Abstract
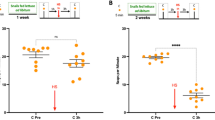
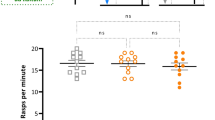
Taste aversion learning is universal. In animals, a single presentation of a novel food substance followed hours later by visceral illness causes animals to avoid that taste. This is known as bait-shyness or the Garcia effect. Humans demonstrate this by avoiding a certain food following the development of nausea after ingesting that food (‘Sauce Bearnaise effect’). Here, we show that the pond snail Lymnaea stagnalis is capable of the Garcia effect. A single ‘pairing’ of a novel taste, a carrot slurry followed hours later by a heat shock stressor (HS) is sufficient to suppress feeding response elicited by carrot for at least 24 h. Other food tastes are not suppressed. If snails had previously been exposed to carrot as their food source, the Garcia-like effect does not occur when carrot is ‘paired’ with the HS. The HS up-regulates two heat shock proteins (HSPs), HSP70 and HSP40. Blocking the up-regulation of the HSPs by a flavonoid, quercetin, before the heat shock, prevented the Garcia effect in the snails. Finally, we found that snails exhibit Garcia effect following a period of food deprivation but the long-term memory (LTM) phenotype can be observed only if the animals are tested in a food satiated state.









Similar content being viewed by others
Abbreviations
- C:
-
Carrot slurry
- CTA:
-
Conditioned taste aversion
- HS:
-
Heat shock
- HSP:
-
Heat shock protein
- ISI:
-
Inter-stimulus intervals
- LTM:
-
Long-term memory
- PW:
-
Pond water
- Q:
-
Quercetin
References
Ambrosini MV, Mariucci G, Tantucci M, Van Hooijdonk L, Ammassari-Teule M (2005) Hippocampal 72-kDa heat shock protein expression varies according to mice learning performance independently from chronic exposure to stress. Hippocampus 15:413–417. https://doi.org/10.1002/hipo.20069
Andrews EA, Braveman NS (1975) The combined effects of dosage level and interstimulus interval on the formation of one-trial poison-based aversions in rats. Anim Learn Behav 3:287–289. https://doi.org/10.3758/BF03213446
Aonuma H, Kaneda M, Hatakeyama D, Kaneda M, Hatakeyama D, Watanabe T, Lukowiak K, Ito E (2016) Relationship between the grades of a learned aversive-feeding response and the dopamine contents in Lymnaea. Biol Open 5:1869–1873. https://doi.org/10.1242/bio.021634
Aonuma H, Totani Y, Kaneda M, Nakamura R, Watanabe T, Hatakeyama D, Dyakonova VE, Lukowiak K, Ito E (2018) Effects of 5-HT and insulin on learning and memory formation in food-deprived snails. Neurobiol Learn Mem 148:20–29. https://doi.org/10.1016/j.nlm.2017.12.010
Asnicar F, Berry SE, Valdes AM, Valdes AM, Nguyen L, Piccinno GM, Drew D, Leeming E, Gibson R, Le Roy C, Khatib HA, Francis L, Mazidi M, Mompeo O, Valles-Colomer M, Tett A, Beghini F, Dubois L, Bazzani D, Thomas AM, Mirzayi C, Khleborodova A, Oh S, Hine R, Bonnett C, Capdevila J, Danzanvilliers S, Giordano F, Geistlinger L, Waldron L, Davies R, Hadjigeorgiou G, Wolf J, Ordovás JM, Gardner C, Franks PW, Chan AT, Huttenhower C, Spector TD, Segata N (2021) Microbiome connections with host metabolism and habitual diet from 1098 deeply phenotyped individuals. Nat Med. https://doi.org/10.1038/s41591-020-01183-8
Becker JA, Stewart LK (2011) Heat-related illness. AFP 83:1325–1330
