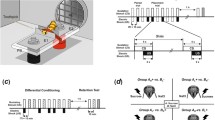
Abstract
Associative learning enables animals to predict rewards or punishments by their associations with predictive stimuli, while non-associative learning occurs without reinforcement. The latter includes latent inhibition (LI), whereby animals learn to ignore an inconsequential ‘familiar’ stimulus. Individual honey bees display heritable differences in expression of LI. We examined the behavioral and neuronal responses between honey bee genetic lines exhibiting high and low LI. We observed, as in previous studies, that high LI lines learned a familiar odor more slowly than low LI bees. By measuring gustatory responses to sucrose, we determined that perception of sucrose reward was similar between both lines, thereby not contributing to the LI phenotype. We then used extracellular electrophysiology to determine differences in neural responses of the antennal lobe (AL) to familiar and novel odors between the lines. Low LI bees responded significantly more strongly to both familiar and novel odors than the high LI bees, but the lines showed equivalent differences in response to the novel and familiar odors. This work suggests that some effects of genotype are present in early olfactory processing, and those effects could complement how LI is manifested at later stages of processing in brains of bees in the different lines.




Similar content being viewed by others
Availability of data and material
References
Chandra SBC, Hosler JS, Smith BH (2000) Heritable variation for latent inhibition and its correlation with reversal learning in honeybees (Apis mellifera). J Comp Psychol 114(1):86–97. https://doi.org/10.1037/0735-7036.114.1.86
Chandra SBC, Hunt GJ, Cobey S, Smith BH (2001) Quantitative trait loci associated with reversal learning and latent inhibition in honeybees (Apis mellifera). Behav Genet 31(3):275–285. https://doi.org/10.1023/A:1012227308783
Chandra SBC, Wright GA, Smith BH (2010) Latent inhibition in the honey bee, Apis mellifera: Is it a unitary phenomenon? Anim Cogn 13(6):805–815. https://doi.org/10.1007/s10071-010-0329-6
Clark AJM, Feldon J, Rawlins JNP (1992) Aspiration lesions of rat ventral hippocampus disinhibit responding in conditioned suppression or extinction, but spare latent inhibition and the partial reinforcement extinction effect. Neuroscience 48(4):821–829. https://doi.org/10.1016/0306-4522(92)90270-C
Cook CN, Mosqueiro T, Brent CS, Ozturk C, Gadau J, Pinter‐Wollman N, Smith BH (2018) Individual differences in learning and biogenic amine levels influence the behavioural division between foraging honeybee scouts and recruits. J Anim Ecol 88(2):9. https://doi-org.ezproxy1.lib.asu.edu/10.1111/1365-2656.12911
Cook CN, Lemanski NJ, Mosqueiro T, Ozturk C, Gadau J, Pinter-Wollman N, Smith BH (2020) Individual learning phenotypes drive collective behavior. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.1920554117
Das S, Sadanandappa MK, Dervan A, Larkin A, Lee JA, Sudhakaran IP, Priya R, Heidari R, Holohan EE, Pimentel A, Gandhi A, Ito K, Sanyal S, Wang JW, Rodrigues V, Ramaswami M (2011) Plasticity of local GABAergic interneurons drives olfactory habituation. Proc Natl Acad Sci USA 108(36):E646–E654. https://doi.org/10.1073/pnas.1106411108
Drezner-Levy T, Smith BH, Shafir S (2009) The effect of foraging specialization on various learning tasks in the honey bee (Apis mellifera). Behav Ecol Sociobiol 64(1):135. https://doi.org/10.1007/s00265-009-0829-z
Galizia CG, Vetter R (2004) Optical methods for analyzing odor-evoked activity in the insect brain. In: Christensen TA (ed) Advances in insect sensory neuroscience. CRC Press, Boca Raton, pp 349–392
Ginton A, Urca G, Lubow RE (1975) The effects of preexposure to a nonattended stimulus on subsequent learning: Latent inhibition in adults. Bull Psych Soc 5(1):5–8. https://doi.org/10.3758/BF03336682
Gray CM, Maldonado PE, Wilson M, McNaughton B (1995) Tetrodes markedly improve the reliability and yield of multiple single-unit isolation from multi-unit recordings in cat striate cortex. J Neurosci Met 63(1):43–54. https://doi.org/10.1016/0165-0270(95)00085-2
Hattori D, Aso Y, Swartz KJ, Rubin GM, Abbott LF, Axel R (2017) Representations of novelty and familiarity in a mushroom body compartment. Cell 169(5):956-969.e917. https://doi.org/10.1016/j.cell.2017.04.028
Latshaw JS, Smith BH (2005) Heritable variation in learning performance affects foraging preferences in the honey bee (Apis mellifera). Behav Ecol Sociobiol 58(2):200–207. https://doi.org/10.1007/s00265-004-0904-4
Lei H, Christensen TA, Hildebrand JG (2004) Spatial and temporal organization of ensemble representations for different odor classes in the moth antennal lobe. J Neurosci 24:11108–11119
