Abstract
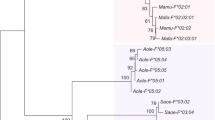
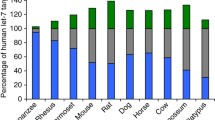
In primates, the unigenic growth hormone (GH) locus of prosimians expressed primarily in the anterior pituitary, evolved by gene duplications, independently in New World Monkeys (NWM) and Old World Monkeys (OWMs)/apes, to give complex clusters of genes expressed in the pituitary and placenta. In human and chimpanzee, the GH locus comprises five genes, GH-N being expressed as pituitary GH, whereas GH-V (placental GH) and CSHs (chorionic somatomammotropins) are expressed (in human and probably chimpanzee) in the placenta; the CSHs comprise CSH-A, CSH-B and the aberrant CSH-L (possibly a pseudogene) in human, and CSH-A1, CSH-A2 and CSH-B in chimpanzee. Here, the GH locus in two additional great apes, gorilla (Gorilla gorilla gorilla) and orangutan (Pongo abelii), is shown to contain six and four GH-like genes, respectively. The gorilla locus possesses six potentially expressed genes, gGH-N, gGH-V and four gCSHs, whereas the orangutan locus has just three functional genes, oGH-N, oGH-V and oCSH-B, plus a pseudogene, oCSH-L. Analysis of regulatory sequences, including promoter, enhancer and P-elements, shows significant variation; in particular the proximal Pit-1 element of GH-V genes differs markedly from that of other genes in the cluster. Phylogenetic analysis shows that the initial gene duplication led to distinct GH-like and CSH-like genes and that a second duplication provided separate GH-N and GH-V. However, evolution of the CSH-like genes remains unclear. Rapid adaptive evolution gave rise to the distinct CSHs, after the first duplication, and to GH-V after the second duplication. Analysis of transcriptomic databases derived from gorilla tissues establishes that the gGH-N, gGH-V and several gCSH genes are expressed, but the significance of the many CSH genes in gorilla remains unclear.







Similar content being viewed by others
References
Adkins RM, Nekrutenko A, Li WH (2001) Bushbaby growth hormone is much more similar to nonprimate growth hormones than to rhesus monkey and human growth hormones. Mol Biol Evol 18:55–60
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Barrera-Saldaña HA (1998) Growth hormone and placental lactogen: biology, medicine and biotechnology. Gene 211:11–18
Barrera-Saldaña HA, Seeburg PH, Saunders GF (1983) Two structurally different genes produce the same secreted human placental lactogen hormone. J Biol Chem 258:3787–3793
Brawand D, Soumillon M, Necsulea A, Julien P, Csárdi G, Harrigan P, Weier M, Liechti A, Aximu-Petri A, Kircher M, Albert FW, Zeller U, Khaitovich P, Grützner F, Bergmann S, Nielsen R, Pääbo S, Kaessmann H (2011) The evolution of gene expression levels in mammalian organs. Nature 478:343–348
Chen EY, Liao YC, Smith DH, Barrera-Saldaña HA, Gelinas RE, Seeburg PH (1989) The human growth hormone locus: nucleotide sequence, biology, and evolution. Genomics 4:479–497
De Vos AM, Ultsch M, Kossiakoff AA (1992) Human growth hormone and extracellular domain of its receptor: crystal structure of the complex. Science 255:306–312
Eberhardt NL, Jiang SW, Shepard AR, Arnold AM, Trujillo MA (1996) Hormonal and cell-specific regulation of the human growth hormone and chorionic somatomammotropin genes. Prog Nucleic Acid Res Mol Biol 54:127–163
Elefant F, Su Y, Liebhaber SA, Cooke NE (2000) Patterns of histone acetylation suggest dual pathways for gene activation by a bifunctional locus control region. EMBO J 19:6814–6822
Emera D, Wagner GP (2012) Transposable element recruitments in the mammalian placenta: impacts and mechanisms. Brief Funct Genomics 11:267–276
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791
Glass CK, Franco R, Weinberger C, Albert VR, Evans RM, Rosenfeld MG (1987) A c-erb-A binding-site in rat growth hormone gene mediates trans-activation by thyroid-hormone. Nature 329:738–741
González Alvarez R, Revol de Mendoza A, Esquivel Escobedo D, Corrales Félix G, Rodríguez Sánchez I, González V, Dávila G, Cao Q, de Jong P, Fu YX, Barrera Saldaña HA (2006) Growth hormone locus expands and diverges after the separation of New and Old World monkeys. Gene 380:38–45
