Abstract
Objective
We aimed to investigate the efficacy of anti-IL-6 receptor antibody (aIL-6) and other biologic disease-modifying antirheumatic drugs (bDMARDs), such as TNF inhibitor and CTLA4-Ig in the treatment of rheumatoid arthritis (RA) in patients with knee joint involvement.
Methods
We retrospectively analyzed 1059 treatment courses of patients with RA who visited our hospitals and were treated with bDMARDs. We categorized them into two groups, with or without knee joint involvement. We investigated the clinical disease activity index (CDAI) at baseline and 12 weeks after the initiation of bDMARDs. We compared the improvement of the markers between aIL-6 and other bDMARDs.
Results
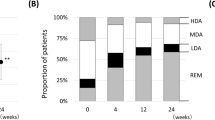
Treatment with aIL-6 significantly increased ΔCDAI (n = 91, 15.4 ± 1.1; mean ± SEM) in patients with knee joint involvement, compared to other bDMARDs (n = 232, 11.0 ± 0.7) at 12 weeks (P = 0.006). Following the multivariate analysis adjusted by the CDAI levels at baseline, age, gender, concomitant use of methotrexate, and the first use of bDMARDs, ΔCDAI levels were significantly higher in aIL-6, compared to other bDMARDs (P = 0.02). However, there was no significant difference in ΔCDAI improvement between aIL-6 (n = 162, 5.9 ± 0.6) and other bDMARDs (n = 573, 6.2 ± 0.4) in patients without swollen knee joints. ΔCDAI levels were equally increased in patients with shoulder and elbow joint involvement.
Conclusion
aIL-6 was more effective in the patients with RA and knee joint involvement, compared to other bDMARDs.



Similar content being viewed by others
References
Scott DL, Wolfe F, Huizinga TW (2010) Rheumatoid arthritis. Lancet 376(9746):1094–1108. https://doi.org/10.1016/s0140-6736(10)60826-4
Maeda Y, Kurakawa T, Umemoto E, Motooka D, Ito Y, Gotoh K et al (2016) Dysbiosis contributes to arthritis development via activation of autoreactive T cells in the intestine. Arthritis Rheumatol 68(11):2646–2661. https://doi.org/10.1002/art.39783
Kishikawa T, Maeda Y, Nii T, Motooka D, Matsumoto Y, Matsushita M et al (2020) Metagenome-wide association study of gut microbiome revealed novel aetiology of rheumatoid arthritis in the Japanese population. Ann Rheum Dis 79(1):103–111. https://doi.org/10.1136/annrheumdis-2019-215743
Okada Y, Wu D, Trynka G, Raj T, Terao C, Ikari K et al (2014) Genetics of rheumatoid arthritis contributes to biology and drug discovery. Nature 506(7488):376–381. https://doi.org/10.1038/nature12873
Hashimoto M, Yamazaki T, Hamaguchi M, Morimoto T, Yamori M, Asai K et al (2015) Periodontitis and Porphyromonas gingivalis in preclinical stage of arthritis patients. PLoS ONE 10(4):e0122121. https://doi.org/10.1371/journal.pone.0122121
Nishimoto N, Hashimoto J, Miyasaka N, Yamamoto K, Kawai S, Takeuchi T et al (2007) Study of active controlled monotherapy used for rheumatoid arthritis, an IL-6 inhibitor (SAMURAI): evidence of clinical and radiographic benefit from an X ray reader-blinded randomised controlled trial of tocilizumab. Ann Rheum Dis 66(9):1162–1167. https://doi.org/10.1136/ard.2006.068064
Aletaha D, Smolen JS (2018) Diagnosis and management of rheumatoid arthritis: a review. JAMA 320(13):1360–1372. https://doi.org/10.1001/jama.2018.13103
Narazaki M, Tanaka T, Kishimoto T (2017) The role and therapeutic targeting of IL-6 in rheumatoid arthritis. Expert Rev Clin Immunol 13(6):535–551. https://doi.org/10.1080/1744666x.2017.1295850
Bijlsma JWJ, Welsing PMJ, Woodworth TG, Middelink LM, Pethö-Schramm A, Bernasconi C et al (2016) Early rheumatoid arthritis treated with tocilizumab, methotrexate, or their combination (U-Act-Early): a multicentre, randomised, double-blind, double-dummy, strategy trial. Lancet 388(10042):343–355. https://doi.org/10.1016/s0140-6736(16)30363-4
Linn-Rasker SP, van der Helm-van Mil AH, Breedveld FC, Huizinga TW (2007) Arthritis of the large joints—in particular, the knee—at first presentation is predictive for a high level of radiological destruction of the small joints in rheumatoid arthritis. Ann Rheum Dis 66(5):646–650. https://doi.org/10.1136/ard.2006.066704
