Abstract
Background
Few studies have reported a possible involvement of pleiotrophin (PTN) in the pathophysiology of osteoarthritis (OA) and very little is known about its role in rheumatoid arthritis (RA). This study is to measure PTN in the sera and synovial fluids in RA and OA and to assess its relation to activity, functional class and radiological staging.
Subjects and methods
Serum and synovial fluid samples were collected from 35 RA patients and 40 knee OA patients and serum samples were withdrawn from 20 healthy controls. Demographic, clinical and serological data were prospectively assessed. Functional and radiographic grades were also assessed. Serum and synovial fluid PTN levels were measured using enzyme-linked immunosorbent assay (ELISA).
Results
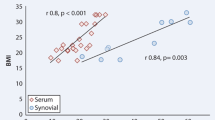
There was no statistical significant differences (p > 0.05) on comparing the mean PTN level in sera of RA, OA patients and healthy controls. However the mean synovial fluid level of PTN in both patient groups was significantly higher than mean serum level (p < 0.001). Significant correlations between the serum PTN level and both morning stiffness duration (p = 0.008) and mHAQ score (p = 0.039) were only observed in RA patients.
Conclusion
Our results point to a possible important role of PTN in RA and OA. We firstly report a serological pattern of PTN in the sera and synovial fluids of RA patients. However its implementation as a disease marker or a potential target therapy in both diseases awaits larger studies and further investigations.
Zusammenfassung
Hintergrund
In einigen wenigen Studien wurde über eine mögliche Beteiligung von Pleiotrophin (PTN) an den pathophysiologischen Prozessen der Arthrose berichtet. Nur wenig ist über die Rolle bei der rheumatoiden Arthritis (RA) bekannt. Zweck der vorliegenden Studie war es, die PTN-Spiegel in Serum und Synovia bei RA und Arthrose zu bestimmen. Zudem wurde der Zusammenhang mit der Aktivität, dem Funktionsstatus und dem radiologischen Stadium untersucht.
Probanden und Methoden
Serum- und Synoviaproben wurden von 35 Patienten mit RA und 40 Patienten mit Kniegelenksarthrose gewonnen, Serumproben auch von 20 gesunden Kontrollen. Demografische, klinische und serologische Daten wurden prospektiv erfasst. Die funktionellen und röntgenologischen Stadien wurden ebenfalls ermittelt. Die PTN-Konzentrationen in Serum und Synovia wurden mithilfe eines „enzyme-linked immunosorbent assay“ (ELISA) bestimmt.
Ergebnisse
Es fand sich kein statistisch signifikanter Unterschied (p > 0,05) bei Vergleich der durchschnittlichen PTN-Spiegel im Serum von Patienten mit RA, Patienten mit Arthrose und gesunden Kontrollen. Die durchschnittliche Konzentration in der Synovia war dagegen in beiden Patientengruppen signifikant höher als der durchschnittliche Serumspiegel (p < 0,001). Eine signifikante Korrelation fand sich nur zwischen den Serum-PTN-Spiegeln und der Dauer der Morgensteifigkeit (p = 0,008) sowie dem mHAQ-Score (p = 0,039) bei Patienten mit RA.
Schlussfolgerungen
Die Ergebnisse deuten darauf hin, dass PTN eine wichtige Rolle bei RA und Arthrose spielen könnte. Dies ist der erste Bericht über ein serologisches PTN-Muster in Serum und Synovia von Patienten mit RA. Bevor aber PTN als Erkrankungsmarker etabliert oder als potenzieller Ansatzpunkt für eine zielgerichtete Therapie beider Erkrankungen herangezogen werden kann, müssen größere Studien und weitere Untersuchungen durchgeführt werden.



Similar content being viewed by others
References
Jiao K, Zhang J, Zhang M, Wei Y, Wu Y (2013) The identification of CD163 expressing phagocytic chondrocytes in joint cartilage and its novel scavenger role in cartilage degradation. PLOS ONE 8(1):e53312
Ashraf S, Walsh DA (2008) Angiogenesis in osteoarthritis. Curr Opin Rheumatol 20(5):573–580
Takeuchi T, Yamanaka H, Ishiguro N, Miyasaka N, Mukai M, Matsubara T, Uchida S (2014) Adalimumab, outcome study for the prevention of joint damage in Japanese patients with early rheumatoid arthritis: the HOPEFUL 1 study. Ann Rheum Dis 73(3):536–543
Tanaka S (2013) Regulation of bone destruction in rheumatoid arthritis through RANKL-RANK pathways. World J Orthop 4(1):1–6
Szekanecz Z, Koch AE (2008) Targeting angiogenesis in rheumatoid arthritis. Curr Rheumatol Rev 4(4):298–303
Kaspiris A, Mikelis C, Heroult M, Khaldi L, Grivas TB, Kouvaras I, Dangas S, Vasiliadis E, Lioté F, Courty J, Papadimitriou E (2013) Expression of the growth factor pleiotrophin and its receptor protein tyrosine phosphatase beta/zeta in the serum, cartilage and subchondral bone of patients with osteoarthritis. Joint Bone Spine 80(4):407–413
Houard X, Goldring MB, Berenbaum F (2013) Homeostatic mechanisms in articular cartilage and role of inflammation in osteoarthritis. Curr Rheumatol Rep 15(11):375
Mentlein R (2007) Targeting pleiotropin to treat osteoarthritis. Expert Opin Ther Targets 11(7):861–867
Pufe T, Bartscher M, Petersen W, Tillmann B, Mentlein R (2003) Expression of pleiotrophin, an embryonic growth and differentiation factor, in rheumatoid arthritis. Arthritis Rheum 48(3):660–667
