Abstract
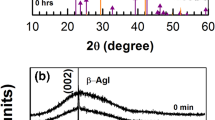
Commercial silver foils—typically 10 µ thick—were subjected to a regulated flux of iodine (I2) vapors produced in a “figure of eight” iodinator. The effect of iodization time, varied in steps from 0 to 720 s, on the microstructure, structure and optical response is reported. The uniodized Ag foil shows large clusters which transform into sub 100 nm sizes on exposure to iodine vapors up to a duration of 540 s, beyond which there is re-growth of large clusters. Crystallization of AgI is not observed up to an iodine exposure duration of 720 s. The foils continue to exhibit diffraction peaks corresponding to crystalline Ag up to this duration of iodine exposure. In the first 180 s, there is very little difference between the optical absorption spectra of the exposed and unexposed foils. At an exposure time of 360 s, a localized surface plasmon peak of Ag appears at a wavelength of 550 nm. The intensity of the plasmon peak increases with further increase in exposure time to 540 and 720 s. Interestingly, the intensity of the plasmon peak and the optical absorption value at 700 nm increase linearly beyond an exposure time of 180 s. The process was monitored gravimetrically to obtain a mass loss of I2 versus iodination time profile. This profile is apparently linear for iodination times in the range 180–720 s, suggesting a linear uptake of I2 by silver foil. This feature together with the gravimetry result makes the process applicable as detectors in situations where ppm levels of I2 need to be detected.






Similar content being viewed by others
References
J. Zhang, X. Li, K. Liu, Z. Cui, G. Zhang, B. Zhao, B. Yang, J. Colloid Interface Sci. 255, 115 (2002)
X.F. Liang, Y. Chen, L. Shi, J. Lin, J. Yin, Z.G. Liu, J. Phys. D Appl. Phys. 40, 4767 (2007)
C. Yang, Y.T. Xie, M.M. Yuen, X. Xiong, C.P. Wong, Phys. Chem. Chem. Phys. 12, 14459 (2010)
C. Yang, Y.T. Xie, M.M. Yuen, B. Xu, B. Gao, X. Xiong, C.P. Wong, Adv. Funct. Mater. 20, 2580 (2010)
D.Y. Cho, S. Tappertzhofen, R. Waser, I. Valov, Sci. Rep. 3, 1169 (2013)
P. Senthil Kumar, C.S. Sunandana, Thin Solid Films 323, 110 (1998)
P. Senthil Kumar, P. Babu Dayal, C.S. Sunandana, Thin Solid Films 357, 111 (1999)
D. BharathiMohan, C.S. Sunandana, J. Appl. Phys. 100, 64314 (2006)
D. Rajesh, C.S. Sunandana, Thin Solid Films 524, 316 (2012)
D. Rajesh, C.S. Sunandana, Appl. Surf. Sci. 259, 276 (2012)
M. Gnanavel, C.S. Sunandana, Physics of Iodized Silver and Silver–Copper Thin Films Nanostructures (Lambert, Germany, 2013)
D. BharathiMohan, K. Sreejith, C.S. Sunandana, Appl. Phys. B 89, 59 (2007)
R.C. Prost, Anal. Chem. 49, 1199 (1977)
S.K. Bose, S.C. Sircar, Metall. Trans. 5, 2015 (1974)
M. Bahadir, J. Fleckenstein, E. Schnug, Commun. Soil Sci. Plant Anal. 27, 741 (1996)
X. Han, L. Cao, H. Cheng, J. Liu, Z. Xu, Anal. Methods 4, 3471 (2012)
C.P. Shelor, P.K. Dasgupta, Anal. Chim. Acta 702, 16 (2011)
C. Kiferle, S. Gonzali, H.T. Holwerda, R. RealIbaceta, P. Perata, Front. Plant Sci. 4, 205 (2013). doi:10.3389/fpls.2013.00205
Acknowledgments
The authors acknowledge the School of Physics, University of Hyderabad for providing experimental facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Desapogu, R., Krishna, M.G. & Sunandana, C.S. Briefly iodized Ag foils: microstructure, structure, optical response and potential as iodine detectors. Appl. Phys. A 119, 1311–1316 (2015). https://doi.org/10.1007/s00339-015-9097-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00339-015-9097-0