Abstract
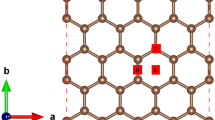
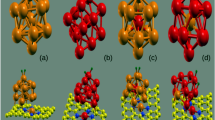
The hydrogen adsorption and desorption capacity of polycrystalline graphene sheets (PGs) with and without titanium (Ti) decoration is investigated using molecular dynamics simulations. Interatomic interactions of PGs are modeled using Tersoff potential, and the remainder of interactions are calculated via Lennard‒Jones potential. The effect of grain size and Ti concentration on the mechanical properties and hydrogen adsorption capacity of PGs is studied. The presence of grain boundaries in PGs reduces their mechanical properties, while the decoration of Ti adatoms does not significantly alter the mechanical properties of PGs. PGs showed a ~ 57% increase in the gravimetric density of H2 at 300 K and 50 bar compared to the pristine graphene sheet. At 100 bar pressure, PGs with 1% Ti concentration achieved a gravimetric density of 9.9 wt.% and 3.2 wt.% at 77 and 300 K, respectively. In Ti-decorated PGs, the desorption curve follows the same path at 300 K as the adsorption curve with increasing Ti concentration, and the desorption curve diverges from the adsorption curve after 1.5% Ti concentration at 77 K. The potential use of the isosteric enthalpy of adsorption to determine the adsorbent’s capability for adsorbing H2 molecules is also discussed.










Similar content being viewed by others
References
L. Schlapbach, A. Züttel, Hydrogen-storage materials for mobile applications. Nature 414(6861), 353–358 (2001). https://doi.org/10.1038/35104634
G.W. Crabtree, M.S. Dresselhaus, M.V. Buchanan, The hydrogen economy. Phys. Today 57(12), 39–44 (2004). https://doi.org/10.1063/1.1878333
A. Léon, Introduction. Hydrogen Technol. 1, 11–13 (2008). https://doi.org/10.1007/978-3-540-69925-5_1
L. Schlapbach, Hydrogen-fuelled vehicles. Nature 460(7257), 809–811 (2009). https://doi.org/10.1038/460809a
K. Mazloomi, C. Gomes, Hydrogen as an energy carrier: prospects and challenges. Renew. Sustain. Energy Rev. 16(5), 3024–3033 (2012). https://doi.org/10.1016/j.rser.2012.02.028
P. Jena, Materials for hydrogen storage: Past, present, and future. J. Phys. Chem. Lett. 2(3), 206–211 (2011). https://doi.org/10.1021/jz1015372
A. Léon, Hydrogen Storage. Hydrogen Technol. 3, 81–128 (2008). https://doi.org/10.1007/978-3-540-69925-5_3
J. Li, T. Furuta, H. Goto, T. Ohashi, Y. Fujiwara, S. Yip, Theoretical evaluation of hydrogen storage capacity in pure carbon nanostructures. J. Chem. Phys. 119(4), 2376–2385 (2003). https://doi.org/10.1063/1.1582831
N.A.A. Rusman, M. Dahari, A review on the current progress of metal hydrides material for solid-state hydrogen storage applications. Int. J. Hydrogen Energy 41(28), 12108–12126 (2016). https://doi.org/10.1016/j.ijhydene.2016.05.244
E. Boateng, A. Chen, Recent advances in nanomaterial-based solid-state hydrogen storage. Mater. Today Adv. 6, 100022 (2020). https://doi.org/10.1016/j.mtadv.2019.100022
K.S. Chan, M.A. Miller, X. Peng, First-principles computational study of hydrogen storage in silicon clathrates. Mater. Res. Lett. 6(1), 72–78 (2018). https://doi.org/10.1080/21663831.2017.1396261
K.S. Novoselov, A.K. Geim, S.V. Morozov, D. Jiang, Y. Zhang, S.V. Dubonos, I.V. Grigorieva, A.A. Firsov, Electric field in atomically thin carbon films. Science 306(5696), 666–669 (2004). https://doi.org/10.1126/science.1102896
M. Pumera, Graphene-based nanomaterials for energy storage. Energy Environ. Sci. 4(3), 668–674 (2011). https://doi.org/10.1039/c0ee00295j
C. Lee, X. Wei, J.W. Kysar, J. Hone, Measurement of the elastic properties and intrinsic strength of monolayer graphene. Science 321(5887), 385–388 (2008). https://doi.org/10.1126/science.1157996