Achour A, M’bika JP, Baudouin F, Caruelle D, Courty J (2008) Pleiotrophin induces expression of inflammatory cytokines in peripheral blood mononuclear cells. Biochimie 90(11-12):1791–1795
Aletaha D, Neogi T, Silman AJ, Funovits J, Felson DT, Bingham CO 3rd, Birnbaum NS, Burmester GR, Bykerk VP, Cohen MD, Combe B, Costenbader KH, Dougados M, Emery P, Ferraccioli G, Hazes JM, Hobbs K, Huizinga TW, Kavanaugh A, Kay J, Kvien TK, Laing T, Mease P, Ménard HA, Moreland LW, Naden RL, Pincus T, Smolen JS, Stanislawska-Biernat E, Symmons D, Tak PP, Upchurch KS, Vencovský J, Wolfe F, Hawker G (2010) Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League against Rheumatism Collaborative Initiative. Arthritis Rheum 62(9):2569–2581
Altman R, Asch E, Bloch D, Bole G, Borenstein D, Brandt K, Christy W, Cooke TD, Greenwald R, Hochberg M (1986) Development of criteria for the classification and reporting of osteoarthritis. Classification of osteoarthritis of the knee. Diagnostic References and Therapeutic Criteria Committee of the American Rheumatism Association. Arthritis Rheum 29(8):1039–1049
Prevoo ML, van ’t Hof MA, Kuper HH, van Leeuwen MA, van de Putte LB, van Riel PL (1995) Modified disease activity scores that include twenty-eight-joint counts. Development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum 38(1):44–48
Pincus T, Summey JA, Soraci SA Jr, Wallston KA, Hummon NP (1983) Assessment of patient satisfaction in activities of daily living using a modified Stanford Health Assessment Questionnaire. Arthritis Rheum 26(11):1346–1353
Rau R, Herborn G (1995) A modified version of Larsen’s scoring method to assess radiologic changes in rheumatoid arthritis. J Rheumatol 22(10):1976–1982
Kellgren JH, Lawrence JS (1957) Radiological assessment of Osteo-Arthrosis. Ann Rheum Dis 16(4):494–502
Bellamy N (1982) Osteoarthritis – An evaluative index for clinical trials. MSc Thesis, McMaster University, Hamilton, Canada.
Burska A, Boissinot M, Ponchel F (2014) Cytokines as biomarkers in rheumatoid arthritis. Mediators Inflamm. doi:10.1155/2014/545493
National Cancer Institute (2014) Targeted cancer therapies. www.cancer.gov/cancertopics/factsheet/Therapy/targeted
Kuek A, Hazleman BL, Östör AJK (2007) Immune-mediated inflammatory diseases (IMIDs) and biologic therapy: a medical revolution. Post Grad Med J 83(978):251–260
Souttou B, Carvalho NB, Raulais D, Vigny M (2001) Activation of anaplastic lymphoma kinase receptor tyrosine kinase induces neuronal differentiation through the mitogen-activated protein kinase pathway. J Biol Chem 276(12):9526–9531
Tsirmoula S, Dimas K, Hatziapostolou M, Lamprou M, Ravazoula P, Papadimitriou E (2012) Implications of pleiotrophin in human PC3 prostate cancer cell growth in vivo. Cancer Sci 103(10):1826–1832
Lynn KD, Roland CL, Brekken RA (2010) VEGF and pleiotrophin modulate the immune profile of breast cancer. Cancers (Basel) 2(2):970–988
Straub RH, Cutolo M (2007) Circadian rhythms in rheumatoid arthritis: implications for pathophysiology and therapeutic management. Arthritis Rheum 56(2):399–408
Ebrahimi AA, Noshad H, Sadreddini S et al (2009) Serum levels of TNF-alpha, TNF-alpha RI, TNF-alpha RII and IL-12 in treated rheumatoid arthritis patients. Iran J Immunol 6(3):147–153
Goëb V, Aegerter P, Parmar R et al (2013) Progression to rheumatoid arthritis in early inflammatory arthritis is associated with low IL-7 serum levels. Ann Rheum Dis 72(6):1032–1036
Pufe T, Bartscher M, Petersen W, Tillmann B, Mentlein R (2003) Pleiotrophin, an embryonic differentiation and growth factor, is expressed in osteoarthritis. Osteoarthr Cartil 11(4):260–264
Peterson WJ, Tachiki KH, Yamaguchi DT (2004) Serial passage of MC3T3-E1 cells down-regulates proliferation during osteogenesis in vitro. Cell Prolif 37(5):325–336
Li G, Cui Y, Mcilmurray L, We A, Wang H (2005) rhBMP-2, rhVEGF (165), rhPTN and thrombin related peptide, TP508 induce chemotaxis of human osteoblasts and microvascular endothelial cells. J Orthop Res 23:680–685
Yang X, Tare RS, Partridge KA (2003) Induction of human osteoprogenitor chemotaxis, proliferation, differentiation, and bone formation by osteoblast stimulating factor-1/pleiotrophin:osteoconductive biomimetic scaffolds for tissue engineering. J Bone Miner Res 18:47–57
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
S. M. H. Fadda, I. H. Bassyouni, R. H. Khalifa and N. Y. Elsaid declare that they have no competing interests.
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Redaktion
U. Müller-Ladner, Bad Nauheim
U. Lange, Bad Nauheim
Rights and permissions
About this article
Cite this article
Fadda, S.M.H., Bassyouni, I.H., Khalifa, R.H. et al. Pleiotrophin, the angiogenic and mitogenic growth factor: levels in serum and synovial fluid in rheumatoid arthritis and osteoarthritis. Z Rheumatol 77, 322–329 (2018). https://doi.org/10.1007/s00393-016-0234-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00393-016-0234-8