Benatti C, Alboni S, Montanari C, Alboni S, Montanari C, Caggia F, Tascedda F, Brunello N, Blom JMC (2011) Central effects of a local inflammation in three commonly used mouse strains with a different anxious phenotype. Behav Brain Res 224:23–34. https://doi.org/10.1016/j.bbr.2011.05.011
Benjamin P, Kemenes G (2010) Lymnaea learning and memory. Scholarpedia 5:4247. https://doi.org/10.4249/scholarpedia.4247
Bermúdez-Rattoni F (2004) Molecular mechanisms of taste-recognition memory. Nat Rev Neurosci 5:209–217. https://doi.org/10.1038/nrn1344
Chambers KC (2018) Conditioned taste aversions. World J Otorhinolaryngol Head Neck Surg 4:92–100. https://doi.org/10.1016/j.wjorl.2018.02.003
Crossley M, Staras K, Kemenes G (2016) A two-neuron system for adaptive goal-directed decision-making in Lymnaea. Nat Comm 7(1):1–13
Crossley M, Staras K, Kemenes G (2018) A central control circuit for encoding perceived food value. Sci Adv. https://doi.org/10.1126/sciadv.aau9180
Dalesman S, Rundle S (2010) Influence of rearing and experimental temperatures on predator avoidance behaviour in a freshwater pulmonate snail. Freshw Biol 55:2107–2113. https://doi.org/10.1111/j.1365-2427.2010.02470.x
Davey VA, Biederman GB (1996) Conditioned antisickness: Heat as an internal stimulus in conditioning taste aversion and aversion failure in rats. J Exp Psychol Anim Behav Proc 22:235–243. https://doi.org/10.1037/0097-7403.22.3.235
Domjan M, Bowman TG (1974) Learned safety and the cs-us delay gradient in taste-aversion learning. Learn Motiv 5:409–423. https://doi.org/10.1016/0023-9690(74)90001-0
Dong Y, Dong S (2008) Induced thermotolerance and expression of heat shock protein 70 in sea cucumber Apostichopus japonicus. Fish Sci 74:573–578. https://doi.org/10.1111/j.1444-2906.2008.01560.x
Dragoin WB (1971) Conditioning and extinction of taste aversions with variations in intensity of the CS and UCS in two strains of rats. Psychon Sci 22:303–305. https://doi.org/10.3758/BF03335967
Elkins RL (1973) Individual differences in bait shyness: effects of drug dose and measurement technique. Psychol Rec 23:349–358. https://doi.org/10.1007/BF03394178
Etscorn F, Stephens R (1973) Establishment of conditioned taste aversions with a 24-hour CS-US interval. Psychobiol 1:251–253. https://doi.org/10.3758/BF03326916
Foster NL, Lukowiak K, Henry TB (2015) Time-related expression profiles for heat shock protein gene transcripts (HSP40, HSP70) in the central nervous system of Lymnaea stagnalis exposed to thermal stress. Commun Integr Biol 8:e1040954. https://doi.org/10.1080/19420889.2015.1040954
Fulton D, Kemenes I, Andrew RJ, Benjamin PR (2005) A single time-window for protein synthesis-dependent long-term memory formation after one-trial appetitive conditioning. Eur J Neurosci 21:1347–1358. https://doi.org/10.1111/j.1460-9568.2005.03970.x
Garb JL, Stunkard AJ (1974) Taste aversions in man. AJP 131:1204–1207. https://doi.org/10.1176/ajp.131.11.1204
Garcia J, Kimeldorf DJ, Koelling RA (1955) Conditioned aversion to saccharin resulting from exposure to gamma radiation. Science 122:157–158. https://doi.org/10.1126/science.122.3160.157
Garcia J, Ervin FR, Koelling RA (1966) Learning with prolonged delay of reinforcement. Psychon Sci 5:121–122. https://doi.org/10.3758/BF03328311