Y. Zhu, S. Murali, W. Cai, X. Li, J.W. Suk, J.R. Potts, R.S. Ruoff, Graphene and graphene oxide: Synthesis, properties, and applications. Adv. Mater. 22(35), 3906–3924 (2010). https://doi.org/10.1002/adma.201001068
S. Patchkovskii, J.S. Tse, S.N. Yurchenko, L. Zhechkov, T. Heine, G. Seifert, Graphene nanostructures as tunable storage media for molecular hydrogen. Proc. Natl. Acad. Sci. U.S.A. 102(30), 10439–10444 (2005). https://doi.org/10.1073/pnas.0501030102
Y. Okamoto, Y. Miyamoto, Ab initio investigation of physisorption of molecular hydrogen on planar and curved graphenes. J. Phys. Chem. B 105(17), 3470–3474 (2001). https://doi.org/10.1021/jp003435h
T. Heine, L. Zhechkov, G. Seifert, Hydrogen storage by physisorption on nanostructured graphite platelets. Phys. Chem. Chem. Phys. 6(5), 980–984 (2004). https://doi.org/10.1039/b316209e
V. Tozzini, V. Pellegrini, Reversible hydrogen storage by controlled buckling of graphene layers. J. Phys. Chem. C 115(51), 25523–25528 (2011). https://doi.org/10.1021/jp208262r
S.I. Kundalwal, M.C. Ray, Shear lag analysis of a novel short fuzzy fiber-reinforced composite. Acta Mech. 225(9), 2621–2643 (2014). https://doi.org/10.1007/S00707-014-1095-3
S.I. Kundalwal, M.C. Ray, S.A. Meguid, Shear lag model for regularly staggered short fuzzy fiber reinforced composite. ASME J. Appl. Mech. 81(9), 091001 (2014). https://doi.org/10.1115/1.4027801
S.I. Kundalwal, M.C. Ray, Improved thermoelastic coefficients of a novel short fuzzy fiber-reinforced composite with wavy carbon nanotubes. J. Mech. Mater. Struct. 9(1), 1–25 (2014). https://doi.org/10.2140/JOMMS.2014.9.1
S.I. Kundalwal, M.C. Ray, Thermoelastic properties of a novel fuzzy fiber-reinforced composite. ASME J. Appl. Mech. 80(6), 061011 (2013). https://doi.org/10.1115/1.4023691/370366
B. Szczęśniak, J. Choma, M. Jaroniec, Gas adsorption properties of graphene-based materials. Adv. Coll. Interface. Sci. 243, 46–59 (2017). https://doi.org/10.1016/j.cis.2017.03.007
H.G. Shiraz, O. Tavakoli, Investigation of graphene-based systems for hydrogen storage. Renew. Sustain. Energy Rev. 74, 104–109 (2017). https://doi.org/10.1016/j.rser.2017.02.052
K.K. Gangu, S. Maddila, S.B. Mukkamala, S.B. Jonnalagadda, Characteristics of MOF, MWCNT and graphene containing materials for hydrogen storage: A review. J. Energy Chem. 30, 132–144 (2019). https://doi.org/10.1016/j.jechem.2018.04.012
V. Jain, B. Kandasubramanian, Functionalized graphene materials for hydrogen storage. J. Mater. Sci. 55(5), 1865–1903 (2020). https://doi.org/10.1007/s10853-019-04150-y
O.V. Yazyev, Y.P. Chen, Polycrystalline graphene and other two-dimensional materials. Nat. Nanotechnol. 9(10), 755–767 (2014). https://doi.org/10.1038/nnano.2014.166
P.Y. Huang, C.S. Ruiz-Vargas, Z.A.M. Van Der, W.S. Whitney, M.P. Levendorf, J.W. Kevek, S. Garg, J.S. Alden, C.J. Hustedt, Y. Zhu et al., Grains and grain boundaries in single-layer graphene atomic patchwork quilts. Nature 469(7330), 389–392 (2011). https://doi.org/10.1038/nature09718
M.Q. Chen, S.S. Quek, Z.D. Sha, C.H. Chiu, Q.X. Pei, Y.W. Zhang, Effects of grain size, temperature and strain rate on the mechanical properties of polycrystalline graphene - A molecular dynamics study. Carbon 85, 135–146 (2015). https://doi.org/10.1016/j.carbon.2014.12.092
A.R. Alian, S.A. Meguid, S.I. Kundalwal, Unraveling the influence of grain boundaries on the mechanical properties of polycrystalline carbon nanotubes. Carbon 125, 180–188 (2017). https://doi.org/10.1016/j.carbon.2017.09.056
M. Izadifar, R. Abadi, A. Namazian, T. Rabczuk, Investigation into the effect of doping of boron and nitrogen atoms in the mechanical properties of single-layer polycrystalline graphene. Comput. Mater. Sci. 138, 435–447 (2017). https://doi.org/10.1016/j.commatsci.2017.06.038
