Abstract
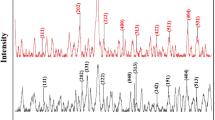
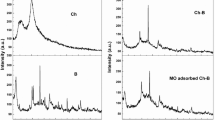
In the present work, nanocomposites of CeO2/ZnO and CeO2/SiO2 were prepared to be used as adsorbing materials for the Cr (VI) heavy metal from water. According to the non-toxic and low-cost properties of zinc oxide and silica, they have been prepared as a nanocomposite with anti-oxidant cerium oxide. The prepared CeO2/SiO2 nanocomposite showed larger removal efficiency for Cr (VI) from aqueous solution of (55%) at optimum conditions of pH = 7 after 100 min. The adsorption kinetics for Cr (VI) on surface of CeO2/Zno and CeO2/SiO2 were well fitted with pseudo-second-order kinetic model. The adsorption mechanism follows monolayer Langmuir isotherm model. The prepared samples were investigated and characterized using X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FT-IR), UV-spectroscopy, field emission scanning electron microscopy (FESEM) and N2 adsorption–desorption isotherm (BET) isotherm. The efficiency of the prepared samples for removing of Cr (VI) from aqueous solution was investigated in different experimental conditions (different PH and contact time).














Similar content being viewed by others
References
K. Bhattacharya, D. Parasar, B. Mondal et al., Mesoporous magnetic secondary nanostructures as versatile adsorbent for efficient scavenging of heavy metals. Sci. Rep. 5, 17072 (2015)
O. Olanipekun, A. Oyefusi, G.M. Neelgunf, A. Oki, Adsorption of lead over graphite oxide. Spectrochim. Acta. A Mol. Biomol. Spectrosc. 118, 857–860 (2014)
Moazeni M, Ebrahimi A, Rafiei N, Pourzamani HR (2016) Removal of lead ions from aqueous solution by nano zero-valent iron (nZVI). Heath Scope
M. Manjuladevi, S.M. Oviyaa, Heavy metal removal from industrial wastewater by nano adsorbent prepared from cucumis melopeel activated carbon. J. Nanomed. Res. 5(1), 00102 (2017)
D.K. Yadav, S. Srivastava, Carbon nanotubes as adsorbent to remove heavy metal ion (Mn+7) in wastewater treatment. Mater. Today 4, 4089–4094 (2017)
L.Z. Lee, M.A.A. Zaini, S.H. Tang, Porous Nanomaterials for Heavy Metal Removal Handbook of Ecomaterials (Springer, Cham, 2019)
A.B. Albadarin, C. Mangwandi, G.M. Walker et al., Influence of solution chemistry on Cr(VI) reduction and complexation onto date-pits/tea-waste biomaterials. J. Environ. Manage. 114, 190–201 (2013)
S. Recillas, J. Colón, E. Casals et al., Chromium VI adsorption on cerium oxide nanoparticles and morphology changes during the process. J. Hazard. Mater. 184, 425–431 (2010)
H.B. Zeng, W.P. Cai, P.S. Liu, X.X. Xu, H.J. Zhou, C. Klingshirn, H. Kalt, ZnO-based hollow nanoparticles by selective etching: Elimination and reconstruction of metal–semiconductor interface, improvement of blue emission and photocatalysis. ACS Nano 2, 1661–1670 (2008)
G.H. Chen, B.S. Fu, C.J. Cai, M.Q. Lu, Y. Yang, S.H. Yi, C. Xu, H. Li, G.S. Wang, T. Zhang, A single-center experience of transplantation for liver transplant recipients with a failing graft. Transplant. Proc. 40, 1485–1487 (2008)
Z.H. Jing, J.H. Zhan, Fabrication, Gas-sensing properties of porous ZnO nanoplates. Adv. Mater. 20, 4547–4551 (2008)
T.P. Chou, Q.F. Zhang, G.E. Fryxell, G.Z. Cao, Hierarchically structured ZnO film for dye-sensitized solar cells with enhanced energy conversion efficiency. Adv. Mater. 19, 2588–2592 (2007)
X.B. Wang, W.P. Cai, Y.X. Lin, G.Z. Wang, C.H. Liang, Mass production of micro/nanostructured porous ZnO plates and their strong structurally enhanced and selective adsorption performance for environmental remediation. J. Mater. Chem. 20, 8582–8590 (2010)
J.H. Lee, B.S. Kim, J.C. Lee, S. Park, Removal of Cu++ ions from aqueous Cu-EDTA solution using ZnO nanopowder, in Eco-Materials Processing & Design Vi. ed. by H.S. Kim, S.-Y. Park, B.Y. Hur, S.W. Lee (Trans Tech Publications, Korea, 2005), pp. 510–513
