Abstract
Key message
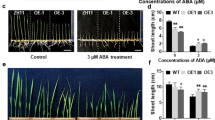
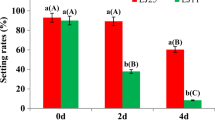
OsERF096 negatively regulates rice cold tolerance and mediates IAA biosynthesis and signaling under cold stress.
Abstract
The APETALA2/ethylene-responsive factor (AP2/ERF) transcription factors play important roles in regulating plant tolerance to abiotic stress. OsERF096 was previously identified as a direct target of miR1320, and was suggested to negatively regulate rice cold tolerance. In this study, we performed RNA-sequencing and targeted metabolomics assays to reveal the regulatory roles of OsERF096 in cold stress response. GO and KEGG analysis of differentially expressed genes showed that the starch and sucrose metabolism, plant–pathogen interaction, and plant hormone signal transduction pathways were significantly enriched. Quantification analysis confirmed a significant difference in sugar contents among WT and OsERF096 transgenic lines under cold treatment. Targeted metabolomics analysis uncovered that IAA accumulation and signaling were modified by OsERF096 in response to cold stress. Expectedly, qRT-PCR assays confirmed significant OsIAAs and OsARFs expression changes in OsERF096 transgenic lines. Finally, we identified three targets of OsERF096 based on RNA-seq, qRT-PCR, and dual-LUC assays. In summary, these results revealed the multiple regulatory roles of OsERF096 in cold stress response.






Similar content being viewed by others
Data availability
The data generated or analyzed during the current study are available from the corresponding author on reasonable request.
References
Aslam M, Sugita K, Qin Y, Rahman A (2020) Aux/IAA14 regulates microrna-mediated cold stress response in Arabidopsis roots. Int J Mol Sci 21:8441
Ba L, Kuang J, Chen J, Lu W (2016) MaJAZ1 attenuates the MaLBD5-mediated transcriptional activation of jasmonate biosynthesis gene MaAOC2 in regulating cold tolerance of banana fruit. J Agric Food Chem 64:738–745
Challam C, Ghosh T, Rai M, Tyagi W (2015) Allele mining across DREB1A and DREB1B in diverse rice genotypes suggest a highly conserved pathway inducible by low temperature. J Genet 94:231–238
Chen C, Chen H, Zhang Y, Thomas HR, Frank MH, He Y, Xia R (2020a) TBtools: an integrative toolkit developed for interactive analyses of big biological data. Mol Plant 13:1194–1202
Chen L, Huang X, Zhao S, Xiao D, Xiao L, Tong J, Wang W, Li Y, Ding Z, Hou B (2020b) IPyA glucosylation mediates light and temperature signaling to regulate auxin-dependent hypocotyl elongation in Arabidopsis. Proc Natl Acad Sci USA 117:6910–6917
Chen H, Chien T, Chen T, Chiang M, Lai M, Chang M (2021) Overexpression of a novel ERF-X-Type transcription factor, OsERF106MZ, reduces shoot growth and tolerance to salinity stress in rice. Rice 14:82
Ding Y, Sun T, Ao K, Peng Y, Zhang Y, Li X, Zhang Y (2018) Opposite roles of salicylic acid receptors NPR1 and NPR3/NPR4 in transcriptional regulation of plant immunity. Cell 173:1454-1467.e1415
Dombrecht B, Xue GP, Sprague SJ, Kirkegaard JA, Ross JJ, Reid JB, Fitt GP, Sewelam N, Schenk PM, Manners JM, Kazan K (2007) MYC2 differentially modulates diverse jasmonate-dependent functions in Arabidopsis. Plant Cell 19:2225–2245
Du H, Wu N, Fu J, Wang S, Li X, Xiao J, Xiong L (2012) A GH3 family member, OsGH3-2, modulates auxin and abscisic acid levels and differentially affects drought and cold tolerance in rice. J Exp Bot 63:6467–6480
Du H, Wu N, Chang Y, Li X, Xiao J, Xiong L (2013) Carotenoid deficiency impairs ABA and IAA biosynthesis and differentially affects drought and cold tolerance in rice. Plant Mol Biol 83:475–488
Dubois M, Gilles KA, Hamilton JK, Rebers PA (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Fonseca S, Fernández-Calvo P, Fernández GM, Díez-Díaz M, Gimenez-Ibanez S, López-Vidriero I, Godoy M, Fernández-Barbero G, Van Leene J, De Jaeger G, Franco-Zorrilla JM, Solano R (2014) bHLH003, bHLH013 and bHLH017 are new targets of jaz repressors negatively regulating JA responses. PLoS ONE 9:e86182
