Abstract
Objectives
The aim of the study was to establish the setup and workflow for delivering focal MRI-guided high-dose-rate (HDR) brachytherapy for prostate cancer (PCA) and to assess patient comfort and safety aspects of MRI-guided single-fraction HDR.
Methods
Patients with histologically proven focal low- to intermediate-risk PCA with a single PIRADS 4/5 lesion were treated with percutaneous interstitial HDR brachytherapy in a single fraction with a minimum dose for the gross tumor volume of 20 Gy while sparing the organ at risk (OAR). Using a 3T-MRI, brachytherapy catheters were placed transgluteal in freehand technique. No antibiotic therapy or general analgesics were administered. Patient data, procedure time, patient discomfort, and complications were recorded. Quarterly PSA controls, biannual follow-up imaging, and annual re-biopsy were planned.
Results
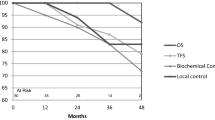
So far, 9 patients were successfully treated and followed for 6 months. Mean intervention time was 34 min. Using the VAS scale, the pain reported for the intervention ranged from 2 to 3. Short-term follow-up showed no acute genitourinary or gastrointestinal toxicity so far. None of the patients displayed signs of infection. PSA levels in all patients decreased significantly. On follow up no residual PCA was detected treated region so far. PSA levels in all patients decreased significantly. On follow-up, no residual PCA was detected so far.
Conclusions
MR-guided single-fraction focal HDR brachytherapy for localized PCA is feasible as well as safe for the individual patient. Catheters can be placed accurately and maximum therapeutic dose distribution can be restricted to the tumor. Countersigning the minimally invasive character of the procedure, no general anesthesia or antibiosis is necessary.
Key Points
• MR-guided focal HDR brachytherapy allows an accurate placement of catheters with maximum therapeutic dose distribution restricted to the tumor.
• No major anesthesia or antibiosis is necessary emphasizing the minimal invasive character of the procedure.
• Patients with low- and intermediate-risk prostate carcinoma in particular may benefit to halt disease progression whereas treatment-related morbidity is reduced compared with radical therapy.




Similar content being viewed by others
Abbreviations
- ACR:
-
American College of Radiology
- AS:
-
Active surveillance
- EBRT:
-
External beam radiation therapy
- GTV:
-
Gross tumor volume
- HDR:
-
High-dose-rate brachytherapy
- HIFU:
-
High-focused ultrasound
- LDR :
-
Low-dose rate brachytherapy
- OAR:
-
Organs at risk
- PCA :
-
Prostate cancer
- PSA :
-
Prostate-specific antigen
- PTV:
-
Planning target volume
- RT:
-
Percutaneous radiotherapy
- VAS:
-
Virtual analog scale
References
Wilt TJ, Brawer MK, Jones KM et al (2012) Radical prostatectomy versus observation for localized prostate cancer. Prostate Cancer Intervention versus Observation Trial (PIVOT) Study Group. N Engl J Med 367:203–213
Hamdy FC, Donovan JL, Lane JA et al (2016) 10-year outcomes after monitoring, surgery, or radiotherapy for localized prostate cancer. ProtecT Study Group. N Engl J Med 375:1415–1424
Bokhorst LP, Valdagni R, Rannikko A et al (2016) PRIAS study group. A decade of active surveillance in the PRIAS study: an update and evaluation of the criteria used to recommend a switch to active treatment. Eur Urol 70:954–960
van der Poel HG, van den Bergh RCN, Briers E et al (2018) Focal therapy in primary localised prostate cancer: the European Association of Urology Position in 2018. Eur Urol 74:84–91
Galalae RM, Martinez A, Mate T et al (2004) Long-term outcome by risk factors using conformal high-dose-rate brachytherapy (HDR-BT) boost with or without neoadjuvant androgen suppression for localized prostate cancer. Int J Radiat Oncol Biol Phys 58:1048–1055
Zamboglou N, Tselis N, Baltas D et al (2012) High-dose-rate interstitial brachytherapy as monotherapy for clinically localized prostate cancer: treatment evolution and mature results. Int J Radiat Oncol Biol Phys 85:672–678
