Abstract
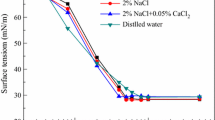
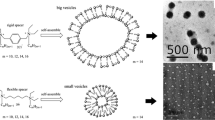
A series of anionic gemini surfactants have been synthesized. The surface properties and micellization process of as-prepared sulfonate gemini surfactants (SGS) and carboxylate gemini surfactant (CGS) have been studied by surface tension measurement and isothermal titration microcalorimetry. Meanwhile, the interaction of these five surfactants with polyacrylamide (PAM) was investigated using surface tension, steady-state fluorescence measurement, and isothermal titration microcalorimetry. The results show that the critical micelle concentrations (CMCs) of above-mentioned surfactants are more than 1 order of magnitude lower than those of corresponding single chain surfactants. Moreover, the enthalpy of micelle formation (ΔH mic) for the investigated gemini surfactants is negative. In the surfactant–PAM systems, the thermodynamic parameters of binding have also been determined. The conclusion may be drawn that the binding strength of SGS onto PAM is stronger than that of CGS, resulting from more compact structure of SGS aggregates. With increasing surfactant hydrophobicity, the values of ΔH agg become more exothermic and a ΔS agg decrease was observed. Therefore, the interaction between SGS and PAM is enthalpy-driven.







Similar content being viewed by others
Abbreviations
- SGS:
-
Sulfonate gemini surfactant
- CGS:
-
Carboxylate gemini surfactant
- CMC:
-
Critical micelle concentration
- CAC:
-
Critical aggregation concentration
- PAM:
-
Polyacrylamide
- ITC:
-
Isothermal titration microcalorimetry
References
Cheng L, Jiang Z, Dong J, Cai B, Yang Y, Li X, Chen C (2013) Monolayers of novel gemini amphiphiles with phthalimide headgroups at the air/water interface: pH and alkyl chain length effects. J Colloid Interface Sci 401:97–106
Cai B, Dong J, Cheng L, Jiang Z, Yang Y, Li X (2013) Adsorption and micellization of gemini surfactants with pyrrolidinium head groups: effect of the spacer length. Soft Matter 9(31):7637–7646
Yoshimura T, Esumi K (2004) Synthesis and surface properties of anionic gemini surfactants with amide groups. J Colloid Interface Sci 276(1):231–238
He Y, Xu S, Sun D, Shang Y, Zhao X, Liu H (2013) Decondensation of cationic gemini surfactant-induced DNA aggregates using triblock copolymer (PEO)20–(PPO)70–(PEO)20. Colloid Polym Sci 291(9):2139–2146
Zhao J, Zou W (2013) Foams stabilized by mixed cationic gemini/anionic conventional surfactants. Colloid Polym Sci 291(6):1471–1478
Iglauer S, Wu Y, Shuler P, Tang Y, Goddard WA (2010) New surfactant classes for enhanced oil recovery and their tertiary oil recovery potential. J Pet Sci Eng 71(1):23–29
Sekhon BS (2013) Surfactants: pharmaceutical and medicinal aspects. Management 1:11–36
Bell PC, Bergsma M, Dolbnya IP, Bras W, Stuart MC, Rowan AE, Feiters MC, Engberts JB (2003) Transfection mediated by gemini surfactants: engineered escape from the endosomal compartment. J Am Chem Soc 125(6):1551–1558
Pettersson E, Topgaard D, Stilbs P, Söderman O (2004) Surfactant/nonionic polymer interaction. a NMR diffusometry and NMR electrophoretic investigation. Langmuir 20(4):1138–1143
Mészáros R, Thompson L, Bos M, Varga I, Gilányi T (2003) Interaction of sodium dodecyl sulfate with polyethyleneimine: surfactant-induced polymer solution colloid dispersion transition. Langmuir 19(3):609–615
