Abstract
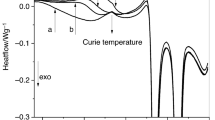
The structural and crystallization kinetics of (As50Se50)100−xAgx (x = 0, 5, 10 and 15 at.%) glasses are reported. The glass transition activation energy, Et, decreased from 185.19 to 179.94 eV with increasing the Ag content. The crystalline phases AsSe, AgAsSe2 and Ag2Se phases were found in the annealed glasses. The structure of the annealed samples at different temperatures was examined using scanning electron microscopy. The crystallization kinetics parameters were calculated using the iso-conversional models. A slight increase in Ec(χ) from with the conversion (χ), which accounts for single-step mechanism, was found. The results of the conversion dependence of the Avrami exponent n(χ) showed an increase from 1.22 to 1.31 with the Ag content and conversion (χ) as well. The average values of n(χ) are accounted for two- and three-dimensional crystal growth with heterogeneous nucleation. Comparing the experimental DSC data with calculated ones indicated that Sestak–Berggren model was found to be suitable for describing the crystallization process of the investigated chalcogenide glasses.















Similar content being viewed by others
References
Z. Mahmoud, M. Mohamed, S. Moustafa, A.M. Abdelraheem, M.A. Abdel-Rahim, Study of non-isothermal crystallization kinetics of Ge20Se70Sn10 chalcogenide glass. J. Therm. Anal. Calorimet. 131, 2433 (2018)
J. Zheng, H. Yin, L. Li, Y. Wang, J. Wei, G. Chen, Effects of Ag additive on structure and crystallization behaviors of As2(Se15Te85)3 glasses. Ceram. Int. 43, 15027–15033 (2017)
Y. Liu, C. Chen, Y. Zhou, R. Kondrotas, J. Tang, Butyldithiocarbamate acid solution processing: its fundamentals and applications in chalcogenide thin film solar cells. J. Mater. Chem. C 7, 11068–11084 (2019)
E. Shaaban, Non-isothermal crystallization kinetic studies on a ternary, Sb0.14 As0.38 Se0.48 chalcogenide semi-conducting glass. Phys. B Condens. Matter 373, 211–216 (2006)
A. Inoue, A. Takeuchi, Recent progress in bulk glassy, nanoquasicrystalline and nanocrystalline alloys. Mater. Sci. Eng. A 375–377, 16–30 (2004)
J.L. Cárdenas-Leal, J. Vázquez, D. García-G, R. Barreda, P.L. González-Palma, P.V. López-Alemany, Analysis of the glass–crystal transformation kinetics by means of the theoretical method developed (TMD) under both non-isothermal and isothermal regimes. Application to the crystallization of the Ag0.16As0.34Se0.50 glassy alloy. J. Alloys Compd. 622, 610–617 (2015)
N. Nedelcu, Applications of the chalcogeide ternary thin films. Roman. J. Mech. 4, 47–64 (2019)
D. Zhao, X.H. Zhang, F. Xia, H. Wang, H.L. Ma, J.L. Adam, G. Chen, Nonisothermal study on crystallization kinetics of GeSe2–As2Se3–CdSe chalcogenide glasses by differential scanning calorimeter. J. Cryst. Growth 285, 228–235 (2005)
A.A. Soliman, Thermal stability of Cu0.3(SSe20)0.7 chalcogenide glass by differential scanning calorimetry. Thermochim. Acta 423, 71–76 (2004)
M.I. Abd-Elrahman, A.Y. Abdel-Latief, R.M. Khafagy, N. Younis, M.M. Hafiz, Thermal annealing effect on the optical properties of Ag10As30S60 thin film. Spectrochim Acta Part A Mol. Biomol. Spectrosc. 137, 29–32 (2015)
M. Frumar, T. Wagner, Ag doped chalcogenide glasses and their applications. Curr. Opin. Solid State Mater. Sci. 7, 117–126 (2003)
F. Wang, W.P. Dunn, M. Jain, C. De Leo, N. Vicker, R. Savage, X. Jin, S. Mamedov, P. Boolchand, The effects of thermal annealing on the obliquely deposited Ag–Ge–S thin films. J. Phys. Chem. Solids 70, 978–981 (2009)
M.A. Abdel-Rahim, M.A.S. Hammam, A.A. Abu-Sehly, M.M. Hafiz, Composition effect on the pre-crystallization and crystallization characteristics for Se90−xTe10Agx. J. Alloys Compd. 728, 1346–1361 (2017)
S.R. Elliott, Physics of Amorphous Materials (Longman Group Limited, London, 1984)
A. Madan, M.P. Shaw, The Physics and Applications of Amorphous Semiconductors (Academic Press Inc., London, 1988)