M. Izadifar, R. Abadi, A.H. Nezhad Shirazi, N. Alajlan, T. Rabczuk, Nanopores creation in boron and nitrogen doped polycrystalline graphene: A molecular dynamics study. Physica E 99, 24–36 (2018). https://doi.org/10.1016/j.physe.2017.12.036
Y. Guo, D.H. Seo, J. Hong, D. Su, H. Wang, J. Zheng, X. Li, A.B. Murphy, K. Ostrikov K (Ken), Controlling the adsorption behavior of hydrogen at the interface of polycrystalline CVD graphene. Int. J. Hydrogen Energy 43(41):18735–18744 (2018). https://doi.org/10.1016/j.ijhydene.2018.07.204
S. Bhattacharya, C. Majumder, G.P. Das, Ti-decorated BC4N sheet: a planar nanostructure for high-capacity hydrogen storage. J. Phys. Chem. C 113(36), 15783–15787 (2009). https://doi.org/10.1021/jp905853x
H.L. Park, S.C. Yi, Y.C. Chung, Hydrogen adsorption on Li metal in boron-substituted graphene: An ab initio approach. Int. J. Hydrogen Energy 35(8), 3583–3587 (2010). https://doi.org/10.1016/j.ijhydene.2010.01.073
F.D. Wang, F. Wang, N.N. Zhang, Y.H. Li, S.W. Tang, H. Sun, Y.F. Chang, R.S. Wang, High-capacity hydrogen storage of Na-decorated graphene with boron substitution: First-principles calculations. Chem. Phys. Lett. 555, 212–216 (2013). https://doi.org/10.1016/j.cplett.2012.11.015
S. Lee, M. Lee, H. Choi, D.S. Yoo, Y.C. Chung, Effect of nitrogen induced defects in Li dispersed graphene on hydrogen storage. Int. J. Hydrogen Energy 38(11), 4611–4617 (2013). https://doi.org/10.1016/j.ijhydene.2013.01.180
L. Zhang, S. Zhang, P. Wang, C. Liu, S. Huang, H. Tian, The effect of electric field on Ti-decorated graphyne for hydrogen storage. Comput. Theor. Chem. 1035, 68–75 (2014). https://doi.org/10.1016/j.comptc.2014.02.032
A. Lebon, J. Carrete, L. J. Gallego, A. Vega A. Ti-decorated zigzag graphene nanoribbons for hydrogen storage. A van der Waals-corrected density-functional study. Int. J. Hydrogen Energy 40(14):4960–4968 (2018). https://doi.org/10.1016/j.ijhydene.2014.12.134
L. Yuan, L. Kang, Y. Chen, D. Wang, J. Gong, C. Wang, M. Zhang, X. Wu, Hydrogen storage capacity on Ti-decorated porous graphene: First-principles investigation. Appl. Surf. Sci. 434, 843–849 (2018). https://doi.org/10.1016/j.apsusc.2017.10.231
Z. Goharibajestani, A. Yürüm, Y. Yürüm, Effect of transition metal oxide nanoparticles on gas adsorption properties of graphene nanocomposites. Appl. Surf. Sci. 475, 1070–1076 (2019). https://doi.org/10.1016/j.apsusc.2019.01.052
M. Rafique, M.A. Uqaili, N.H. Mirjat, M.A. Tunio, Y. Shuai, Ab-initio investigations on titanium (Ti) atom-doped divacancy monolayer h-BN system for hydrogen storage systems. Physica E 109, 169–178 (2019). https://doi.org/10.1016/j.physe.2019.01.015
D. Kag, N. Luhadiya, N.D. Patil, S.I. Kundalwal, Strain and defect engineering of graphene for hydrogen storage via atomistic modelling. Int. J. Hydrogen Energy 46(43), 22599–22610 (2021). https://doi.org/10.1016/j.ijhydene.2021.04.098
A. Nuhnen, C. Janiak, A practical guide to calculate the isosteric heat/enthalpy of adsorption via adsorption isotherms in metal–organic frameworks. MOFs. Dalton Trans. 49(30), 10295–10307 (2020). https://doi.org/10.1039/D0DT01784A
Y. Wei, C. Jiang, Y. Zhang, X. Li, L. Zhang, P. Wang, Y. Fang, Investigation of photocatalysis reactions on the single-crystal and polycrystalline graphenes. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 251, 119441 (2021). https://doi.org/10.1016/J.SAA.2021.119441
J.S. Roh, J.K. Jang, N. Kwon, S. Bok, Y.J. Kim, C. Jeon, H.W. Yoon, H.W. Kim, B. Lim, H.B. Park, Macroscopic properties of single-crystalline and polycrystalline graphene on soft substrate for transparent electrode applications. Carbon 178, 181–189 (2021). https://doi.org/10.1016/J.CARBON.2021.02.097