Garcia J, Ervin FR, Yorke CH, Koelling RA (1967) Conditioning with delayed vitamin injections. Science 155:716–718. https://doi.org/10.1126/science.155.3763.716
Garcia J, Hankins WG, Rusiniak KW (1974) Behavioral regulation of the milieu interne in man and rat. Science 185:824–831. https://doi.org/10.1126/science.185.4154.824
Green L, Rachlin H (1976) Learned taste aversions in rats as a function of delay, speed, and duration of rotation. Learn Motiv 7:283–289. https://doi.org/10.1016/0023-9690(76)90035-7
Harris CA, Passaro PA, Kemenes I, Kemenes G, O’Shea M (2010) Sensory driven multi-neuronal activity and associative learning monitored in an intact CNS on a multielectrode array. J Neurosci Methods 186:171–178. https://doi.org/10.1016/j.jneumeth.2009.11.014
Hatakeyama D, Okuta A, Otsuka E, Lukowiak K, Ito E (2013) Consolidation of long-term memory by insulin in Lymnaea is not brought about by changing the number of insulin receptors. Commun Integr Biol. https://doi.org/10.4161/cib.23955
Hermann PM, Park D, Beaulieu E, Wildering WC (2013) Evidence for inflammation-mediated memory dysfunction in gastropods: putative PLA2 and COX inhibitors abolish long-term memory failure induced by systemic immune challenges. BMC Neurosci 14:83. https://doi.org/10.1186/1471-2202-14-83
Hosokawa N, Hirayoshi K, Nakai A, Marui N, Yoshida M, Sakai T, Nishino H, Aoike A, Kawai K (1990) Flavonoids inhibit the expression of heat shock proteins. Cell Struct Funct 15:393–401. https://doi.org/10.1247/csf.15.393
Hung C-H, Lin M-T, Liao J-F, Wang J-J (2004) Scopolamine-induced amnesia can be prevented by heat shock pretreatment in rats. Neurosci Lett 364:63–66. https://doi.org/10.1016/j.neulet.2004.02.074
Igaz LM, Bekinschtein P, Izquierdo I, Medina JH (2004) One-trial aversive learning induces late changes in hippocampal CaMKIIalpha, Homer 1a, Syntaxin 1a and ERK2 protein levels. Brain Res Mol Brain Res 132:1–12. https://doi.org/10.1016/j.molbrainres.2004.08.016
Ito E, Yamagishi M, Hatakeyama D, Watanabe T, Fujito Y, Dyakonova V, Lukowiak K (2015a) Memory block: a consequence of conflict resolution. J Exp Biol 218:1699–1704. https://doi.org/10.1242/jeb.120329
Ito E, Yamagishi M, Takigami S, Sakakibara M, Fujito Y, Lukowiak K (2015b) The Yerkes-Dodson law and appropriate stimuli for conditioned taste aversion in Lymnaea. J Exp Biol 218:336–339. https://doi.org/10.1242/jeb.113266
Kagan D, Lukowiak K (2019) Configural learning in freshly collected, smart, wild Lymnaea. J Exp Biol. https://doi.org/10.1242/jeb.212886
Kalat JW, Rozin P (1971) Role of interference in taste-aversion learning. J Comp Physiol Psychol 77:53–58. https://doi.org/10.1037/h0031585
Kawai R, Sunada H, Horikoshi T, Sakakibara M (2004) Conditioned taste aversion with sucrose and tactile stimuli in the pond snail Lymnaea stagnalis. Neurobiol Learn Mem 82:164–168. https://doi.org/10.1016/j.nlm.2004.06.003
Kojima S, Yamanaka M, Fujito Y, Ito E (1996) Differential neuroethological effects of aversive and appetitive reinforcing stimuli on associative learning in Lymnaea stagnalis. J Zool 13:803–812. https://doi.org/10.2108/zsj.13.803
La M, Jj H (2007) Examination of KNK437- and quercetin-mediated inhibition of heat shock-induced heat shock protein gene expression in Xenopus laevis cultured cells. Comp Biochem Physiol A Mol Integr Physiol 148:521–530. https://doi.org/10.1016/j.cbpa.2007.06.422
