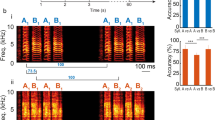
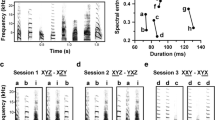
Abstract
Auditory feedback (AF) plays a critical role in vocal learning. Previous studies in songbirds suggest that low-frequency (<~1 kHz) components may be salient cues in AF. We explored this with auditory stimuli including the bird’s own song (BOS) and BOS variants with increased relative power at low frequencies (LBOS). We recorded single units from BOS-selective neurons in two forebrain nuclei (HVC and Area X) in anesthetized zebra finches. Song-evoked responses were analyzed based on both rate (spike counts) and temporal coding of spike trains. The BOS and LBOS tended to evoke similar spike-count responses in substantially overlapping populations of neurons in both HVC and Area X. Analysis of spike patterns demonstrated temporal coding information that discriminated among the BOS and LBOS stimuli significantly better than spike counts in the majority of HVC (94 %) and Area X (85 %) neurons. HVC neurons contained more and a broader range of temporal coding information to discriminate among the stimuli than Area X neurons. These results are consistent with a role of spike timing in coding differences in the spectral components of BOS in HVC and Area X neurons.






Similar content being viewed by others
Abbreviations
- AF:
-
Auditory Feedback
- BOS:
-
Bird's own song
- LBOS:
-
Low frequency BOS
- revBOS:
-
time-reversed BOS
References
Anisimov VN, Herbst JA, Abramchuk AN, Latanov AV, Hahnloser RHR, Vyssotski AL (2014) Reconstruction of vocal interactions in a group of small songbirds. Nat Methods 11:1135–1137. doi:10.1038/nmeth.3114
Aronov D (2003) Fast algorithm for the metric-space analysis of simultaneous responses of multiple single neurons. J Neurosci Methods 124:175–179
Billimoria CP, Kraus BJ, Narayan R, Maddox RK, Sen K (2008) Invariance and sensitivity to intensity in neural discrimination of natural sounds. J Neurosci 28:6304–6308. doi:10.1523/JNEUROSCI.0961-08.2008
Borden GJ (1979) An interpretation of research of feedback interruption in speech. Brain Lang 7:307–319
Bottjer SW, Arnold AP (1984) The role of feedback from the vocal organ. I. Maintenance of stereotypical vocalizations by adult zebra finches. J Neurosci 4:2387–2396
Dave AS, Yu AC, Margoliash D (1998) Behavioral state modulation of auditory activity in a vocal motor system. Science 282:2250–2254
Di Lorenzo PM, Victor JD (2003) Taste response variability and temporal coding in the nucleus of the solitary tract of the rat. J Neurophysiol 90:1418–1431. doi:10.1152/jn.00177.2003
Doupe AJ (1997) Song- and order-selective neurons in the songbird anterior forebrain and their emergence during vocal development. J Neurosci 17:1147–1167
Doupe AJ, Konishi M (1991) Song-selective auditory circuits in the vocal control system of the zebra finch. Proc Natl Acad USA 88:11339–11343
Doupe AJ, Kuhl PK (1999) Birdsong and human speech: common themes and mechanisms. Annu Rev Neurosci 22:567–631. doi:10.1146/annurev.neuro.22.1.567
Farries MA, Ding L, Perkel DJ (2005) Evidence for “direct” and “indirect” pathways through the song system basal ganglia. J Comp Neurol 484:93–104. doi:10.1002/cne.20464
Fee MS, Mitra PP, Kleinfeld D (1996) Automatic sorting of multiple unit neuronal signals in the presence of anisotropic and non-Gaussian variability. J Neurosci Methods 69:175–188. doi:10.1016/S0165-0270(96)00050-7
Fee MS, Shraiman B, Pesaran B, Mitra PP (1998) The role of nonlinear dynamics of the syrinx in the vocalizations of a songbird. Nature 395:67–71. doi:10.1038/25725