Locatelli FF, Fernandez PC, Villareal F, Muezzinoglu K, Huerta R, Galizia CG, Smith BH (2013) Nonassociative plasticity alters competitive interactions among mixture components in early olfactory processing. Eur J Neurosci 37(1):63–79
Lubow RE (1973) Latent inhibition. Psychol Bull 79(6):398–407. https://doi.org/10.1037/h0034425
Lubow RE, Gewirtz JC (1995) Latent inhibition in humans: Data, theory, and implications for schizophrenia. Psychol Bull 117(1):87–103. https://doi.org/10.1037/0033-2909.117.1.87
Lubow RE, Moore AU (1959) Latent inhibition: The effect of nonreinforced pre-exposure to the conditional stimulus. J Comp Physiol Psychol 52(4):415–419. https://doi.org/10.1037/h0046700
Meyer A, Galizia CG, Nawrot MP (2013) Local interneurons and projection neurons in the antennal lobe from a spiking point of view. J Neurophysiol 110(10):2465–2474. https://doi.org/10.1152/jn.00260.2013
Moauro MA, Balbuena MS, Farina WM (2018) Assessment of appetitive behavior in honey bee dance followers. Front Behav Neurosci 12 (74). doi:https://doi.org/10.3389/fnbeh.2018.00074
Page RE Jr, Erber J, Fondrk MK (1998) The effect of genotype on response thresholds to sucrose and foraging behavior of honey bees (Apis mellifera L.). J Comp Physiol A 182(4):489–500. https://doi.org/10.1007/s003590050196
Pankiw T, Waddington KD, Page RE (2001) Modulation of sucrose response thresholds in honey bees (Apis mellifera L.): influence of genotype, feeding, and foraging experience. J Comp Physiol A 187(4):293–301. https://doi.org/10.1007/s003590100201
Pearce JM, Bouton ME (2001) Theories of associative learning in animals. Ann Rev Psychol 52(1):111–139. https://doi.org/10.1146/annurev.psych.52.1.111
Puga F, Barrett DW, Bastida CC, Gonzalez-Lima F (2007) Functional networks underlying latent inhibition learning in the mouse brain. NeuroImage 38(1):171–183. https://doi.org/10.1016/j.neuroimage.2007.06.031
R Core Team (2019) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Rey HG, Pedreira C, Quian Quiroga R (2015) Past, present and future of spike sorting techniques. Brain Res Bull 119:106–117. https://doi.org/10.1016/j.brainresbull.2015.04.007
Scheiner R, Abramson CI, Brodschneider R, Crailsheim K, Farina WM, Fuchs S, Grünewald B, Hahshold S, Karrer M, Koeniger G, Koeniger N, Menzel R, Mujagić S, Radspieler G, Schmickl T, Schneider CW, Siegel AJ, Szopek M, Thenius R (2013) Standard methods for behavioural studies of Apis mellifera. J Api Res 52:58
Smith BH, Burden CM (2014) A proboscis extension response protocol for investigating behavioral plasticity in insects: Application to basic, biomedical, and agricultural research. JoVE 91:51057. https://doi.org/10.3791/51057
Strube-Bloss MF, Herrera-Valdez MA, Smith BH (2012) Ensemble response in mushroom body output neurons of the honey bee outpaces spatiotemporal odor processing two synapses earlier in the antennal lobe. PLoS ONE 7(11):e50322
Weiner I, Feldon J (1997) The switching model of latent inhibition: an update of neural substrates. Behav Brain Res 88(1):11–25. https://doi.org/10.1016/S0166-4328(97)02314-0
Winnington AP, Napper RM, Mercer A (1996) Structural plasticity of identified glomeruli in the antennal lobes of the adult worker honey bee. J Comp Neurol 365:479–490
Acknowledgements
We would like to thank the undergraduates Eda Sezen and Alexa Phillips for their help collecting behavioral data; and Drs. Patel, Iaci and Navas Zuloaga for assistance in statistics. Furthermore, we greatly appreciate Cahit Ozturk for doing the artificial insemination of queens and maintaining the learning lines. This research is funded by National Science Foundation (1556337) and National Institute of Health (NIGMS) (GM113967) to BHS, NIH NIGMS (F32GM126728) to CNC, and a subaward to BHS as part of the NSF/CIHR/DFG/FRQ/UKRI-MRC Next Generation Networks for Neuroscience Program.
Funding
This research is funded by National Science Foundation (1556337) and National Institute of Health (NIGMS) (GM113967) to BHS; NIH NIGMS (F32GM126728) to CNC; a subaward to BHS as part of the NSF/CIHR/DFG/FRQ/UKRI-MRC Next Generation Networks for Neuroscience Program.
Author information
Authors and Affiliations
Contributions
MB and CC performed the experiments; MB, CC and HL analyzed the data and revised manuscript; MB wrote the manuscript; BS and HL conceptualized the project and revised the manuscript; BS secured financial support.
Corresponding author
Ethics declarations
Conflict of interest
The authors report no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bennett, M.M., Cook, C.N., Smith, B.H. et al. Early olfactory, but not gustatory processing, is affected by the selection of heritable cognitive phenotypes in honey bee. J Comp Physiol A 207, 17–26 (2021). https://doi.org/10.1007/s00359-020-01451-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-020-01451-5