Fu J, Yu Q, Zhang C, Xian B, Fan J, Huang X, Yang W, Zou X, Chen S, Su L, He Y, Li Q (2023) CsAP2-09 confers resistance against citrus bacterial canker by regulating CsGH3.1L-mediated phytohormone biosynthesis. Int J Biol Macromol 229:964–973
Hong Y, Wang H, Gao Y, Bi Y, Xiong X, Yan Y, Wang J, Li D, Song F (2022) ERF transcription factor OsBIERF3 positively contributes to immunity against fungal and bacterial diseases but negatively regulates cold tolerance in rice. Int J Mol Sci 23:606
Hu Y, Jiang L, Wang F, Yu D (2013) Jasmonate regulates the inducer of cbf expression–c-repeat binding factor/DRE binding factor1 cascade and freezing tolerance in Arabidopsis. Plant Cell 25:2907–2924
Jackson RG, Lim E-K, Li Y, Kowalczyk M, Sandberg G, Hoggett J, Ashford DA, Bowles DJ (2001) Identification and biochemical characterization of an Arabidopsis indole-3-acetic acid glucosyltransferase. J Biol Chem 276:4350–4356
Jisha V, Dampanaboina L, Vadassery J, Mithöfer A, Kappara S, Ramanan R (2015) Overexpression of an AP2/ERF type transcription factor OsEREBP1 confers biotic and abiotic stress tolerance in rice. PLoS ONE 10:e0127831
Jung H, Chung PJ, Park S, Redillas MCFR, Kim YS, Suh J, Kim J (2017) Overexpression of OsERF48 causes regulation of OsCML16, a calmodulin-like protein gene that enhances root growth and drought tolerance. Plant Biotechnol J 15:1295–1308
Ke Y, Yang Z, Yu S, Li T, Wu J, Gao H, Fu Y, Luo L (2014) Characterization of OsDREB6 responsive to osmotic and cold stresses in rice. J Plant Biol 57:150–161
Kumar M, Kesawat MS, Ali A, Lee SC, Gill SS, Kim AHU (2019) Integration of abscisic acid signaling with other signaling pathways in plant stress responses and development. Plants 8:592
Leisner CP, Potnis N, Sanz-Saez A (2022) Crosstalk and trade-offs: plant responses to climate change-associated abiotic and biotic stresses. Plant Cell Environ 46(10):2946–2963
Li W, Zhang M, Qiao L, Chen Y, Zhang D, Jing X, Gan P, Huang Y, Gao J, Liu W, Shi C, Cui H, Li H, Chen K (2022) Characterization of wavy root 1, an agravitropism allele, reveals the functions of OsPIN2 in fine regulation of auxin transport and distribution and in ABA biosynthesis and response in rice (Oryza sativa L.). Crop J 10:980–992
Liu D, Chen X, Liu J, Ye J, Guo Z (2012) The rice ERF transcription factor OsERF922 negatively regulates resistance to Magnaporthe oryzae and salt tolerance. J Exp Bot 63:3899–3911
Liu CT, Wang W, Mao BG, Chu CC (2018) Cold stress tolerance in rice: physiological changes, molecular mechanism, and future prospects. Hereditas 40:171–185
Liu C, Li C, Bing H, Zhao J, Li L, Sun P, Li T, Du D, Zhao J, Wang X, Xiang W (2023) Integrated physiological, transcriptomic, and metabolomic analysis reveals the mechanism of guvermectin promoting seed germination in direct-seeded rice under chilling stress. J Agric Food Chem 71:7348–7358
Luo J, Zhou J, Zhang J (2018) Aux/IAA gene family in plants: Molecular structure, regulation, and function. Int J Mol Sci 19:259
Madore MA (1990) Carbohydrate metabolism in photosynthetic and nonphotosynthetic tissues of variegated leaves of coleus blumei benth. Plant Physiol 93:617–622
Mao D, Chen C (2012) Colinearity and similar expression pattern of rice DREB1s reveal their functional conservation in the cold-responsive pathway. PLoS ONE 7:e47275
Mao X, Cai T, Olyarchuk JG, Wei L (2005) Automated genome annotation and pathway identification using the KEGG Orthology (KO) as a controlled vocabulary. Bioinformatics 21:3787–3793
Mizoi J, Shinozaki K, Yamaguchi-Shinozaki K (2012) AP2/ERF family transcription factors in plant abiotic stress responses. Biochim Biophys Acta 1819:86–96
Moon S-J, Min MK, Kim J-A, Kim DY, Yoon IS, Kwon TR, Byun MO, Kim B-G (2019) Ectopic expression of OsDREB1G, a member of the OsDREB1 subfamily, confers cold stress tolerance in rice. Front Plant Sci 10:297
