Abstract
Objectives
To construct and validate a nomogram model that integrated the CT radiomic features and the TNM staging for risk stratification of thymic epithelial tumors (TETs).
Methods
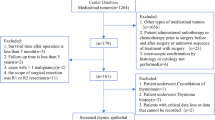
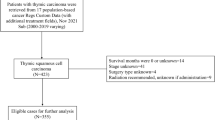
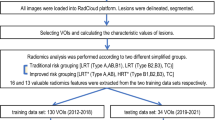
A total of 136 patients with pathology-confirmed TETs who underwent CT examination were collected from two institutions. According to the WHO pathological classification criteria, patients were classified into low-risk and high-risk groups. The TNM staging was determined in terms of the 8th edition AJCC/UICC staging criteria. LASSO regression was performed to extract the optimal features correlated to risk stratification among the 704 radiomic features calculated. A nomogram model was constructed by combining the Radscore and the TNM staging. The clinical performance was evaluated by ROC analysis, calibration curve, and decision curve analysis (DCA). The Kaplan-Meier (KM) analysis was employed for survival analysis.
Results
Five optimal features identified by LASSO regression were employed to calculate the Radscore correlated to risk stratification. The nomogram model showed a better performance in both training cohort (AUC = 0.84, 95%CI 0.75–0.91) and external validation cohort (AUC = 0.79, 95%CI 0.69–0.88). The calibration curve and DCA analysis indicated a better accuracy of the nomogram model for risk stratification than either Radscore or the TNM staging alone. The KM analysis showed a significant difference between the two groups stratified by the nomogram model (p = 0.02).
Conclusions
A nomogram model that integrated the radiomic signatures and the TNM staging could serve as a reliable model of risk stratification in predicting the prognosis of patients with TETs.
Key Points
• The radiomic features could be associated with the TET pathophysiology.
• TNM staging and Radscore could independently stratify the risk of TETs.
• The nomogram model is more objective and more comprehensive than previous methods.







Similar content being viewed by others
Abbreviations
- ADC:
-
Apparent diffusion coefficient
- AJCC:
-
American Joint Committee on Cancer
- AUC:
-
Area under the curve
- DCA:
-
Decision curve analysis
- GLCM:
-
Gray-level co-occurrence matrix
- GLRLM:
-
Gray-level run-length matrix
- GLZSM:
-
Gray-level zone size matrix
- IASLC:
-
International Association for the Study of Lung Cancer
- ICC:
-
Intraclass correlation coefficient
- ITMIG:
-
Thymic malignancy interest group
- KM:
-
Kaplan-Meier
- LASSO:
-
Least Absolute Shrinkage and Selection Operator
- ROC:
-
Receiver operating characteristic
- ROI:
-
Regions of interest
- TC:
-
Thymic carcinomas
- TETs:
-
Thymic epithelial tumors
- UICC:
-
Union for International Cancer Control
- WHO:
-
World Health Organization
References
Engels EA (2010) Epidemiology of thymoma and associated malignancies. J Thorac Oncol 5:S260–S265
Travis WD, Brambilla E, Nicholson AG et al (2015) The 2015 World Health Organization classification of lung tumors: impact of genetic, clinical and radiologic advances since the 2004 classification. J Thorac Oncol 10:1243–1260
Detterbeck FC, Moran C, Huang J et al (2014) Which way is up? Policies and procedures for surgeons and pathologists regarding resection specimens of thymic malignancy. Zhongguo Fei Ai Za Zhi 17:95–103
Filosso PL, Ruffini E, Lausi PO, Lucchi M, Oliaro A, Detterbeck F (2014) Historical perspectives: The evolution of the thymic epithelial tumors staging system. Lung Cancer 83:126–132
Kondo K, Van Schil P, Detterbeck FC et al (2014) The IASLC/ITMIG Thymic epithelial tumors staging project: proposals for the N and M components for the forthcoming (8th) edition of the TNM classification of malignant tumors. J Thorac Oncol 9:S81–S87
Jeong YJ, Lee KS, Kim J, Shim YM, Han J, Kwon OJ (2004) Does CT of thymic epithelial tumors enable us to differentiate histologic subtypes and predict prognosis? AJR Am J Roentgenol 183:283–289
Sadohara J, Fujimoto K, Muller NL et al (2006) Thymic epithelial tumors: comparison of CT and MR imaging findings of low-risk thymomas, high-risk thymomas, and thymic carcinomas. Eur J Radiol 60:70–79
Yakushiji S, Tateishi U, Nagai S et al (2008) Computed tomographic findings and prognosis in thymic epithelial tumor patients. J Comput Assist Tomogr 32:799–805
Shen Q, Hu W, Feng Z (2015) Perplexing histologic classification of thymic epithelial tumor. Radiology 275:929–930
Strobel P, Marx A, Zettl A, Muller-Hermelink HK (2005) Thymoma and thymic carcinoma: an update of the WHO Classification 2004. Surg Today 35:805–811