Harper ME, Barrera-Saldaña HA, Saunders GF (1982) Chromosomal localization of the human placental lactogen-growth hormone gene cluster to 17q22-24. Am J Hum Genet 34:227–234
Higgins DG, Sharp PM (1988) CLUSTAL: a package for performing multiple sequence alignment on a microcomputer. Gene 73:237–244
Ho Y, Liebhaber SA, Cooke NE (2004) Activation of the human GH gene cluster: roles for targeted chromatin modification. Trends Endocrinol Metab 15:40–45
Jacquemin P, Oury C, Peers B, Morin A, Belayew A, Martial JA (1994) Characterization of a single strong tissue-specific enhancer downstream from the three human genes encoding placental lactogen. Mol Cell Biol 14:93–103
Jacquemin P, Alsat E, Oury C, Belayew A, Muller M, Evain-Brion D, Martial JA (1996) The enhancers of the human placental lactogen B, A, and L genes: progressive activation during in vitro trophoblast differentiation and importance of the DF-3 element in determining their respective activities. DNA Cell Biol 15:845–854
Jiang SW, Shepard AR, Eberhardt NL (1995) An initiator element is required for maximal human chorionic somatomammotropin gene promoter and enhancer function. J Biol Chem 270:3683–3692
Kent WJ (2002) BLAT—The BLAST-Like alignment tool. Genome Res 12:656–664
Leidig F, Shepard AR, Zhang W, Stelter A, Cattini PA, Baxter JD, Eberhardt NL (1992) Thyroid hormone responsiveness in human growth hormone-related genes. Possible correlation with receptor-induced DNA conformational changes. J Biol Chem 267:913–921
Lemaigre FP, Courtois SJ, Lafontaine DA, Rousseau GG (1989) Evidence that the upstream stimulatory factor and the Sp1 transcription factor bind in vitro to the promoter of the human-growth-hormone gene. Eur J Biochem 181:555–561
Locke DP et al (2011) Comparative and demographic analysis of orang-utan genomes. Nature 469:529–533
Lynch VJ, Leclerc RD, May G, Wagner GP (2011) Transposon-mediated rewiring of gene regulatory networks contributed to the evolution of pregnancy in mammals. Nat Genet 11:1154–1159
Lynch VJ, Nnamani MC, Kapusta A, Brayer K, Plaza SL, Mazur EC, Emera D, Sheikh SZ, Grutzner F, Bauersachs S, Graf A, Young SL, Lieb JD, DeMayo FJ, Feschotte C, Wagner GP (2015) Ancient transposable elements transformed the uterine regulatory landscape and transcriptome during the evolution of mammalian pregnancy. Cell Rep 10:1–11
Lytras A, Surabhi RM, Zhang JF, Jin Y, Cattini PA (1996) ‘Repair’ of the chorionic somatomammotropin-A ‘enhancer’ region reveals a novel functional element in the chorionic somatomammotropin-B enhancer. Mol Cell Endocrinol 119:1–10
Misra-Press A, Cooke NE, Liebhaber SA (1994) Complex alternative splicing partially inactivates the human chorionic somatomammotropin-like (hCS-L) gene. J Biol Chem 269:23220–23229
Nachtigal MW, Nickel BE, Cattini PA (1993) Pituitary-specific repression of placental members of the human growth hormone gene family. A possible mechanism for locus regulation. J Biol Chem 268:8473–8479
Necsulea A, Soumillon M, Warnefors M, Liechti A, Daish T, Zeller U, Baker JC, Grützner F, Kaessmann H (2014) The evolution of lncRNA repertoires and expression patterns in tetrapods. Nature 505:635–640
Norquay LD, Yang X, Sheppard P, Gregoire S, Dodd JG, Reith W, Cattini PA (2003) RFX1 and NF-1 associate with P sequences of the human growth hormone locus in pituitary chromatin. Mol Endocrinol 17:1027–1038
Norquay LD, Yang X, Jin Y, Detillieux KA, Cattini PA (2006) Hepatocyte nuclear factor-3α binding at P sequences of the human growth hormone locus is associated with pituitary repressor function. Mol Endocrinol 20:598–607
Pérez-Ibave DC, Rodríguez-Sánchez IP, Garza-Rodríguez ML, Barrera-Saldaña HA (2014) Extrapituitary growth hormone synthesis in humans. Growth Horm IGF Res 24:47–53
Pérez-Maya AA, Rodríguez-Sánchez IP, de Jong P, Wallis M, Barrera-Saldaña HA (2012) The chimpanzee GH locus: composition, organization, and evolution. Mamm Genome 23:387–398