N. Abd-el Salam, E.R. Mansour Mohamed, M.A. Shaaban, A.-L. Abdel-Rahim, The crystallization kinetics studies of the two crystallization stages of As37.5Se37.5Ag25 glass using the model-fitting and model-free approaches. Chin. J. Phys. 60, 35–47 (2019)
M.J. Starink, Analysis of aluminium based alloys by calorimetry: quantitative analysis of reactions and reaction kinetics. Int. Mater. Rev. 49, 191–226 (2004)
A.A. Joraid, Estimating the activation energy for the non-isothermal crystallization of an amorphous Sb9.1Te20.1Se70.8 alloy. Thermochim. Acta 456, 1–6 (2007)
L. Liu, F.W. Zhi, L. Chen, A kinetic study of the non-isothermal crystallization of a Zr-based bulk metallic glass. Phys. Lett. 19, 1483–1486 (2002)
S. Vyazovkin, C.A. Wight, Isothermal and non isothermal reaction kinetics in solids. J. Phys. Chem. A 101, 8279–8284 (1997)
S. Vyazovkin, C.A. Wight, Model-free and model-fitting approaches to kinetic analysis of isothermal and nonisothermal data. Thermochim. Acta 340–341, 53–68 (1999)
S. Vyazovkin, Advanced isoconversional method. J. Therm. Anal. 49, 1493–1499 (1997)
B.S. Patial, N. Thakur, S.K. Tripathi, On the crystallization kinetics of In additive Se–Te chalcogenide glasses. Thermochim. Acta 513, 1–8 (2011)
M.A. Abdel-Rahim, M.M. Hafiz, A.Z. Mahmoud, Crystallization kinetics of overlapping phases in Se70Te15Sb15 using isoconversional methods. Progr. Nat. Sci. Mater. Int. 25, 169–177 (2015)
S. Vyazovkin, A.K. Burnham, J.M. Criado, Perez-Maqueda LA, Popescu CSN. ICTAC kinetics committee recommendations for performing kinetic computations on thermal analysis data, Thermochim. Acta. 520, 1–19 (2011)
J. Šesták, Thermal analysis: their measurements and theoretical thermal analysis. Part D Thermophys. Prop. Solids 12, 172–259 (1984)
P. Šimon, Isoconversional methods. J. Therm. Anal. Calorim. 76, 123–132 (2004)
P. Šimon, Single-step kinetics approximation employing non-Arrhenius temperature functions. J. Therm. Anal. Calorim. 79, 703–708 (2005)
J. Malek, Kinetic analysis of non-isothermal calorimetric data. Sci. Pap. Univ. Pardubice 2, 177–209 (1996)
J. Sestak, G. Berggren, Study of the kinetics of the mechanism of solid-state reactions at increasing temperatures. Thermochim. Acta 3, 1–12 (1971)
M.J. Starink, Activation energy determination for linear heating experiments: deviations due to neglecting the low temperature end of the temperature integral. J. Mater. Sci. 42, 483–489 (2007)
J.H. Flynn, L.A. Wall, Adirect method for the determination of activation energy from thermo-gravimetric data. Polym. Lett. 4, 323–328 (1966)
S. Vyazovkin, N. Sbirrazzuoli, Estimating the activation energy for non-isothermal crystallization of polymer melts. J. Therm. Anal. Calorim. 72, 681–686 (2003)
S. Vyazovkin, N. Sbirrazzuoli, Isoconversional approach to evaluating the Hoffman–Lauritzen parameters (U* and Kg) from the overall rates of nonisothermal crystallization. Macromol. Rapid Commun. 25, 733 (2004)
A. Khawam, D.R. Flanagan, Role of isoconversional methods in varying activation energies of solid-state kinetics: II. Nonisothermal kinetic studies. Thermochim. Acta 436, 101–112 (2005)
S. Vyazovkin, Modification of the integral isoconversional method to account for variation in the activation energy. J. Comput. Chem. 22, 178–183 (2001)
T. Ozawa, A new method of analyzing thermo-gravimetric data. Bull. Chem. Soc. Jp. 38, 188–1886 (1965)
F. Liu, S. Song, J. Xu, J. Wang, Determination of nucleation and growth modes from evaluation of transformed fraction in solid-state transformation. Acta Mater. 56, 6003–6012 (2008)
H.E. Kissinger, Variation of peak temperature with heating rate in differential thermal analysis. J. Res. Natl. Bur. Stand. 57, 217–221 (1956)
H.L. Friedman, Kinetics of thermal degradation of char-forming plastics from thermogravimetry. Appl. Phenol. Plast. J. Polym. Sci. C 6, 183–195 (1964)
M. Imran, N. Saxena, D. Bhandari, M. Husain, Glass transition phenomena, crystallization kinetics and enthalpy released in binary Se100–xInx (x = 2, 4 and 10) semiconducting glasses. Phys. Status Solidi 181, 357–368 (2000)