Masaoka A, Monden Y, Nakahara K, Tanioka T (1981) Follow-up study of thymomas with special reference to their clinical stages. Cancer 48:2485–2492
Koga K, Matsuno Y, Noguchi M et al (1994) A review of 79 thymomas: modification of staging system and reappraisal of conventional division into invasive and non-invasive thymoma. Pathol Int 44:359–367
Carter BW, Benveniste MF, Madan R et al (2017) IASLC/ITMIG Staging system and lymph node map for thymic epithelial neoplasms. Radiographics 37:758–776
Roden AC, Yi ES, Jenkins SM et al (2015) Modified Masaoka stage and size are independent prognostic predictors in thymoma and modified Masaoka stage is superior to histopathologic classifications. J Thorac Oncol 10:691–700
Detterbeck F, Youssef S, Ruffini E, Okumura M (2014) A review of prognostic factors in thymic malignancies. Zhongguo Fei Ai Za Zhi 17:130–136
Li B, Xin YK, Xiao G et al (2019) Predicting pathological subtypes and stages of thymic epithelial tumors using DWI: value of combining ADC and texture parameters. Eur Radiol. https://doi.org/10.1007/s00330-019-06080-4
Iannarelli A, Sacconi B, Tomei F et al (2018) Analysis of CT features and quantitative texture analysis in patients with thymic tumors: correlation with grading and staging. Radiol Med 123:345–350
Nakajo M, Jinguji M, Shinaji T et al (2018) Texture analysis of (18)F-FDG PET/CT for grading thymic epithelial tumours: usefulness of combining SUV and texture parameters. Br J Radiol 91:20170546
Priola AM, Priola SM, Di Franco M, Cataldi A, Durando S, Fava C (2010) Computed tomography and thymoma: distinctive findings in invasive and noninvasive thymoma and predictive features of recurrence. Radiol Med 115:1–21
Chang S, Hur J, Im DJ et al (2017) Volume-based quantification using dual-energy computed tomography in the differentiation of thymic epithelial tumours: an initial experience. Eur Radiol 27:1992–2001
Lee HN, Yun SJ, Kim JI, Ryu CW (2020) Diagnostic outcome and safety of CT-guided core needle biopsy for mediastinal masses: a systematic review and meta-analysis. Eur Radiol 30:588–599
Priola AM, Priola SM, Giraudo MT et al (2016) Diffusion-weighted magnetic resonance imaging of thymoma: ability of the apparent diffusion coefficient in predicting the World Health Organization (WHO) classification and the Masaoka-Koga staging system and its prognostic significance on disease-free survival. Eur Radiol 26:2126–2138
Hayes SA, Huang J, Plodkowski AJ et al (2014) Preoperative computed tomography findings predict surgical resectability of thymoma. J Thorac Oncol 9:1023–1030
Qu YJ, Liu GB, Shi HS, Liao MY, Yang GF, Tian ZX (2013) Preoperative CT findings of thymoma are correlated with postoperative Masaoka clinical stage. Acad Radiol 20:66–72
Padda SK, Terrone D, Tian L et al (2018) Computed tomography features associated with the eighth edition TNM stage classification for thymic epithelial tumors. J Thorac Imaging 33:176–183
Davnall F, Yip CS, Ljungqvist G et al (2012) Assessment of tumor heterogeneity: an emerging imaging tool for clinical practice? Insights Imaging 3:573–589
Ganeshan B, Miles KA (2013) Quantifying tumour heterogeneity with CT. Cancer Imaging 13:140–149
Lambin P, Rios-Velazquez E, Leijenaar R et al (2012) Radiomics: extracting more information from medical images using advanced feature analysis. Eur J Cancer 48:441–446
Shen Q, Shan Y, Hu Z et al (2018) Quantitative parameters of CT texture analysis as potential markersfor early prediction of spontaneous intracranial hemorrhage enlargement. Eur Radiol 28:4389–4396
Lubner MG, Smith AD, Sandrasegaran K, Sahani DV, Pickhardt PJ (2017) CT texture analysis: definitions, applications, biologic correlates, and challenges. Radiographics 37:1483–1503
Thrall JH (2016) Trends and Developments Shaping the Future of Diagnostic Medical Imaging: 2015 Annual Oration in Diagnostic Radiology. Radiology 279:660–666
Li B, Xin YK, Xiao G et al (2019) Predicting pathological subtypes and stages of thymic epithelial tumors using DWI: value of combining ADC and texture parameters. Eur Radiol 29:5330–5340
Xiao G, Rong WC, Hu YC et al (2020) MRI radiomics analysis for predicting the pathologic classification and TNM staging of thymic epithelial tumors: a pilot study. AJR Am J Roentgenol 214:328–340
Marx A, Strobel P, Badve SS et al (2014) ITMIG consensus statement on the use of the WHO histological classification of thymoma and thymic carcinoma: refined definitions, histological criteria, and reporting. J Thorac Oncol 9:596–611
Vasquez MM, Hu C, Roe DJ, Chen Z, Halonen M, Guerra S (2016) Least absolute shrinkage and selection operator type methods for the identification of serum biomarkers of overweight and obesity: simulation and application. BMC Med Res Methodol 16:154