Winnik FM, Regismond ST (1996) Fluorescence methods in the study of the interactions of surfactants with polymers. Colloids Surf A Physicochem Eng Asp 118(1):1–39
Li Y, Guo Y, Xu G, Wang Z, Bao M, Sun N (2013) Dissipative particle dynamics simulation on the properties of the oil/water/surfactant system in the absence and presence of polymer. Mol Simul 39(4):299–308
Claesson PM, Makuska R, Varga I, Meszaros R, Titmuss S, Linse P, Pedersen JS, Stubenrauch C (2010) Bottle-brush polymers: adsorption at surfaces and interactions with surfactants. Adv Colloid Interface Sci 155(1):50–57
Guo Y, Chen T, Zhao N, Shang Y, Liu H (2013) Dilational properties of gemini surfactant/polymer systems at the air–water surface. Colloid Polym Sci 291(4):845–854
Zhang L, Wang XC, Yan F, Luo L, Zhang L, Zhao S, Yu JY (2008) Interfacial dilational properties of partly hydrolyzed polyacrylamide and gemini surfactant at the decane–water interface. Colloid Polym Sci 286(11):1291–1297
Goddard E (2002) Polymer/surfactant interaction: interfacial aspects. J Colloid Interface Sci 256(1):228–235
Taylor D, Thomas R, Penfold J (2007) Polymer/surfactant interactions at the air/water interface. Adv Colloid Interface Sci 132(2):69–110
Mohr A, Nylander T, Piculell L, Lindman BR, Boyko V, Bartels FW, Liu Y, Kurkal-Siebert V (2012) Mixtures of cationic copolymers and oppositely charged surfactants: effect of polymer charge density and ionic strength on the adsorption behavior at the silica–aqueous interface. ACS Appl Mater Interface 4(3):1500–1511
Liu J, Sun D, Wei X, Wang S, Yu L, Zheng L (2012) Interaction between 1-dodecyl-3-Methylimidazolium bromide and sodium carboxymethylcellulose in aqueous solution: effect of polymer concentration. J Dispersion Sci Technol 33(1):5–14
Mukherjee S, Dan A, Bhattacharya SC, Panda AK, Moulik SP (2011) Physicochemistry of interaction between the cationic polymer poly(diallyldimethylammonium chloride) and the anionic surfactants sodium dodecyl sulfate, sodium dodecylbenzenesulfonate, and sodium N-dodecanoylsarcosinate in water and isopropyl alcohol–water media. Langmuir 27(9):5222–5233
Zhang X, Taylor D, Thomas R, Penfold J (2011) Adsorption of polyelectrolyte/surfactant mixtures at the air–water interface: Modified poly(ethyleneimine) and sodium dodecyl sulfate. Langmuir 27(6):2601–2612
Martinez JC, Murciano-Calles J, Cobos ES, Iglesias-Bexiga M, Luque I, Ruiz-Sanz J (2013) Isothermal titration calorimetry: thermodynamic analysis of the binding thermograms of molecular recognition events by using equilibrium models. Intech, Croatia, pp 73–104
Becker AL, Welsch N, Schneider C, Ballauff M (2011) Adsorption of RNase A on cationic polyelectrolyte brushes: a study by isothermal titration calorimetry. Biomacromolecules 12(11):3936–3944
Chiappisi L, Li D, Wagner NJ, Gradzielski M (2014) An improved method for analyzing isothermal titration calorimetry data from oppositely charged surfactant polyelectrolyte mixtures. J Chem Thermodyn 68:48–52
Han Y, Wang Y (2011) Aggregation behavior of gemini surfactants and their interaction with macromolecules in aqueous solution. Phys Chem Chem Phys 13(6):1939–1956
Han Y, Xia L, Zhu L, Zhang S, Li Z, Wang Y (2012) Association behaviors of dodecyltrimethylammonium bromide with double hydrophilic block co-polymer poly(ethylene glycol)-block-poly(glutamate sodium). Langmuir 28(43):15134–15140