Nakano T, Suzuki K, Fujimura T, Shinshi H (2006) Genome-wide analysis of the ERF gene family in Arabidopsis and rice. Plant Physiol 140:411–432
Nayyar H, Bains T, Kumar S (2005) Low temperature induced floral abortion in chickpea: relationship to abscisic acid and cryoprotectants in reproductive organs. Environ Exp Bot 53:39–47
Niu Y, Fan S, Cheng B, Li H, Wu J, Zhao H, Huang Z, Yan F, Qi B, Zhang L, Zhang G (2023) Comparative transcriptomics and co-expression networks reveal cultivar-specific molecular signatures associated with reproductive-stage cold stress in rice. Plant Cell Rep 42:707–722
Olate E, Jiménez-Gómez JM, Holuigue L, Salinas J (2018) NPR1 mediates a novel regulatory pathway in cold acclimation by interacting with HSFA1 factors. Nat Plants 4:811–823
Pillai SE, Kumar C, Dasgupta M, Kumar BK, Vungarala S, Patel HK, Sonti RV (2020) Ectopic expression of a cell-wall-degrading enzyme-induced OsAP2/ERF152 leads to resistance against bacterial and fungal infection in Arabidopsis. Phytopathology 110:726–733
Rahman A (2013) Auxin: a regulator of cold stress response. Physiol Plant 147:28–35
Rashid M, Guangyuan H, Guangxiao Y, Hussain J, Xu Y (2012) AP2/ERF transcription factor in rice: genome-wide canvas and syntenic relationships between monocots and eudicots. Evol Bioinform Online 8:321–355
Ritonga FN, Ngatia JN, Wang Y, Khoso MA, Farooq U, Chen S (2021) AP2/ERF, an important cold stress-related transcription factor family in plants: a review. Physiol Mol Biol Plants 27:1953–1968
Ruan J, Zhou Y, Zhou M, Yan J, Khurshid M, Weng W, Cheng J, Zhang K (2019) Jasmonic acid signaling pathway in plants. Int J Mol Sci 20:2479
Sakuma Y, Liu Q, Dubouzet JG, Abe H, Shinozaki K, Yamaguchi-Shinozaki K (2002) DNA-binding specificity of the ERF/AP2 domain of Arabidopsis DREBs, transcription factors involved in dehydration- and cold-inducible gene expression. Biochem Biophys Res Commun 290:998–1009
Schmidt R, Mieulet D, Hubberten HM, Obata T, Hoefgen R, Fernie AR, Fisahn J, San Segundo B, Guiderdoni E, Schippers JHM, Mueller-Roeber B (2013) Salt-responsive ERF1 regulates reactive oxygen species–dependent signaling during the initial response to salt stress in rice. Plant Cell 25:2115–2131
Serra TS, Figueiredo DD, Cordeiro AM, Almeida DM, Lourenço T, Abreu IA, Sebastián A, Fernandes L, Contreras-Moreira B, Oliveira MM, Saibo NJM (2013) OsRMC, a negative regulator of salt stress response in rice, is regulated by two AP2/ERF transcription factors. Plant Mol Biol 82:439–455
Sheng Y (2015) Function analysis of stress tolerance gene OsSTAP1 in rice. Master, University of Chinese Academy of Agricultural Sciences
Shi Y, Yang S (2015) COLD1: a cold sensor in rice. Sci China Life Sci 58:409–410
Sun M, Shen Y, Chen Y, Wang Y, Cai X, Yang J, Jia B, Dong W, Chen X, Sun X (2022) Osa-miR1320 targets the ERF transcription factor OsERF096 to regulate cold tolerance via JA-mediated signaling. Plant Physiol 189:2500–2516
Szerszen JB, Szczyglowski K, Bandurski RS (1994) iaglu, a gene from zea mays involved in conjugation of growth hormone indole-3-acetic acid. Science 265:1699–1701
Thines B, Katsir L, Melotto M, Niu Y, Mandaokar A, Liu G, Nomura K, He SY, Howe GA, Browse J (2007) JAZ repressor proteins are targets of the SCFCOI1 complex during jasmonate signalling. Nature 448:661–665
Tognetti VB, Van Aken O, Morreel K, Vandenbroucke K, van de Cotte B, De Clercq I, Chiwocha S, Fenske R, Prinsen E, Boerjan W, Genty B, Stubbs KA, Inzé D, Van Breusegem F (2010) Perturbation of indole-3-butyric acid homeostasis by the UDP-Glucosyltransferase UGT74E2 modulates arabidopsis architecture and water stress tolerance. Plant Cell 22:2660–2679