Tomiyama N, Muller NL, Ellis SJ et al (2001) Invasive and noninvasive thymoma: distinctive CT features. J Comput Assist Tomogr 25:388–393
Zhao Y, Chen H, Shi J, Fan L, Hu D, Zhao H (2014) The correlation of morphological features of chest computed tomographic scans with clinical characteristics of thymoma. Eur J Cardiothorac Surg. https://doi.org/10.1093/ejcts/ezu475
Liu GB, Qu YJ, Liao MY, Hu HJ, Yang GF, Zhou SJ (2012) Relationship between computed tomography manifestations of thymic epithelial tumors and the WHO pathological classification. Asian Pac J Cancer Prev 13:5581–5585
Green DB, Eliades S, Legasto AC, Askin G, Port JL, Gruden JF (2019) Multilobulated thymoma with an acute angle: a new predictor of lung invasion. Eur Radiol 29:4555–4562
Inoue A, Tomiyama N, Fujimoto K et al (2006) MR imaging of thymic epithelial tumors: correlation with World Health Organization classification. Radiat Med 24:171–181
Abdel Razek AA, Khairy M, Nada N (2014) Diffusion-weighted MR imaging in thymic epithelial tumors: correlation with World Health Organization classification and clinical staging. Radiology 273:268–275
Priola AM, Priola SM, Parlatano D et al (2017) Apparent diffusion coefficient measurements in diffusion-weighted magnetic resonance imaging of the anterior mediastinum: inter-observer reproducibility of five different methods of region-of-interest positioning. Eur Radiol 27:1386–1394
Priola AM, Priola SM, Gned D, Giraudo MT, Veltri A (2018) Nonsuppressing normal thymus on chemical-shift MR imaging and anterior mediastinal lymphoma: differentiation with diffusion-weighted MR imaging by using the apparent diffusion coefficient. Eur Radiol 28:1427–1437
Masaoka A (2010) Staging system of thymoma. J Thorac Oncol 5:S304–S312
Detterbeck FC, Stratton K, Giroux D et al (2014) The IASLC/ITMIG Thymic Epithelial Tumors Staging Project: proposal for an evidence-based stage classification system for the forthcoming (8th) edition of the TNM classification of malignant tumors. J Thorac Oncol 9:S65–S72
Ried M, Eicher MM, Neu R et al (2018) Comparison of the Masaoka-Koga classification with the new TNM staging of the IASLC/ITMIG for thymoma and thymic carcinoma. Zentralbl Chir 143:S44–S50
Liang G, Gu Z, Li Y et al (2016) Comparison of the Masaoka-Koga and the IASLC/ITMIG proposal for the TNM staging systems based on the Chinese Alliance for Research in Thymomas (ChART) Retrospective Database. Zhongguo Fei Ai Za Zhi 19:425–436
White DB, Hora MJ, Jenkins SM et al (2019) Efficacy of chest computed tomography prediction of the pathological TNM stage of thymic epithelial tumours. Eur J Cardiothorac Surg. https://doi.org/10.1093/ejcts/ezz013
Choe J, Lee SM, Lim S et al (2017) Doubling time of thymic epithelial tumours on CT: correlation with histological subtype. Eur Radiol 27:4030–4036
Acknowledgments
This study was supported in part by Translational Medicine Research Center, Key Laboratory of Clinical Cancer Pharmacology, and Toxicology Research of Zhejiang Province under Grant No. 2020E10021.
Funding
This study has received funding by the Clinical Science Research of Zhejiang University, China (No.YYJJ2019Z06). This study has received funding by the Natural Science Foundation of Zhejiang Province, China (No. LSY19H180009). This study has received funding by the Medical Health Science and Technology Commission of Zhejiang Province, China (No. 2019KY123). This study has received funding by the Medical Health Science and Technology Commission of Zhejiang Province, China (No. 2018KY582).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Guarantor
The scientific guarantor of this publication is Wenli Cai.
Conflict of interest
One of the authors of this manuscript (Peipei Pang) is an employee of GE Healthcare. The remaining authors declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was not required for this study because of the retrospective nature of the study.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• retrospective
• diagnostic or prognostic study
• multicenter study
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Wenli Cai and Zhongxiang Ding are equal contributors to the final manuscript and supervising all the data.
Electronic supplementary material
ESM 1
(DOCX 130 kb)
Rights and permissions
About this article
Cite this article
Shen, Q., Shan, Y., Xu, W. et al. Risk stratification of thymic epithelial tumors by using a nomogram combined with radiomic features and TNM staging. Eur Radiol 31, 423–435 (2021). https://doi.org/10.1007/s00330-020-07100-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-020-07100-4