Pipes L, Li S, Bozinoski M, Palermo R, Peng X, Blood P, Kelly S, Weiss JM, Thierry-Mieg J, Thierry-Mieg D, Zumbo P, Chen R, Schroth GP, Mason CE, Katze MG (2013) The non-human primate reference transcriptome resource (NHPRTR) for comparative functional genomics. Nucl Acids Res 41:D906–D914
Reséndez-Pérez D, Ramirez-Solís R, Varela-Echavarría A, Martínez-Rodríguez HG, Barrera-Saldaña HA (1990) Coding potential of transfected human placental lactogen genes. Nucl Acids Res 18:4665–4670
Rodríguez-Sánchez IP, Tejero ME, Cole SA, Comuzzie AG, Nathanielsz PW, Wallis M, Barrera-Saldaña HA (2010) Growth hormone-related genes from baboon (Papio hamadryas): characterization, placental expression and evolutionary aspects. Gene 450:1–7
Sawyer SA (1999) GENECONV: a computer package for the statistical detection of gene conversion. Distributed by the author, Department of Mathematics, Washington University, St. Louis, MO. Available at http://www.math.wustl.edu/_sawyer
Scally A et al (2012) Insights into hominid evolution from the gorilla genome sequence. Nature 483:169–175
Slater EP, Rabenau O, Karin M, Baxter JD, Beato M (1985) Glucocorticoid receptor binding and activation of a heterologous promoter by dexamethasone by the first intron of the human growth hormone gene. Mol Cell Biol 5:2984–2992
Smit AFA, Hubley R, Green P (1996-2010) RepeatMasker Open-3.0. Available at http://www.repeatmasker.org
Su Y, Liebhaber SA, Cooke NE (2000) The human growth hormone gene cluster locus control region supports position-independent pituitary- and placenta-specific expression in the transgenic mouse. J Biol Chem 275:7902–7909
Swofford DL (1998) PAUP*. Phylogenetic analysis using parsimony (* and other methods), ver. 4. Sinauer, Sunderland
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739
Walker WH, Fitzpatrick SL, Barrera-Saldaña HA, Reséndez-Perez D, Saunders GF (1991) The human placental lactogen genes: structure, function, evolution and transcriptional regulation. Endocrine Rev. 12:316–328
Wallis M (2008) Mammalian genome projects reveal new growth hormone (GH) sequences. Characterization of the GH-encoding genes of armadillo (Dasypus novemcinctus), hedgehog (Erinaceus europaeus), bat (Myotis lucifugus), hyrax (Procavia capensis), shrew (Sorex araneus), ground squirrel (Spermophilus tridecemlineatus), elephant (Loxodonta africana), cat (Felis catus) and opossum (Monodelphis domestica). Gen Comp Endocrinol 155:271–279
Wallis OC, Wallis M (2002) Characterization of the GH gene cluster in a new-world monkey, the marmoset (Callithrix jacchus). J Mol Endocrinol 29:89–97
Wallis OC, Wallis M (2006) Evolution of growth hormone in primates: the GH gene clusters of the New World Monkeys marmoset (Callithrix jacchus) and white-fronted capuchin (Cebus albifrons). J Mol Evol 63:591–601
Wallis OC, Zhang YP, Wallis M (2001) Molecular evolution of GH in primates: characterization of the GH genes from slow loris and marmoset defines an episode of rapid evolutionary change. J Mol Endocrinol 26:249–258
Yang Z (2007) PAML 4: phylogenetic analysis by maximum likelihood. Mol Biol Evol 24:1586–1591
Yang Z, Bielawski JP (2000) Statistical methods for detecting molecular adaptation. Trends Ecol Evol 15:496–503
Zhang J, Nielsen R, Yang Z (2005) Evaluation of an improved branch-site likelihood method for detecting positive selection at the molecular level. Mol Biol Evol 22:2472–2479
Acknowledgments
This research was possible, thanks to grants contributed by CONACYT (Q-43987). We thank Sergio Lozano-Rodriguez, MD, for his critical reading of the manuscript and Irám Sánchez for his help in isolating GH loci-containing BACs from CHORI genome projects BAC resource, accessed thanks to the generosity of Peter de Jong, Ph D.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pérez-Maya, A.A., Wallis, M. & Barrera-Saldaña, H.A. Structure and evolution of the gorilla and orangutan growth hormone loci. Mamm Genome 27, 511–523 (2016). https://doi.org/10.1007/s00335-016-9654-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00335-016-9654-7