Nakagawa J, Koyama Y, Kawakami A, Ueki Y, Tsukamoto H, Horiuchi T et al (2017) A novel scoring system based on common laboratory tests predicts the efficacy of TNF-inhibitor and IL-6 targeted therapy in patients with rheumatoid arthritis: a retrospective, multicenter observational study. Arthritis Res Ther 19(1):185. https://doi.org/10.1186/s13075-017-1387-9
Lansbury J, Haut DD (1956) Quantitation of the manifestations of rheumatoid arthritis. 4. Area of joint surfaces as an index to total joint inflammation and deformity. Am J Med Sci. 232(2):150–155
Hashimoto M, Furu M, Yamamoto W, Fujimura T, Hara R, Katayama M et al (2018) Factors associated with the achievement of biological disease-modifying antirheumatic drug-free remission in rheumatoid arthritis: the ANSWER cohort study. Arthritis Res Ther 20(1):165. https://doi.org/10.1186/s13075-018-1673-1
Ebina K, Hirano T, Maeda Y, Yamamoto W, Hashimoto M, Murata K et al (2020) Drug retention of 7 biologics and tofacitinib in biologics-naïve and biologics-switched patients with rheumatoid arthritis: the ANSWER cohort study. Arthritis Res Ther 22(1):142. https://doi.org/10.1186/s13075-020-02232-w
Ebina K, Hirano T, Maeda Y, Yamamoto W, Hashimoto M, Murata K et al (2020) Drug retention of secondary biologics or JAK inhibitors after tocilizumab or abatacept failure as first biologics in patients with rheumatoid arthritis—the ANSWER cohort study. Clin Rheumatol 39(9):2563–2572. https://doi.org/10.1007/s10067-020-05015-5
Jinno S, Onishi A, Dubreuil M, Akashi K, Hashimoto M, Yamamoto W et al (2020) Comparison of the efficacy and safety of biologic agents between elderly-onset and young-onset RA patients: the ANSWER cohort study. Rheumatol Int. https://doi.org/10.1007/s00296-020-04660-y
Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS et al (1988) The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 31(3):315–324. https://doi.org/10.1002/art.1780310302
Aletaha D, Neogi T, Silman AJ, Funovits J, Felson DT, Bingham CO III et al (2010) 2010 Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum 62(9):2569–2581. https://doi.org/10.1002/art.27584
Thompson PW, Silman AJ, Kirwan JR, Currey HL (1987) Articular indices of joint inflammation in rheumatoid arthritis. Correlation with the acute-phase response. Arthritis Rheum 30(6):618–623. https://doi.org/10.1002/art.1780300603
Lansbury J (1968) Clinical appraisal of the activity index as a measure of rheumatoid activity. Arthritis Rheum 11(4):599–604. https://doi.org/10.1002/art.1780110411
Holt I, Cooper RG, Hopkins SJ (1991) Relationships between local inflammation, interleukin-6 concentration and the acute phase protein response in arthritis patients. Eur J Clin Investig 21(5):479–484. https://doi.org/10.1111/j.1365-2362.1991.tb01398.x
Chen DY, Hsieh TY, Chen YM, Hsieh CW, Lan JL, Lin FJ (2009) Proinflammatory cytokine profiles of patients with elderly-onset rheumatoid arthritis: a comparison with younger-onset disease. Gerontology 55(3):250–258. https://doi.org/10.1159/000164393
Yoshihara Y, Obata K, Fujimoto N, Yamashita K, Hayakawa T, Shimmei M (1995) Increased levels of stromelysin-1 and tissue inhibitor of metalloproteinases-1 in sera from patients with rheumatoid arthritis. Arthritis Rheum 38(7):969–975. https://doi.org/10.1002/art.1780380713
Sasaki S, Iwata H, Ishiguro N, Obata K, Miura T (1994) Detection of stromelysin in synovial fluid and serum from patients with rheumatoid arthritis and osteoarthritis. Clin Rheumatol 13(2):228–233. https://doi.org/10.1007/bf02249017
Gorai M, Ogasawara M, Matsuki Y, Yamada Y, Murayama G, Sugisaki N et al (2014) Weighting with the Lansbury Articular Index improves the correlation of ultrasound score with serum matrix metalloproteinase-3 level in rheumatoid arthritis patients. Mod Rheumatol 24(6):915–919. https://doi.org/10.3109/14397595.2014.888794
Lipina M, Makarov M, Makarov S, Novikov A (2017) The degree of cartilage degradation assessed by serum biomarker levels changes after arthroscopic knee synovectomy in rheumatoid arthritis patients. Int Orthop 41(11):2259–2264. https://doi.org/10.1007/s00264-017-3634-8