Fehér O, Wang H, Saar S, Mitra PP, Tchernichovski O (2009) De novo establishment of wild-type song culture in the zebra finch. Nature 459:564–568. doi:10.1038/nature07994
Fukushima M, Margoliash D (2015) The effects of delayed auditory feedback revealed by bone conduction microphone in adult zebra finches. Sci Rep 5:8800. doi:10.1038/srep08800
Gentner TQ, Fenn KM, Margoliash D, Nusbaum HC (2006) Recursive syntactic pattern learning by songbirds. Nature 440:1204–1207. doi:10.1038/nature04675
Goldberg DH, Victor JD, Gardner EP, Gardner D (2009) Spike train analysis toolkit: enabling wider application of information-theoretic techniques to neurophysiology. Neuroinformatics 7:165–178. doi:10.1007/s12021-009-9049-y
Goller F, Daley MA (2001) Novel motor gestures for phonation during inspiration enhance the acoustic complexity of birdsong. Proc Biol Sci 268:2301–2305. doi:10.1098/rspb.2001.1805
Goller F, Mallinckrodt MJ, Torti SD (2004) Beak gape dynamics during song in the zebra finch. J Neurobiol 59:289–303. doi:10.1002/(ISSN)1097-4695
Hahnloser RHR, Kozhevnikov AA, Fee MS (2002) An ultra-sparse code underlies the generation of neural sequences in a songbird. Nature 419:65–70. doi:10.1038/nature00974
Huetz C, Del Negro C, Lebas N, Tarroux P, Edeline J-M (2006) Contribution of spike timing to the information transmitted by HVC neurons. Eur J Neurosci 24:1091–1108. doi:10.1111/ejn.2006.24.issue-4
Janata P, Margoliash D (1999) Gradual emergence of song selectivity in sensorimotor structures of the male zebra finch song system. J Neurosci 19:5108–5118
Jensen KK, Cooper BG, Larsen ON, Goller F (2007) Songbirds use pulse tone register in two voices to generate low-frequency sound. Proc Biol Sci 274:2703–2710. doi:10.1098/rspb.2007.0781
Konishi M (2004) The role of auditory feedback in birdsong. Ann N Y Acad Sci 1016:463–475. doi:10.1196/annals.1298.010
Lipkind D, Marcus GF, Bemis DK, Sasahara K, Jacoby N, Takahasi M, Suzuki K, Fehér O, Ravbar P, Okanoya K, Tchernichovski O (2013) Stepwise acquisition of vocal combinatorial capacity in songbirds and human infants. Nature 498:104–108. doi:10.1038/nature12173
Margoliash D (1983) Acoustic parameters underlying the responses of song-specific neurons in the white-crowned sparrow. J Neurosci 3:1039–1057
Margoliash D, Fortune ES (1992) Temporal and harmonic combination-sensitive neurons in the zebra finch’s HVc. J Neurosci 12:4309–4326
Margoliash D, Konishi M (1985) Auditory representation of autogenous song in the song system of white-crowned sparrows. Proc Natl Acad USA 82:5997–6000
Margoliash D, Schmidt MF (2010) Sleep, off-line processing, and vocal learning. Brain Lang 115:45–58. doi:10.1016/j.bandl.2009.09.005
McCasland JS, Konishi M (1981) Interaction between auditory and motor activities in an avian song control nucleus. Proc Natl Acad USA 78:7815–7819
Nagel KI, Doupe AJ (2008) Organizing principles of spectro-temporal encoding in the avian primary auditory area field L. Neuron 58:938–955. doi:10.1016/j.neuron.2008.04.028
Nealen PM, Schmidt MF (2006) Distributed and selective auditory representation of song repertoires in the avian song system. J Neurophysiol 96:3433–3447. doi:10.1152/jn.01130.2005
Nick TA, Konishi M (2005) Neural auditory selectivity develops in parallel with song. J Neurobiol 62:469–481. doi:10.1002/neu.20115
Nordeen KW, Nordeen EJ (1992) Auditory feedback is necessary for the maintenance of stereotyped song in adult zebra finches. Behav Neural Biol 57:58–66
Nottebohm F, Nottebohm ME, Crane L (1986) Developmental and seasonal changes in canary song and their relation to changes in the anatomy of song-control nuclei. Behav Neural Biol 46:445–471