Lackie RE, Maciejewski A, Ostapchenko VG, Marques-Lopes J, Choy WY, Duennwald ML, Prado VF, Prado MAM (2017) The HSP70/HSP90 chaperone machinery in neurodegenerative diseases. Front Neurosci. https://doi.org/10.3389/fnins.2017.00254
Lindquist S, Craig EA (1988) The heat-shock proteins. Annu Rev Genet 22:631–677. https://doi.org/10.1146/annurev.ge.22.120188.003215
Logue AW, Ophir I, Strauss KE (1981) The acquisition of taste aversions in humans. Behav Res Ther 19:319–333. https://doi.org/10.1016/0005-7967(81)90053-X
Lorenz K (1965) Evolution and the modification of behavior. Univ. of Chicago Press, Chicago
Lukowiak K, Sangha S, Scheibenstock A, Parvez K, McComb C, Rosenegger D, Varshney N, Sadamoto H (2003) A molluscan model system in the search for the engram. J Physiol Paris 97:69–76. https://doi.org/10.1016/j.jphysparis.2003.10.008
Lukowiak K, Fras M, Smyth K, Wong C, Hittel K (2007) Reconsolidation and memory infidelity in Lymnaea. Neurobiol Learn Mem 87(4):547–560
McDonald SC (1973) Activity patterns of Lymnaea stagnalis (L.) in relation to temperature conditions: a preliminary study. Malacologia 14:395–396
Mclaurin WA, Scarborough BB (1963) Extension of the interstimulus interval in saccharin avoidance conditioning. Radiat Res 20:317–324
Nachman M, Ashe JH (1973) Learned taste aversions in rats as a function of dosage, concentration, and route of administration of LiCl. Physiol Behav 10:73–78. https://doi.org/10.1016/0031-9384(73)90089-9
Nakai J, Totani Y, Kojima S, Kojima S, Sakakibara M, Ito E (2020) Features of behavioral changes underlying conditioned taste aversion in the pond snail Lymnaea stagnalis. Invert Neurosci 20:8. https://doi.org/10.1007/s10158-020-00241-7
Nakamura H, Ito I, Kojima S, Fujito Y, Suzuki H, Ito E (1999) Histological characterization of lip and tentacle nerves in Lymnaea stagnalis. Neurosci Res 33:127–136. https://doi.org/10.1016/S0168-0102(98)00121-7
Ottaviani E, Malagoli D, Franceschi C (2007) Common evolutionary origin of the immune and neuroendocrine systems: from morphological and functional evidence to in silico approaches. Trends Immunol 28:497–502. https://doi.org/10.1016/j.it.2007.08.007
Pizarro JM, Haro LS, Barea-Rodriguez EJ (2003) Learning associated increase in heat shock cognate 70 mRNA and protein expression. Neurobiol Learn Mem 79:142–151. https://doi.org/10.1016/s1074-7427(02)00008-4
Porto RR, Dutra FD, Crestani AP, Crestani AP, Holsinger RMD, Quillfeldt JA, de Homem Bittencourt Jr. PI, Oliveira Alvares L (2018) HSP70 Facilitates memory consolidation of fear conditioning through MAPK pathway in the hippocampus. Neuroscience 375:108–118. https://doi.org/10.1016/j.neuroscience.2018.01.028
Revusky SH (1968) Aversion to sucrose produced by contingent x-irradiation: temporal and dosage parameters. J Comp Physiol Psychol 65:17–22. https://doi.org/10.1037/h0025416
Ribeiro MJ, Schofield MG, Kemenes I, O’Shea M, Kemenes G, Benjamin PR (2005) Activation of MAPK is necessary for long-term memory consolidation following food-reward conditioning. Learn Mem 12:538–545. https://doi.org/10.1101/lm.8305
Rivi V, Benatti C, Colliva C, Radighieri G, Brunello N, Tascedda F, Blom JMC (2020) Lymnaea stagnalis as model for translational neuroscience research: from pond to bench. Neurosci Biobehav Rev 108:602–616. https://doi.org/10.1016/j.neubiorev.2019.11.020