S. Plimpton, Fast parallel algorithms for short-range molecular dynamics. J. Comput. Phys. 117(1), 1–19 (1995). https://doi.org/10.1006/jcph.1995.1039
J. Tersoff, Modeling solid-state chemistry: Interatomic potentials for multicomponent systems. Phys. Rev. B 39(8), 5566–5568 (1989). https://doi.org/10.1103/PhysRevB.39.5566
S.I. Kundalwal, V.K. Choyal, N. Luhadiya, V. Choyal, Effect of carbon doping on electromechanical response of boron nitride nanosheets. Nanotechnology 31(40), 405710 (2020). https://doi.org/10.1088/1361-6528/ab9d43
S.I. Kundalwal, V.K. Choyal, V. Choyal, S.K. Nevhal, N. Luhadiya, Enhancement of piezoelectric and flexoelectric response of boron nitride sheet superlattices via interface and defect engineering. Physica E 127, 114563 (2021). https://doi.org/10.1016/j.physe.2020.114563
B. Faria, C. Guarda, N. Silvestre, J.N.C. Lopes, CNT-reinforced iron and titanium nanocomposites: Strength and deformation mechanisms. Compos. B Eng. 187, 107836 (2020). https://doi.org/10.1016/j.compositesb.2020.107836
S. Zhen, G.J. Davies, Calculation of the Lennard-Jones–m potential energy parameters for metals. Phys. Status Solidi (a) 78(2), 595–605 (1983). https://doi.org/10.1002/pssa.2210780226
R.F. Cracknell, Molecular simulation of hydrogen adsorption in graphitic nanofibres. Phys. Chem. Chem. Phys. 3(11), 2091–2097 (2001). https://doi.org/10.1039/b100144m
S. Chu, L. Hu, X. Hu, M. Yang, J. Deng, Titanium-embedded graphene as high-capacity hydrogen-storage media. Int. J. Hydrogen Energy 36(19), 12324–12328 (2011). https://doi.org/10.1016/j.ijhydene.2011.07.015
Z. Yang, Y. Huang, F. Ma, Y. Sun, K. Xu, P.K. Chu, Temperature and strain-rate effects on the deformation behaviors of nano-crystalline graphene sheets. Euro. Phys. J. B 88, 135 (2015). https://doi.org/10.1140/epjb/e2015-50850-x
N. Luhadiya, S.I. Kundalwal, S.K. Sahu, Investigation of hydrogen adsorption behavior of graphene under varied conditions using a novel energy-centered method. Carbon Lett. 31, 655–666 (2021). https://doi.org/10.1007/s42823-021-00236-3
J. Tóth, Calculation of the BET-compatible surface area from any Type I isotherms measured above the critical temperature. J. Colloid Interface Sci. 225(2), 378–383 (2000). https://doi.org/10.1006/jcis.2000.6723
T.K.A. Hoang, D.M. Antonelli, Exploiting the Kubas Interaction in the Design of Hydrogen Storage Materials. Adv. Mater. 21(18), 1787–1800 (2009). https://doi.org/10.1002/ADMA.200802832
J. Skipper C V., Ahmad Hamaed, M. Antonelli D, Nikolas Kaltsoyannis. The Kubas interaction in M( ii ) (M = Ti, V, Cr) hydrazine-based hydrogen storage materials: a DFT study. Dalton Trans. 41(28):8515–8523 (2012). https://doi.org/10.1039/C2DT30383C
Acknowledgements
This work is supported by the Department of Science and Technology (DST), Ministry of Science and Technology, Government of India. Authors SIK and SKS have received a research grant from the DST (DST/TMD/HFC/2K18/88). Author NL acknowledges the support from the Prime Minister’s Research Fellows (PMRF) Program, Ministry of Education, Government of India (PMRF-192002-780).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declared that they have no conflicts of interest in this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Luhadiya, N., Kundalwal, S.I. & Sahu, S.K. Adsorption and desorption behavior of titanium-decorated polycrystalline graphene toward hydrogen storage: a molecular dynamics study. Appl. Phys. A 128, 49 (2022). https://doi.org/10.1007/s00339-021-05194-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00339-021-05194-1