M.M.A. Imran, D. Bhandari, N.S. Saxena, Glass transition phenomena, crystallization kinetics and thermodynamic properties of ternary Se80Te20−xInx (x = 2, 4, 6, 8 and 10) semiconducting glasses: theoretical and experimental aspects. Mater. Sci. Eng. A 292, 56–65 (2000)
M.F. Kotkata, E.A. Mahmoud, M.K. El-Mously, An X-ray study of the Se−Te system. Acta Phys. Acad. Sci. Hung. 52, 175–187 (1982)
Y.E.A. Voroshilov, Method for orientation of uniaxial nonlinear single crystals. Sov. J. Quant. Electron. 6(3), 326–402 (1976)
U. Wiegers, H. Hilz, A new method using ‘proteinase K’ to prevent mRNA degradation during isolation from HeLa cells. Biochem. Biophys. Res. Commun. 44, 513–519 (1971)
A.J.C. Wilson, Mathematical Theory of X-ray Powder Diffractometry (Centrex Publishing Company, Boston, 1963)
M. Dhanam, R. Balasundaraprabhu, S. Jayakumar, P. Gopalakrishnan, M. Kannan, Preparation and study of structural and optical properties of chemical bath deposited copper indium diselenide thin films. Phys. Stat. Solidi 191, 149–160 (2002)
S. Venkatachalam, D. Mangalaraj, S.K. Narayandass, Characterization of vacuum-evaporated ZnSe thin films. Phys. B 393, 47–55 (2007)
M.I. Abd-Elrahman, R.M. Khafagy, N. Younis, M.M. Hafiz, Structural and calorimetric studies of two crystallization stages of Ag10As30S60 glassy alloys. Phys. B 449, 155–159 (2014)
K. Ogusu, T. Kumagai, Y. Fujimori, M. Kitao, Thermal analysis and Raman scattering study on crystallization and structure of Agx (As04Se06)100–x glasses. J. Non-cryst. Solids 324, 118–126 (2003)
M. Abdel-Rahim, A. Abdel-Latief, M.N. Abd-el Salam, Kinetic analysis of crystallization process of Se–In–Pb glasses—isoconversion method. Thermochim. Acta 573, 57–64 (2013)
M. Lasocka, The effect of scanning rate on glass transition temperature of splatcooled Te85Ge15. Mater. Sci. Eng. 23, 173–177 (1976)
K. He, Variation of peak temperature with heating rate in differential thermal analysis. J Res Natl Bur Stand 57(217), 221 (1956)
N.H. March, R.A. Street, M. Tosi, Amorphous Solids and Liquid State (Plenum, New York, 1985), p. 434
J.M. Cai, L.S. Bi, Kinetic analysis of wheat straw pyrolysis using isoconversional methods. J Therm Anal Calorim 98, 325–330 (2009)
J. Málek, E. C̆ernošková, R. Svejka, J. Sestak, G. Van der Plaats, Crystallization kinetics of Ge03Sb14S27 glass. Thermochim. Acta 280, 353–361 (1996)
S. Vyazovkin, W. Linert, Kinetic analysis of reversible thermal decomposition of solids. Int. J. Chem. Kinet. 27, 73–84 (1995)
J. Malek, The applicability of Johnson–Mehl–Avrami model in the thermal analysis of the crystallization kinetics of glasses. Thermochim. Acta. 267, 61–73 (1995)
W. Lu, B. Yan, W.-H. Huang, Complex primary crystallization kinetics of amorphous Finemet alloy. J. Non-Cryst. Solids 351, 3320–3324 (2005)
K. Tanka, Structural phase transitions in chalcogenide glasses. Phys. Rev. B. 39, 1270–1279 (1989)
P. Duhan, D. Baranock, A. Ondrejka, The study of transformation kinetics of the amorphous Pd–Si alloys. J. Non-Cryst. Solids. 2, 1411–1428 (1976)
J. Wong, H.C. Kou, J.S. Li, X.F. Cu, L.Q. Xing, L. Zhou, Determination of kinetic parameters during isochronal crystallization of Ti40Zr25Ni8Cu9Be18 metallic glass. J. Alloys Compd. 479, 835–893 (2009)
D.W. Henderson, Thermal analysis of non-isothermal crystallization kinetics in glass forming liquids. J. Non-Cryst. Solids 30, 301–315 (1979)
A.A. Joraid, Limitation of the Johnson–Mehl–Avrami (JMA) formula for kinetic analysis of the crystallization of a chalcogenide glass. Thermochim. Acta 436, 78–82 (2005)
M.A.A. Rahim, A.Y.A. Latief, A. El-Korashy, M.A. Sabet, Kinetic analysis of crystallization process in amorphous Se90−xTe10Pbx glasses. Mater. Trans. 51, 428–433 (2010)
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mohamed, M., Moustafa, S. Morphology, structure and thermal analysis of (As50Se50)100−xAgx glasses. Appl. Phys. A 126, 270 (2020). https://doi.org/10.1007/s00339-020-3457-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00339-020-3457-0