Rzóska J (1953) Bait shyness, a study in rat behaviour. Br J Anim Behav 1:128–135. https://doi.org/10.1016/S0950-5601(53)80011-0
Sahley C, Rudy JW, Gelperin A (1981) An analysis of associative learning in a terrestrial mollusc. J Comp Physiol 144:1–8. https://doi.org/10.1007/BF00612791
Schleyer M, Fendt M, Schuller S, Gerber B (2018) Associative Learning of stimuli paired and unpaired with reinforcement: evaluating evidence from maggots, flies, bees, and rats. Front Psychol 9:1494. https://doi.org/10.3389/fpsyg.2018.01494
Seligman ME (1970) On the generality of the laws of learning. Psychol Rev 77:406–418. https://doi.org/10.1037/h0029790
Seppälä O, Jokela J (2011) Immune defence under extreme ambient temperature. Biol Lett 7:119–122. https://doi.org/10.1098/rsbl.2010.0459
Smith JC, Roll DL (1967) Trace conditioning with X-rays as an aversive stimulus. Psychon Sci 9:11–12. https://doi.org/10.3758/BF03330734
Song H-M, Mu X-D, Gu D-E, Luo D, Yang Y-X, Xu M, Luo J-R, Zhang H-C (2014) Molecular characteristics of the HSP70 gene and its differential expression in female and male golden apple snails (Pomacea canaliculata) under temperature stimulation. Cell Stress Chaperones 19:579–589. https://doi.org/10.1007/s12192-013-0485-0
Storniolo A, Raciti M, Cucina A, Bizzarri M, Di Renzo L (2015) Quercetin affects Hsp70/IRE1α mediated protection from death induced by endoplasmic reticulum stress. Oxid Med Cell Longev. https://doi.org/10.1155/2015/645157
Straub VA, Styles BJ, Ireland JS, O’Shea M, Benjamin PR (2004) Central localization of plasticity involved in appetitive conditioning in Lymnaea. Learn Mem 11(6):787–793
Sugai R, Shiga H, Azami S, Watanabe T, Sadamoto H, Fujito Y, Lukowiak K (2006) Taste discrimination in conditioned taste aversion of the pond snail Lymnaea stagnalis. J Exp Biol 209:826–833. https://doi.org/10.1242/jeb.02069
Sunada H, Riaz H, de Freitas E, Lukowiak KS, Swinton C, Swinton E, Protheroe A, Shymansky T, Komatsuzaki Y, Lukowiak K (2016) Heat stress enhances LTM formation in Lymnaea: role of HSPs and DNA methylation. J Exp Biol 219:1337–1345. https://doi.org/10.1242/jeb.134296
Swinton C, Swinton E, Shymansky T, Hughes E, Zhang J, Rothwell C, Kakadiya M, Lukowiak K (2019) Configural learning: a higher form of learning in Lymnaea. J Exp Biol. https://doi.org/10.1242/jeb.190405
Takigami S, Sunada H, Lukowiak K, Sakakibara M (2013) High voltage with little current as an unconditional stimulus for taste avoidance conditioning in Lymnaea stagnalis. Neurosci Lett 555:149–153
Tascedda F, Malagoli D, Accorsi A, Rigillo G, Blom JMC, Ottaviani E (2015) Molluscs as models for translational medicine. Med Sci Monit Basic Res 21:96–99. https://doi.org/10.12659/MSMBR.894221
Teskey ML, Lukowiak KS, Riaz H, Dalesman S, Lukowiak K (2012) What’s hot: the enhancing effects of thermal stress on long-term memory formation in Lymnaea stagnalis. J Exp Biol 215:4322–4329. https://doi.org/10.1242/jeb.075960
Totani Y, Nakai J, Dyakonova VE, Lukowiak K, Sakakibara M, Etsuro I (2020) Induction of LTM following an insulin injection. Eneuro. https://doi.org/10.1523/ENEURO.0088-20.2020
Vaughn CM (1953) Effects of temperature on hatching and growth of Lymnaea stagnalis appressa say. Am Midl Nat 49:214–228. https://doi.org/10.2307/2422289