O'Neill WE, Suga N (1982) Encoding of target range and its representation in the auditory cortex of the mustached bat. J Neurosci 2:17–31
Ohlemiller KK, Kanwal JS, Suga N (1996) Facilitative responses to species-specific calls in cortical FM–FM neurons of the mustached bat. NeuroReport 7:1749–1755
Okanoya K, Dooling RJ (1987) Hearing in passerine and psittacine birds: a comparative study of absolute and masked auditory thresholds. J Comp Psychol 101:7–15
Okanoya K, Yamaguchi A (1997) Adult Bengalese finches (Lonchura striata var. domestica) require real-time auditory feedback to produce normal song syntax. J Neurobiol 33:343–356
Oller DK, Eilers RE (1988) The role of audition in infant babbling. Child Dev 59:441–449
Prather JF, Peters S, Nowicki S, Mooney R (2008) Precise auditory–vocal mirroring in neurons for learned vocal communication. Nature 451:305–310. doi:10.1038/nature06492
Prather JF, Nowicki S, Anderson RC et al (2009) Neural correlates of categorical perception in learned vocal communication. Nat Neurosci 12:221–228. doi:10.1038/nn.2246
Rauske PL, Shea SD, Margoliash D (2003) State and neuronal class-dependent reconfiguration in the avian song system. J Neurophysiol 89:1688–1701. doi:10.1152/jn.00655.2002
Solis MM, Doupe AJ (1997) Anterior forebrain neurons develop selectivity by an intermediate stage of birdsong learning. J Neurosci 17:6447–6462
Suga N, O'Neill WE, Manabe T (1979) Harmonic-sensitive neurons in the auditory cortex of the mustache bat. Sci 203:270–274
Tang C, Chehayeb D, Srivastava K et al (2014) Millisecond-scale motor encoding in a cortical vocal area. PLoS Biol 12:e1002018. doi:10.1371/journal.pbio.1002018.s004
Theunissen FE, Doupe AJ (1998) Temporal and spectral sensitivity of complex auditory neurons in the nucleus HVc of male zebra finches. J Neurosci 18:3786–3802
Victor JD, Purpura KP (1996) Nature and precision of temporal coding in visual cortex: a metric-space analysis. J Neurophysiol 76:1310–1326
Victor JD, Purpura KP (1997) Metric-space analysis of spike trains: theory, algorithms and application. Network 8:127–164
Volman SF (1993) Development of neural selectivity for birdsong during vocal learning. J Neurosci 13:4737–4747
Waldstein RS (1990) Effects of postlingual deafness on speech production: implications for the role of auditory feedback. J Acoust Soc Am 88:2099–2114
Williams H, Nottebohm F (1985) Auditory responses in avian vocal motor neurons: a motor theory for song perception in birds. Science 229:279–282
Woolley SMN, Rubel EW (1997) Bengalese finches Lonchura striata domestica depend upon auditory feedback for the maintenance of adult song. J Neurosci 17:6380–6390
Woolley SMN, Rubel EW (1999) High-frequency auditory feedback is not required for adult song maintenance in Bengalese finches. J Neurosci 19:358–371
Woolley SC, Rajan R, Joshua M, Doupe AJ (2014) Emergence of context-dependent variability across a basal ganglia network. Neuron 82:208–223. doi:10.1016/j.neuron.2014.01.039
Zevin JD, Seidenberg MS, Bottjer SW (2004) Limits on reacquisition of song in adult zebra finches exposed to white noise. J Neurosci 24:5849–5862. doi:10.1523/JNEUROSCI.1891-04.2004
Acknowledgments
We thank Daniel Baleckaitis for technical assistance D.M was supported by NIH Grant MH59831.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fukushima, M., Rauske, P.L. & Margoliash, D. Temporal and rate code analysis of responses to low-frequency components in the bird’s own song by song system neurons. J Comp Physiol A 201, 1103–1114 (2015). https://doi.org/10.1007/s00359-015-1037-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-015-1037-0