Kobayashi A, Naito S, Enomoto H, Shiomoi T, Kimura T, Obata K et al (2007) Serum levels of matrix metalloproteinase 3 (stromelysin 1) for monitoring synovitis in rheumatoid arthritis. Arch Pathol Lab Med 131(4):563–570. https://doi.org/10.1043/1543-2165(2007)131[563:Slomms]2.0.Co;2
Wang J, Devenport J, Low JM, Yu D, Hitraya E (2016) Relationship between baseline and early changes in C-reactive protein and interleukin-6 levels and clinical response to tocilizumab in rheumatoid arthritis. Arthritis Care Res (Hoboken) 68(6):882–885. https://doi.org/10.1002/acr.22765
Zhang W, Doherty M, Peat G, Bierma-Zeinstra MA, Arden NK, Bresnihan B et al (2010) EULAR evidence-based recommendations for the diagnosis of knee osteoarthritis. Ann Rheum Dis 69(3):483–489. https://doi.org/10.1136/ard.2009.113100
Hussain SM, Neilly DW, Baliga S, Patil S, Meek R (2016) Knee osteoarthritis: a review of management options. Scott Med J 61(1):7–16. https://doi.org/10.1177/0036933015619588
Acknowledgements
We thank all the physicians who evaluated detailed joint findings at Kyoto University Hospital, Osaka Medical College Hospital, Osaka Red Cross Hospital, Kansai Medical University Hospital, Kobe University Hospital, Nara Medical University Hospital and Osaka University Hospital. English language editing was performed by editage.
Funding
ANSWER Cohort is supported by grants from pharmaceutical companies (Abbie G.K., Asahi-Kasei Pharma, AYUMI Pharmaceutical Co., Chugai Pharmaceutical Co. Ltd., Eisai Co. Ltd., Janssen Pharmaceutical K.K., Ono Pharmaceutical Co., Sanofi, UCB Japan Co. Ltd., Teijin Healthcare Limited) and an information technology services company (CAC). These companies had no roles in study design, data collection, data analysis, data interpretation or writing of the manuscript.
Author information
Authors and Affiliations
Contributions
Conceived and designed the study: YM. Analyzed the data: YM, collection of data: YM, TH, KE, RH, MH, WY, KM, TK, KH, YS, HA, AO, SJ, MK, AK. YM prepared the initial draft of the manuscript. All the authors were involved in revising the manuscript critically for content. All authors read and approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of interest
YM received a research grant and/or speaker fee from Eli Lilly Japan K.K., Chugai Pharmaceutical Co. Ltd., Pfizer Inc., Bristol Myers Squibb, and Mitsubishi Tanabe Pharma Corporation. TH received a speaker fee from GlaxoSmithKline, Chugai, Eisai, Eli Lilly and NIPPON SHINYAKU. KE is affiliated with the department, which is supported by Taisho. KE has received research grants from Abbie, Asahi-Kasei, Astellas, Chugai, Eisai, Ono Pharmaceutical, and UCB Japan. K.E. has received payments for lectures from Abbie, Asahi-Kasei, Astellas, AYUMI, Bristol-Myers Squibb, Chugai, Eisai, Eli Lilly, Janssen, Mitsubishi Tanabe, Ono Pharmaceutical, Sanofi and UCB Japan. MH belong to the department that is financially supported by Nagahama City, Shiga, Japan, Toyooka City, Hyogo, Japan and five pharmaceutical companies (Mitsubishi-Tanabe, Chugai, UCB Japan, AYUMI and Asahi-Kasei). MH has received a research and/or speaker fee from Bristol-Meyers, Eisai, Ely Lilly, and Tanabe-Mitsubishi. JS reports personal fees from Mitsubishi Tanabe Pharma Corporation, personal fees from Chugai Pharmaceutical Co. Ltd, personal fees from Asahi-Kasei Corporation, personal fees from Eli Lilly and Company, personal fees from AbbVie GK. KM received a research grant from Eisai. AO received a speaker fee from Chugai, Ono Pharmaceutical, Eli Lilly, Mitsubishi Tanabe, Asahi-Kasei and Takeda. AK received a research grant and/or speaker fee from Mitsubishi-Tanabe, Chugai, Eisai, Asahi-Kasei, Astellas, Abbie, Bristol-Myers Squibb, Ono Pharmaceutical and Pfizer.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Maeda, Y., Hirano, T., Ebina, K. et al. Comparison of efficacy between anti-IL-6 receptor antibody and other biological disease-modifying antirheumatic drugs in the patients with rheumatoid arthritis who have knee joint involvement: the ANSWER cohort, retrospective study. Rheumatol Int 41, 1233–1241 (2021). https://doi.org/10.1007/s00296-021-04862-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-021-04862-y