Vogel EH, Soto FA, Castro ME, Solar PA (2007) Stimulus specificity in the acquisition and extinction of conditioned taste aversion. Biol Res 40:123–129. https://doi.org/10.4067/S0716-97602007000200003
Wagatsuma A, Sugai R, Chono K, Azami S, Hatakeyama D, Sadamoto H, Ito E (2004) The early snail acquires the learning. Comparison of scores for conditioned taste aversion between morning and afternoon. Acta Biol Hung 55:149–155. https://doi.org/10.1556/ABiol.55.2004.1-4.18
Wang RE, Kao JL-F, Hilliard CA, Pandita RK, Roti Roti JL, Hunt CR, Taylor J-S (2009) Inhibition of heat shock induction of heat shock protein 70 and enhancement of Heat Shock Protein 27 phosphorylation by quercetin derivatives. J Med Chem 52:1912–1921. https://doi.org/10.1021/jm801445c
Acknowledgements
We thank Diana Kagan and Bevin Wiley for discussions during the research. This research was supported by Regione Emilia-Romagna “L’invertebrato L. stagnalis quale modello per la Medicina Traslazionale” L.R. N. 20/2002 PROGETTI DI RICERCA SUI METODI ALTERNATIVI ALL'UTILIZZO DI ANIMALI; and FAR 2016 Department of Life Sciences, University of Modena and Reggio Emilia. Behavioural trials were performed in the University of Calgary and funding was provided by the Natural Sciences and Engineering Research Council of Canada (NSERC) to K.L.
Funding
This research was supported by Regione Emilia-Romagna “L’invertebrato L. stagnalis quale modello per la Medicina Traslazionale” L.R. N. 20/2002 PROGETTI DI RICERCA SUI METODI ALTERNATIVI ALL'UTILIZZO DI ANIMALI; and FAR 2016 Department of Life Sciences, University of Modena and Reggio Emilia. Behavioural trials were performed in the University of Calgary and funding was provided by the Natural Sciences and Engineering Research Council of Canada (NSERC) to K.L. These sources of funding had no involvement in the study design, data collection, analysis, and interpretation, writing of the report, or in the decision to submit the paper for publication.
Author information
Authors and Affiliations
Contributions
Conceptualization: VR, AB, KJ, MK and KL; methodology: VR, CB, and KL; data collection and analyses: VR and AB; writing—original draft: VR; writing—review and editing: AB and KL; funding acquisition: JMCB and KL; resources: JMCB and KL; supervision: KL.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Ethical approval
The pond snails used in our study, Lymnaea stagnalis are found in the Holarctic geographical region in abundant numbers and are neither an endangered nor a protected species. Experiments on pond snails are not subject to the approval of our respective animal ethics committees. Nonetheless, every effort was made to minimize the number of animals used ensuring adequate food, clean oxygenated water, and low-density conditions. The heat stress treatment used in our experiments has proven to have no long-term negative effects on the animals beyond the brief exposure period.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rivi, V., Batabyal, A., Juego, K. et al. To eat or not to eat: a Garcia effect in pond snails (Lymnaea stagnalis). J Comp Physiol A 207, 479–495 (2021). https://doi.org/10.1007/s00359-021-01491-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-021-01491-5