Tunsagool P, Jutidamrongphan W, Phaonakrop N, Jaresitthikunchai J, Roytrakul S, Leelasuphakul W (2019) Insights into stress responses in mandarins triggered by Bacillus subtilis cyclic lipopeptides and exogenous plant hormones upon Penicillium digitatum infection. Plant Cell Rep 38:559–575
Wan L, Zhang J, Zhang H, Zhang Z, Quan R, Zhou S, Huang R (2011) Transcriptional activation of OsDERF1 in OsERF3 and OsAP2-39 negatively modulates ethylene synthesis and drought tolerance in rice. PLoS ONE 6:e25216
Wang Q, Guan Y, Wu Y, Chen H, Chen F, Chu C (2008) Overexpression of a rice OsDREB1F gene increases salt, drought, and low temperature tolerance in both Arabidopsis and rice. Plant Mol Biol 67:589–602
Wang F, Wang C, Liu P, Lei C, Hao W, Gao Y, Liu Y, Zhao K (2016) Enhanced rice blast resistance by CRISPR/Cas9-targeted mutagenesis of the ERF transcription factor gene OsERF922. PLoS ONE 11:e0154027
Wang H, Li Y, Chern M, Zhu Y, Zhang L, Lu J, Li X, Dang W, Ma X, Yang Z, Yao S, Zhao Z, Fan J, Huang Y, Zhang J, Pu M, Wang J, He M, Li W, Chen X, Wu X, Li S, Li P, Li Y, Ronald PC, Wang W (2021a) Suppression of rice miR168 improves yield, flowering time and immunity. Nat Plants 7:129–136
Wang X, Pan L, Wang Y, Meng J, Deng L, Niu L, Liu H, Ding Y, Yao J-L, Nieuwenhuizen NJ, Ampomah-Dwamena C, Lu Z, Cui G, Wang Z, Zeng W (2021b) PpIAA1 and PpERF4 form a positive feedback loop to regulate peach fruit ripening by integrating auxin and ethylene signals. Plant Sci 313:111084
Wang Y (2013) Cloning and functional identification of four stress-induced genes in rice. Master, University of Huazhong Agricultural Unversity
Xiong H, Yu J, Miao J, Li J, Zhang H, Wang X, Liu P, Zhao Y, Jiang C, Yin Z, Li Y, Guo Y, Fu B, Wang W, Li Z, Ali J, Li Z (2018) Natural variation in OsLG3 increases drought tolerance in rice by inducing ROS scavenging. Plant Physiol 178:451–467
Yao Y, He RJ, Xie QL, Zhao Xh, Xm D, Jb He, Song L, He J, Marchant A, Chen X, Wu A (2017) ETHYLENE RESPONSE FACTOR 74 (ERF74) plays an essential role in controlling a respiratory burst oxidase homolog D (RbohD)-dependent mechanism in response to different stresses in Arabidopsis. New Phytol 213:1667–1681
Young MD, Wakefield MJ, Smyth GK, Oshlack A (2010) Gene ontology analysis for RNA-seq: accounting for selection bias. Genome Biol 11:R14
Zhang Y, Chen C, Jin XF, Xiong AS, Peng RH, Hong YH, Yao QH, Chen JM (2009) Expression of a rice DREB1 gene, OsDREB1D, enhances cold and high-salt tolerance in transgenic Arabidopsis. BMB Rep 42:486–492
Zhang H, Zhang J, Quan R, Pan X, Wan L, Huang R (2013) EAR motif mutation of rice OsERF3 alters the regulation of ethylene biosynthesis and drought tolerance. Planta 237:1443–1451
Zhang Y, Li J, Chen S, Ma X, Wei H, Chen C, Gao N, Zou Y, Kong D, Li T, Liu Z, Yu S, Luo L (2020) An APETALA2/ethylene responsive factor, OsEBP89 knockout enhances adaptation to direct-seeding on wet land and tolerance to drought stress in rice. Mol Genet Genom 295:941–956
Funding
This work was supported by the National Natural Science Foundation of China (32101672, U20A2025), and Key Research and Development Plan of Heilongjiang Province (GZ2022ZX02B05-2).
Author information
Authors and Affiliations
Contributions
XS and MS conceived and designed the experiment. XC and YC completed most of the experiments and wrote the main manuscript. YW prepared the materials for the experiment. YS and JY analyzed experimental data. BJ plotted the resulting images, Figs. 1, 2, 3, 4, 5, 6. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interests
The authors declare that they have no conflicts of interests.
Additional information
Communicated by Leandro Peña.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cai, X., Chen, Y., Wang, Y. et al. A comprehensive investigation of the regulatory roles of OsERF096, an AP2/ERF transcription factor, in rice cold stress response. Plant Cell Rep 42, 2011–2022 (2023). https://doi.org/10.1007/s00299-023-03079-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-023-03079-6