Zeta1 expression in patients with failed ICSI but with normal sperm parameters. J Assist Reprod Genet 31(6):749–756
Meerschaut FV, Nikiforaki D, Heindryckx B, De Sutter P (2014) Assisted oocyte activation following ICSI fertilization failure. Reprod Biomed Online 28(5):560–571
Yelumalai S, Yeste M, Jones C, Amdani S, Kashir J, Mounce G et al (2015) Total levels and proportions of sperm exhibiting phospholipase C Zeta (PLCζ) are significantly correlated with fertilization rates following intracytoplasmic sperm injection. Fert Steril 104:561–568
Mu J, Zhang Z, Wu L, Fu J, Chen B, Yan Z et al (2020) The identification of novel mutations in PLCZ1 responsible for human fertilization failure and a therapeutic intervention by artificial oocyte activation. Mol Hum Reprod 26(2):80–87
Meng X, Melo P, Jones C, Ross C, Mounce G, Turner K et al (2020) Use of phospholipase C zeta analysis to identify candidates for artificial oocyte activation: a case series of clinical pregnancies and a proposed algorithm for patient management. Fertil Steril 114(1):163–174
Tavalaee M, Kiani-Esfahani A, Nasr-Esfahani MH (2017) Relationship between phospholipase C-zeta, semen parameters, and chromatin status. Syst Biol Reprod Med 63(4):259–268
Miyagawa Y, Nishimura H, Tsujimura A, Matsuoka Y, Matsumiya K, Okuyama A et al (2005) Single-nucleotide polymorphisms and mutation analyses of the TNP1 and TNP2 genes of fertile and infertile human male populations. J Androl 26(6):779–786
Funding
This work was supported by the Iran University of Medical Sciences under Grant (number 13580).
Author information
Authors and Affiliations
Contributions
FA contributed to the design of the study, acquisition, revision and gave input at all stages of the study. JSM, ZZ, MM, RA, MST and ASM took part in collection and analysis of data. NS contributed to drafting the article and critical discussion. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
The present study was approved by the Ethics Committee of Iran University of Medical Sciences (IR.IUMS.FMD.REC.1398.179).
Consent to participate
All the participants provided written informed consent before sample collection.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mirsanei, J.S., Sheibak, N., Zandieh, Z. et al. Microfluidic chips as a method for sperm selection improve fertilization rate in couples with fertilization failure. Arch Gynecol Obstet 306, 901–910 (2022). https://doi.org/10.1007/s00404-022-06618-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-022-06618-w