Han Y, Wang W, Tang Y, Zhang S, Li Z, Wang Y (2013) Coassembly of poly (ethylene glycol)-block-poly(glutamate sodium) and gemini surfactants with different spacer lengths. Langmuir 29(30):9316–9323
Fan YR, Li YJ, Yuan GC, Wang YL, Wang JB et al (2005) Comparative studies on the micellization of sodium bis(4-phenylbutyl) sulfosuccinate and sodium bis(2-ethylhexyl) sulfosuccinate and their interaction with hydrophobically modified poly(acrylamide). Langmuir 21(9):3814–3820
Wang MY, Kong SN, Liu S, Li CL, Wang MG, Tan YB (2014) Aggregation behavior of partially fluorinated Gemini surfactants in aqueous solution: effect of headgroups. Colloids Surf A Physicochem Eng Asp 441:25–33
Kästner U, Zana R (1999) Interactions between quaternary ammonium surfactant oligomers and water-soluble modified guars. J Colloid Interface Sci 218(2):468–479
Bai GY, Wang YJ, Yan HK (2002) Thermodynamics of interaction between cationic gemini surfactants and hydrophobically modified polymers in aqueous solutions. J Phys Chem B 106(9):2153–2159
Yoshimura T, Nagata Y, Esumi K (2004) Interactions of quaternary ammonium salt-type gemini surfactants with sodium poly(styrene sulfonate). J Colloid Interface Sci 275(2):618–622
Zana R (1996) Critical micellization concentration of surfactants in aqueous solution and free energy of micellization. Langmuir 12(5):1208–1211
Wang Y, Han Y, Huang X, Cao M, Wang Y (2008) Aggregation behaviors of a series of anionic sulfonate gemini surfactants and their corresponding monomeric surfactant. J Colloid Interface Sci 319(2):534–541
Xin X, Zhu Y, Cao X, Xu G (2014) Interaction between an ethoxylated alkylphenol polymer with formaldehyde and triblock polyEO–polyPO–polyEO Copolymer in aqueous solutions. J Surfactant Deterg 17:71–83
Bain C, Claesson PM, Langevin D et al (2010) Complexes of surfactants with oppositely charged polymers at surfaces and in bulk. Adv Colloid Interface Sci 155(1):32–49
Courtois J, Berret JF (2010) Probing oppositely charged surfactant and copolymer interactions by isothermal titration microcalorimetry. Langmuir 26(14):11750–11758
Fan Y, Li Y, Cao M, Wang J, Wang Y, Thomas RK (2007) Micellization of dissymmetric cationic gemini surfactants and their interaction with dimyristoylphosphatidylcholine vesicles. Langmuir 23(23):11458–11464
De Lisi R, Milioto S, Muratore N (2009) Thermodynamics of surfactants, block copolymers and their mixtures in water: the role of the isothermal calorimetry. Int J Mol Sci 10(7):2873–2895
Li X, Wettig SD, Verrall RE (2004) Interactions between 12-EOx-12 gemini surfactants and pluronic ABA block copolymers (F108 and P103) studied by isothermal titration calorimetry. Langmuir 20(3):579–586
Penfold J, Thomas R, Taylor D (2006) Polyelectrolyte/surfactant mixtures at the air–solution interface. Curr Opin Colloid Interface Sci 11(6):337–344
Acknowledgments
The authors gratefully acknowledge the financial support from PetroChina Innovation Foundation (Grant No. 2011D-5006-0211), Educational Commission of Hubei Province of China (Grant No. Q20141302), National Science Fund for Distinguished Young Scholars of China (Grant No. 21225313).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 356 kb)
Rights and permissions
About this article
Cite this article
Lai, L., Mei, P., Wu, XM. et al. Micellization of anionic gemini surfactants and their interaction with polyacrylamide. Colloid Polym Sci 292, 2821–2830 (2014). https://doi.org/10.1007/s00396-014-3304-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-014-3304-y