M. Hua, S. Zhang, B. Pan, W. Zhang, L. Lv, Q. Zhang, Heavy metal removal from water/wastewater by nanosized metal oxides: A review. J. Hazard. Mater. 211–212, 317–331 (2012)
S. Bernal, J.J. Calvino, M.A. Cauqui, J.M. Gatica, C. Larese, J.A.P. Omil, J.M. Pintado, Some recent results on metal/support interaction effects in NM/CeO2 (NM: noble metal) catalysts. Catal. Today 50, 175–206 (1999)
H. Siddiqui, K.B.M. Ahmed, F. Sami, S. Hayat, Silicon Nanoparticles and Plants: Current Knowledge and Future Perspectives (Springer, Cham, 2020)
H. Rogalla, V. Römheld, Role of leaf apoplast in silicon-mediated manganese tolerance of Cucumis sativus L. Plant Cell Environ. 25, 549–555 (2002)
K.P.V. Da Cunha, C.W.A. do Nascimento, Silicon effects on metal tolerance and structural changes in maize (Zea mays L.) grown on a cadmium and zinc enriched soil Water. Air, Soil Pollut. 197(1–4), 323–330 (2009)
A. Hatamie, A. Khan, M. Golabi et al., Zinc oxide nanostructure-modified textile and its application to biosensing, photocatalysis, and as antibacterial material. Langmuir 31(39), 10913–10921 (2015)
S. Ojha, M. Roy, A. Chamuah, K. Bhattacharya, S. Bhattacharya, Electrical transport of chalcogenide glassy system: interpretation by Hunt’s model and microstructure. SN Appl. Sci. 2, 838 (2020). https://doi.org/10.1007/s42452-020-2518-5
S. Bhattacharya, Phys. Lett. A 384, 12634 (2020)
S. Ojha, M. Roy, A. Chamuah, K. Bhattacharyad, S. Bhattacharya, Phys. Chem. Chem. Phys. 22(42), 24600–24613 (2020)
A. Acharya, K. Bhattacharya, C.K. Ghosh, A.N. Biswas, S. Bhattacharya, Mater. Sci. Eng., B 260, 114612 (2020). https://doi.org/10.1016/j.mseb.2020.114612
R.M. El-Sharkawy, E.A. Allam, M.E. Mahmoud, Environ. Nanotechnol. Monitor. Manag 14, 100367 (2020)
E.J.S. Christy, R. Alagar, M. Dhanu, A. Pius, Environ. Nanotechnol. Monitor. Manag. 14, 100365 (2020)
S. Sobhanardakania, A. Jafari, R. Zandipak, A. Meidanchi, Process Saf. Environ. Prot. 120, 348–357 (2018)
I.A. Salem, M.A. Salem, M.A. El-Ghobashy, J. Mol. Liq. 248, 527–538 (2017)
M. Gu, L. Hao, Y. Wang, X. Li, Y. Chen, W. Lia, L. Liang, Chem. Phys. 534, 110750 (2020)
M. Dinari, A. Haghigh, Ultra. – Sonochem. 41, 75–84 (2018)
M.K. Aroua, F.M. Zuki, N.M. Sulaiman, J. Hazard. Mater. 147, 752–758 (2007)
T.D.C. Dantas, A.D. Neto, M.D.A. Moura, E.L.B. Neto, E.D. Paiva Telemaco, Removal of chromium from aqueous solutions by diatomite treated with microemulsion. Langmuir 35(9), 2219–2224 (2001)
R. Ramadan, M.K. Ahmed, V. Uskoković, J. Alloys Compounds. (2021). https://doi.org/10.1016/j.jallcom.2020.157013
M.K. Ahmed, S.F. Mansour, R. Ramadan, M. Afifi, S. Mostaf, S.I. El-dek, V. Uskoković, J. Water Process Eng. 34, 101090 (2020)
M.K. Ahmed, R. Ramadan, M. Afifi, A.A. Menaze, J. Mater. Res. Technol. 9(4), 8854–8866 (2020)
D.H.K. Reddy, Y.S. Yun, Coord. Chem. Rev. 315, 90–111 (2016)
Q. Zhang, J. Li, Q. Lina, C. Fang, Environ. Res. 188, 109698 (2020)
J. Wu, J. Wang, Y. Du, H. Li, Y. Yang, X. Jia, Appl. Catal. B 174–175, 435–444 (2015)
M. Gua, L. Haoa, Y. Wang, X. Lia, Y. Chen, W. Lia, L. Jiang, Chem. Phys. 534, 110750 (2020)
M.K. Ahmed, R. Ramadan, S.I. El-dek, V. Uskokovi, J. Alloy. Compd. 801, 70–81 (2019)
A.S. Khan, A.N. Hussain, L. Sidra, Z. Sarfraz, H. Khalid, M. Khan, F. Manzoor, L. Shahzadi, M. Yar, I.U. Rehman, Mater. Sci. Eng. C 80, 387–396 (2017)
G. Wu, X. Deng, J. Song, F. Chen, J. Photochem. Photobiol. B Biol. 178, 27–32 (2018)
W. Peng, H. Li, Y. Liu, S. Song, J. Molecul. Liquids 230, 496–504 (2017)
T.S. Badessa, E. Wakuma, A.M. Yimer et al., BMC Chem 14, 71 (2020)
A. Alemuab, B. Lemma, N. Gabbiye, M.T. Alula, M.T. Desta, Heliyon 4, e00682 (2018)
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ramadan, R., El-Masry, M.M. Comparative study between CeO2/Zno and CeO2/SiO2 nanocomposites for (Cr6+) heavy metal removal. Appl. Phys. A 127, 876 (2021). https://doi.org/10.1007/s00339-021-05037-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00339-021-05037-z