Ricke J, Wust P, Stohlmann A et al (2004) CT-Guided brachytherapy. A novel percutaneous technique for interstitial ablation of liver metastases. Strahlenther Onkol 180:274–280
Ricke J, Thormann M, Ludewig M et al (2010) MR-guided liver tumor ablation employing open high-field 1.0T MRI for image-guided brachytherapy. Eur Radiol 20:1985–1993
Bretschneider T, Peters N, Hass P, Ricke J (2012) Update on interstitial brachytherapy. Radiologe 52:70–73
Fischbach F, Wien L, Krueger S et al (2018) Feasibility study of MR-guided transgluteal targeted in-bore biopsy for suspicious lesions of the prostate at 3 Tesla using a freehand approach. Eur Radiol 28:2690–2699
Vargas HA, Hötker AM, Goldman DA et al (2016) Updated prostate imaging reporting and data system (PIRADS v2) recommendations for the detection of clinically significant prostate cancer using multiparametric MRI: critical evaluation using whole-mount pathology as standard of reference. Eur Radiol 26:1606–1612
Fayers PM (2001) Interpreting quality of life data: population-based reference data for the EORTC QLQ-C30. Eur J Cancer 37:1331–1334
Sacco DE, Daller M, Grocela JA, Babayan RK, Zietman AL (2003) Corticosteroid use after prostate brachytherapy reduces the risk of acute urinary retention. BJU Int 91:345–349
Doyle CA, Loadsman JA, Hruby G (2008) An audit of analgesia requirements for high-dose-rate prostate brachytherapy. Anaesth Intensive Care 36:707–779
Peach MS, Trifiletti DM, Libby B (2016) Systematic review of focal prostate brachytherapy and the future implementation of image-guided prostate HDR brachytherapy using MR-ultrasound fusion. Prostate Cancer. https://doi.org/10.1155/2016/4754031
Demanes DJ, Martinez AA, Ghilezan M et al (2011) High dose rate monotherapy: safe and effective brachytherapy for patients with localized prostate cancer. Int J Radiat Oncol Biol Phys 81:1286–1292
Grills IS, Martinez AA, Hollander M et al (2004) High dose rate brachytherapy as prostate cancer monotherapy reduces toxicity compared to low dose rate palladium seeds. J Urol 171:1098–1104
Sartor AO, Hricak H, Wheeler TM et al (2008) Evaluating localized prostate cancer and identifying candidates for focal therapy. Urology 72:12–24
Liu W, Laitinen S, Khan S et al (2009) Copy number analysis indicates monoclonal origin of lethal metastatic prostate cancer. Nat Med 15:559–565
Haffner MC, Mosbruger T, Esopi DM et al (2013) Tracking the clonal origin of lethal prostate cancer. J Clin Invest 123:4918–4922
Bott SR, Ahmed HU, Hindley RG, Abdul-Rahman A, Freeman A, Emberton M (2010) The index lesion and focal therapy: an analysis of the pathological characteristics of prostate cancer. BJU Int 106:1607–1611
Gburek BM, Kollmorgen TA, Qian J, D'Souza-Gburek SM, Lieber MM, Jenkins RB (1997) Chromosomal anomalies in stage D1 prostate adenocarcinoma primary tumors and lymph node metastases detected by fluorescence in situ hybridization. J Urol 157:223–227
Wise AM, Stamey TA, McNeal JE, Clayton JL (2002) Morphologic and clinical significance of multifocal prostate cancers in radical prostatectomy specimens. Urology 60:264–269
Kasivisvanathan V, Emberton M, Ahmed HU (2013) Focal therapy for prostate cancer: rationale and treatment opportunities. Clin Oncol (R Coll Radiol) 25:461–473
Mouraviev V, Villers A, Bostwick DG, Wheeler TM, Montironi R, Polascik TJ (2011) Understanding the pathological features of focality, grade and tumour volume of early-stage prostate cancer as a foundation for parenchyma-sparing prostate cancer therapies: active surveillance and focal targeted therapy. BJU Int 108:1074–1085
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Frank Fischbach.
Conflict of interest
The co-author, Christian Stehning, is a staff of the company Philips Healthcare.
All other authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• Prospective
• Observational
• Performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fischbach, F., Hass, P., Schindele, D. et al. MRI targeted single fraction HDR Brachytherapy for localized Prostate Carcinoma: a feasibility study of focal radiation therapy (ProFocAL). Eur Radiol 30, 2072–2081 (2020). https://doi.org/10.1007/s00330-019-06505-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-019-06505-0